This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates the ability of QuanRecovery Sample Plates with MaxPeak HPS (High Performance Surfaces) to mitigate non-specific binding of monoclonal antibody biotherapeutics and improve analytical method performance.
Large, hydrophobic biomolecules are prone to non-specific binding (NSB). Ensuring labware is designed to mitigate NSB can greatly improve assay performance.
Biotherapeutics are becoming more complex as they are engineered to improve efficacy at safer dosage levels.¹ To support development of these drugs, more sensitive and selective assays are required to meet needed limits of detection. Larger hydrophobic biomolecules, such as monoclonal antibodies (mAbs), often suffer from non-specific binding (NSB) or adsorption to any labware that samples come into contact with (e.g., plates, vials, pipette tips).² This phenomena is dependent on the physicochemical properties of the molecules and typically increases in severity at low concentrations. This can lead to poor recovery, loss of analyte, and poor assay reproducibility, ultimately leading to insufficient limits of detection. It is therefore important to mitigate the effects of NSB wherever possible. A commonly used approach to combat NSB is to add carrier proteins into samples. Carrier proteins will coat the surfaces that samples come in contact with and they decrease the amount of the molecule of interest that is adsorbed to these surfaces. Although generally effective, this strategy adds unwanted complexity back into the samples. To minimize the need for this, samples can be prepared and stored prior to LC-MS/MS analysis in sample storage containers with high performance surfaces to mitigate the effects of NSB. Waters MaxPeak HPS are new and innovative technologies designed to increase analyte recovery and reproducibility by minimizing analyte/surface interactions that can lead to sample losses.
In this study, QuanRecovery Sample Plates with MaxPeak HPS (p/n: 186009184) were used to mitigate non-specific binding of several mAbs prior to LC-MS/MS analysis. Quantification of adalimumab, cetuximab, and the NISTmAb (RM 8671) in the Humanized mAb Mass Check Standard (p/n: 186009125), was performed at the subunit light chain level using a Xevo TQ-XS Mass Spectrometer and a common MS/MS fragment (1329.85 m/z) from the conserved region of the mAb light chains. Chromatographic separation was achieved using an ACQUITY UPLC I-Class PLUS System with a BioResolve RP mAb Polyphenyl, 450 Å, 2.7 µm, 2.1 x 50 mm Column (p/n: 186008944). A shallow gradient of 25–30% B over 2.5 minutes (flow rate 0.4 mL/min) was used to separate biotherapeutics with mobile phases of water and acetonitrile containing 0.1% formic acid.
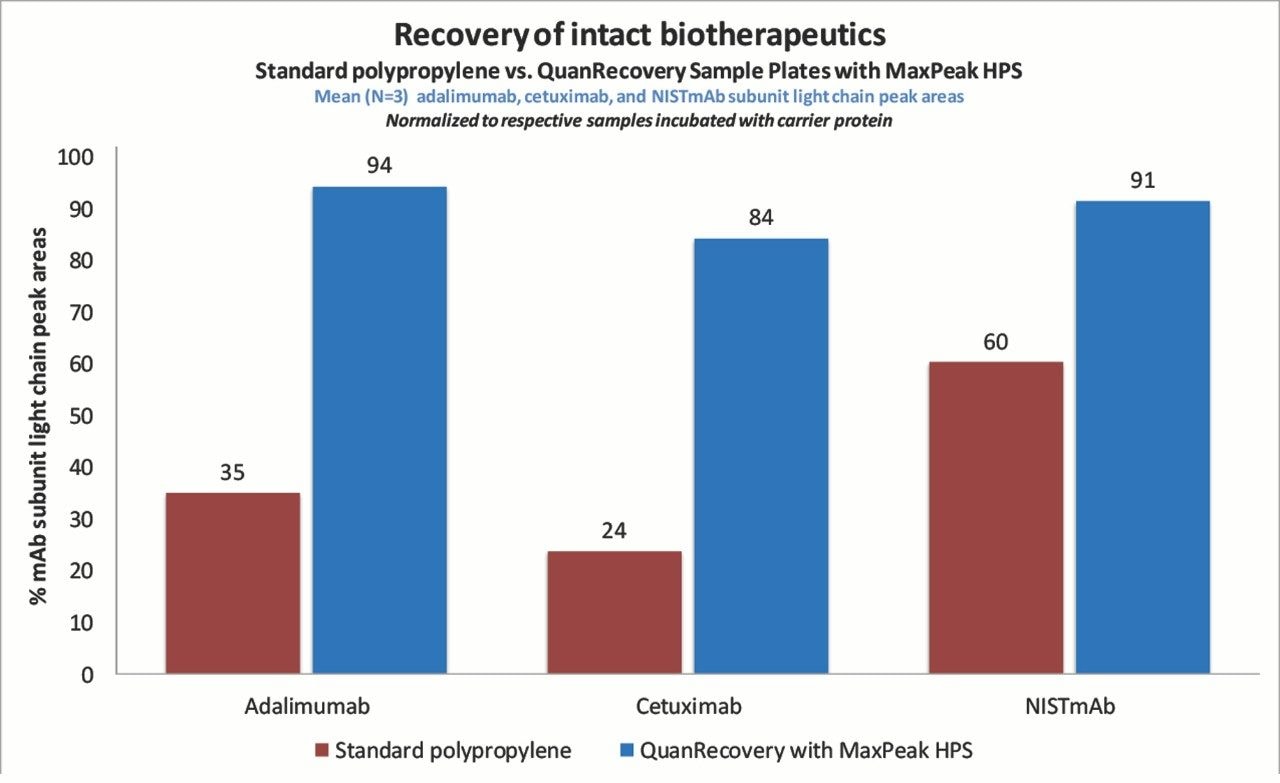
During method development, the three mAbs were stored in either a QuanRecovery Sample Plate or a standard polypropylene plate in order to assess the plates’ effectiveness in mitigating NSB at the intact mAb level. Test samples (100 ng/mL) were prepared and stored in neat solution, while additional samples prepared in a solution containing carrier proteins were used as a benchmark for 100% recovery. Shown in Figure 1, samples stored in QuanRecovery Plates for 24 hours experienced very little NSB with 84–94% recovery, while samples stored in a standard polypropylene plate suffered from variable levels of NSB ranging from only 23–60% recovery.
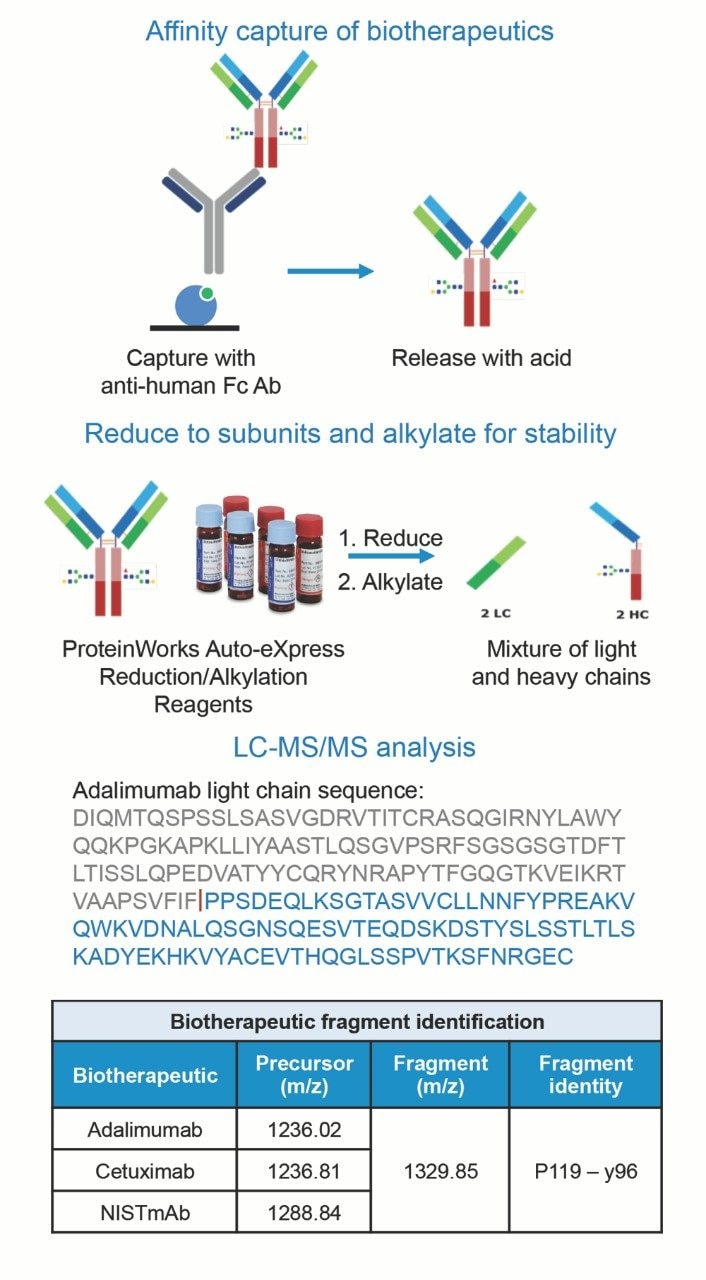
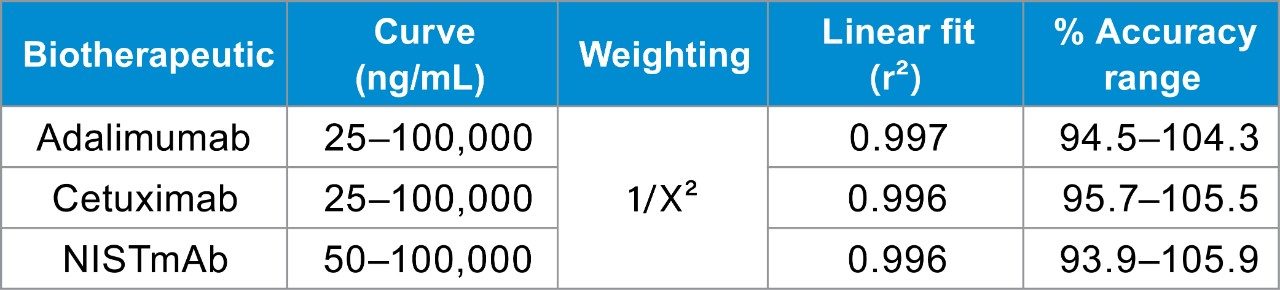
With these results in mind, and as further demonstration of the QuanRecovery Plate's performance, each of the three mAbs were extracted from rat plasma and the resulting samples were stored in QuanRecovery Plates prior to LC-MS/MS analysis. Highlighted in Figure 2, biotherapeutics were immunopurified from rat plasma (10 µL) using biotinylated anti-human Fc Ab (15 µL) conjugated to streptavidin coated magnetic beads (25 µL). Affinity purified samples (50 µL) were reduced and alkylated using the ProteinWorks Reduction Alkylation Kit (p/n: 186008889). Samples were reduced to light and heavy chains with dithiothreitol, then alkylated with iodoacetamide for stability. A 10 µL aliquot of the resulting 70 µL of sample was injected directly from a QuanRecovery Plate for each LC-MS/MS analysis. With these optimized sample preparation methods, storage conditions, and LC-MS/MS methods, LLOQs of 25–50 ng/mL were achieved for all three study mAbs. Calibration curves were linear (r² >0.99) from 25–100,000 ng/mL and statistics indicate excellent accuracy (±15%) and precision with CVs <10% (Table 1). Although the level of NSB mitigation experienced using QuanRecovery Sample Plates is mAb specific, there is a clear benefit to using this technology for this type of molecule, particularly at low protein concentrations.
Care must be taken to mitigate the effects of NSB when working with larger molecules prone to adsorption. QuanRecovery Sample Plates with MaxPeak High Performance Surfaces have been shown to mitigate, and in some cases eliminate, NSB of a diversity of biomolecules at low concentrations and from samples with limited volumes. With selective sample preparation strategies, high performance surface, LC-MS compatible sample storage containers, and sensitive LC-MS/MS methods, the lower limits of quantification required for biotherapeutics can easily be achieved.
720006528, March 2019