
This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates to identify the major sources of post-column dispersion in nanoLC and understand their contributions to the observed peak widths.
Void volume within a union, often caused by a poorly cut capillary or an insecure union structure, creates more significant broadening.
Nanoflow liquid chromatography (nanoLC) interfaced with mass spectrometry, offers significant sensitivity benefits over the same separation carried out using conventional LC.1-3 It is thus favored in applications where the maximum sensitivity is desired from a limited volume sample. In theory, the separation efficiency of a nanocolumn should be the same as that of a standard-bore column of the same length. In practice, however, separations achieved using a nanocolumn have lower observed efficiencies. This is because nanoLC suffers more severely from extra-column dispersion than conventional LC due to the smaller column volume.⁴ Extra-column dispersion that occurs before the column, often referred to as pre-column dispersion, can be effectively mitigated using a clever experimental design. One example is to choose the optimal stationary phases for the trap and analytical columns to maximize the on-column refocusing effect.⁵ Post-column dispersion, however, cannot be reduced using the same principle because there is nothing that can chemically interact with the eluting bands. The only practical solution is to design the post-column flow path so that it contributes minimal band broadening.
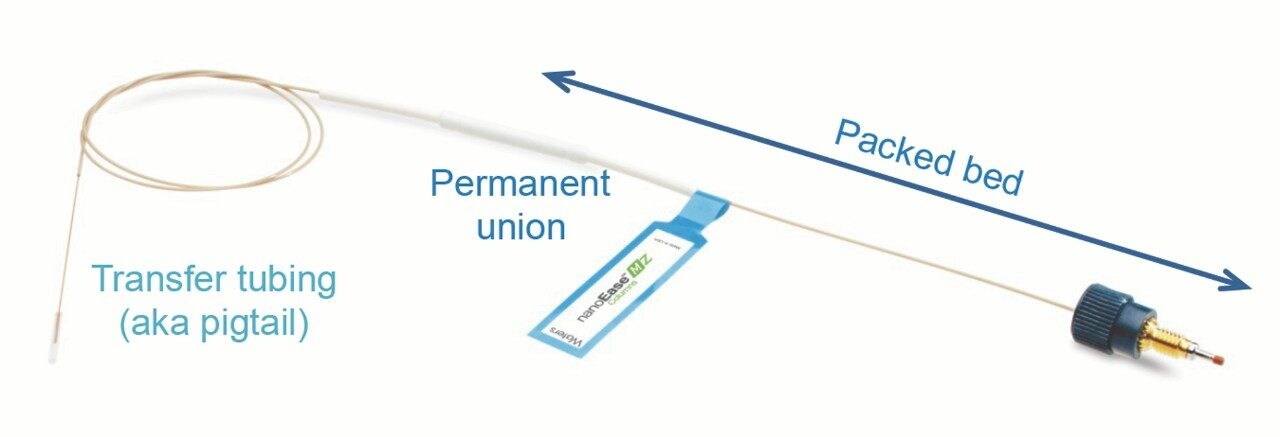
In that sense, some users have expressed concern about band broadening caused by the 20-inch-long post-column transfer tube (“pigtail”) on Waters’ capillary-based nanocolumns, including nanoEase M/Z Columns and ACQUITY UPLC M-Class Columns (Figure 1). Because of this concern, some users have reduced the tube length by cutting it short in their lab. This article examines the true dispersion contribution from the long, post-column transfer tube, and explains why cutting the tube short can hurt separation efficiency.
We measured the peak widths for peptides using typical proteomic separation conditions, before and after cutting down the post-column transfer tube. The capillary tube was cut using a cleaving tile or a circular cutter (Shortix cutter). Figure 2 shows the changes in peak widths for seven peptides from an enolase digest on a trap-and-elute nanoLC-MS setup using a nanoEase M/Z Symmetry C18, 5 µm, 180 µm x 20 mm Trap Column (p/n: 186008821) and a nanoEase M/Z HSS T3, 1.8 µm, 75 µm x 250 mm Column (p/n: 186008818). Contrary to expectations, cutting the tube resulted in significant increases in peak width. The band broadening was more significant when the capillary cut surface was not prepared properly. The uneven cut surface at the capillary end created a large void volume at the downstream capillary end-to-end union with the capillary emitter.
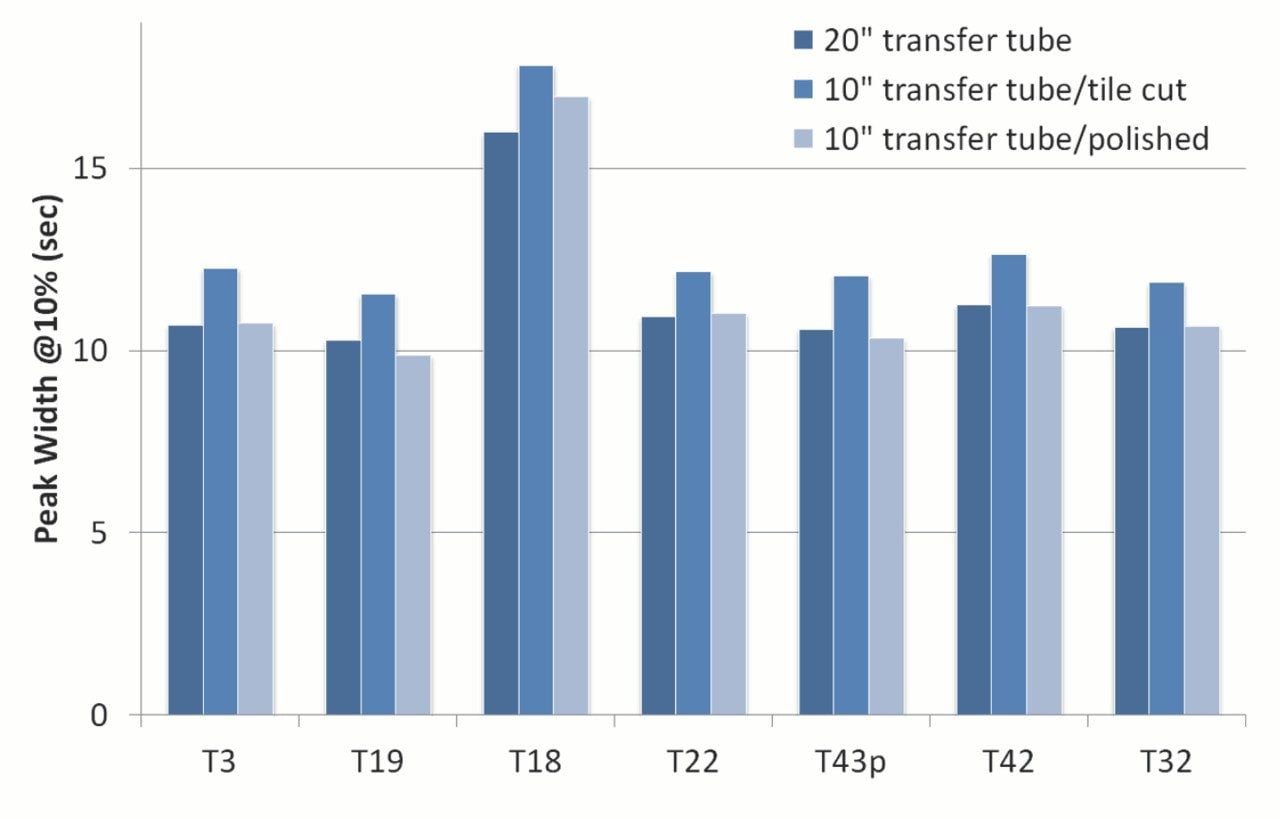
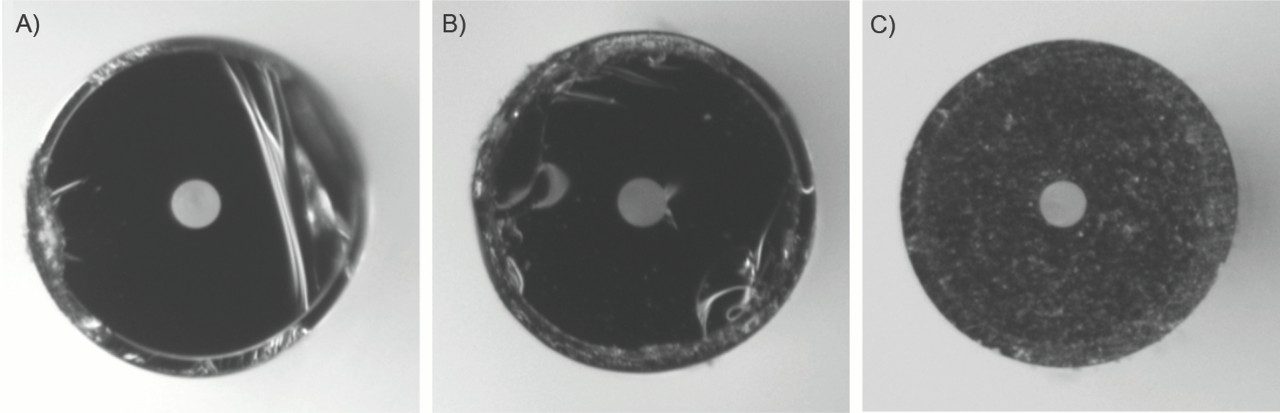
Figure 3 shows representative images of capillary cross-sections prepared using different cutting/polishing methods. A circular cutter tends to make a cleaner cut than does a cleaving tile, but nevertheless it frequently failed to provide a smooth surface. A smooth surface could be reliably achieved only after the capillary end was extensively polished using a special tool and multiple polishing papers. The union made with the cut surface shown in Figure 3C created no extra void volume.
It may be surprising that the chromatographic peak widths did not decrease significantly even when the capillary tube was cut short to 10 inches, and the cut surface was polished properly. The resulting peak widths were equivalent within the 5% experimental uncertainty to the peak widths achieved with the full 20-inch-long transfer tube. To better explain this observation, we can estimate the dispersion of a band traveling through an open capillary using the Taylor-Aris equation.⁶
σ²=(πr4FL)/(24∙D) - Eq. 1
Where σ² is the peak variance, r is the tube radius, L is the tube length, F is the flow rate, and D is the diffusion coefficient.
Under the experimental conditions used here (D ~ 1 x 10-5 cm²/s and F = 300 nL/min), a capillary of 20 µm I.D. and 10 inches in length is expected to contribute a peak variance of 1.7 nL.² This amount of dispersion contributes only a 0.7% increase in peak width to a 10-sec wide chromatographic peak.
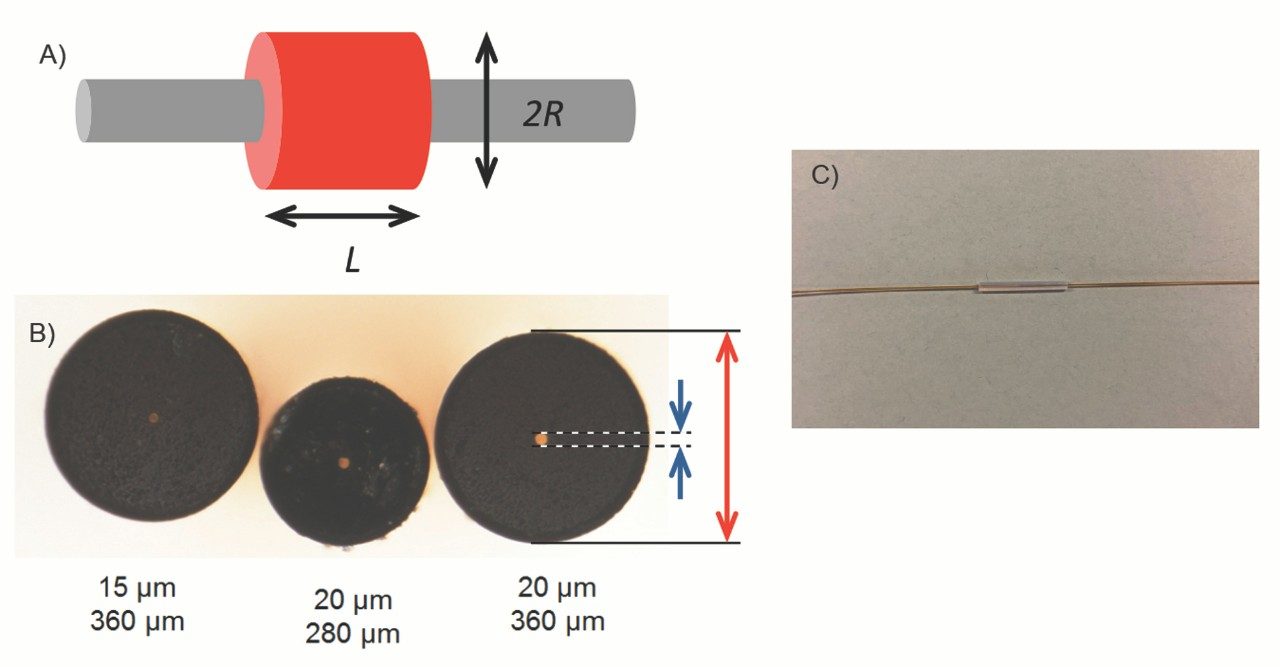
On the other hand, a large void can create dispersion through mixing or eddy effects (Figure 4A). This dispersion depends on the size and the shape of the void and can be estimated using the following equation.4,6
σ²= V²/n =(π2R4L2)/n - Eq. 2
Where σ² is the peak variance, V is the volume of the void, R is the radius of the void, L is the tube length, and n is a constant typically between 3 and 8.
A void formed at the capillary end-to-end union creates much greater dispersion than a long capillary tube does, because the void has a significantly larger volume (created by the 360 µm outer diameter of a typical fused-silica capillary) than a capillary inner volume created by the diameter of 20 µm or less (Figure 4B). Using the same experimental parameters as above, a 100 µm gap may contribute a 5–13% increase to a 10-sec wide peak. It is evident that an improperly cut capillary end can be detrimental to the observed chromatographic efficiency in nanoLC, while the benefit of having a short transfer tube is negligible.
Another common location where a void may form is between the packed capillary and the transfer tube. Most homemade columns and some commercially available nanocolumns have a capillary union held by a polymer sleeve (Figure 4C). This type of simple union keeps the two capillary ends together by the elasticity of the polymer and/or the frictional force. It is not surprising that these unions often fail to create a dispersion-free connection. All Waters’ capillary columns have a permanently attached union between the packed bed and the transfer tube (Figure 1), so that a void cannot be formed.
Post-column dispersion compromises the chromatographic efficiency in nanoLC. With this concern, the long post-column capillary transfer tubing of Waters’ nanocolumns has been suspected to contribute significant band broadening. We showed through experimental measurements and theoretical calculations that the band broadening from the transfer tubing is negligible. A more significant source is a void created in the capillary union, which may be caused by a poorly cut capillary or an insecure union structure.
720006526, April 2019