
This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates the effects of mobile phase composition and column type on RPLC analysis of free fatty acids.
ACQUITY UPLC BEH Columns impact chromatographic performance in the analysis of free fatty acids.
Free fatty acid (FFA) analysis often occurs in the biopharmaceutical industry as part of raw material assessment of surfactants such as polysorbate and on occasion in the analysis of degradation byproducts in formulated drug products. GC-based techniques are often used in the analysis of fatty acids, however derivatization steps are usually needed during sample preparation to increase instrument compatibility. LC-based techniques such as reversed phase chromatography (RPLC) are increasingly being deployed in the analysis of FFAs to bypass derivatization steps and increase productivity. RPLC-based analysis of FFAs can be challenging as physicochemical properties such as chain length and degree of saturation can impact the separation efficiency and detector response.
Furthermore, solvent compositions as well as column selection can yield significantly different chromatograms. Given the wide variety of mobile phase and column configurations possible for RPLC-based analyses, it is important to fundamentally understand the impact of basic organic to aqueous composition when analyzing free fatty acids with different RPLC column chemistries.
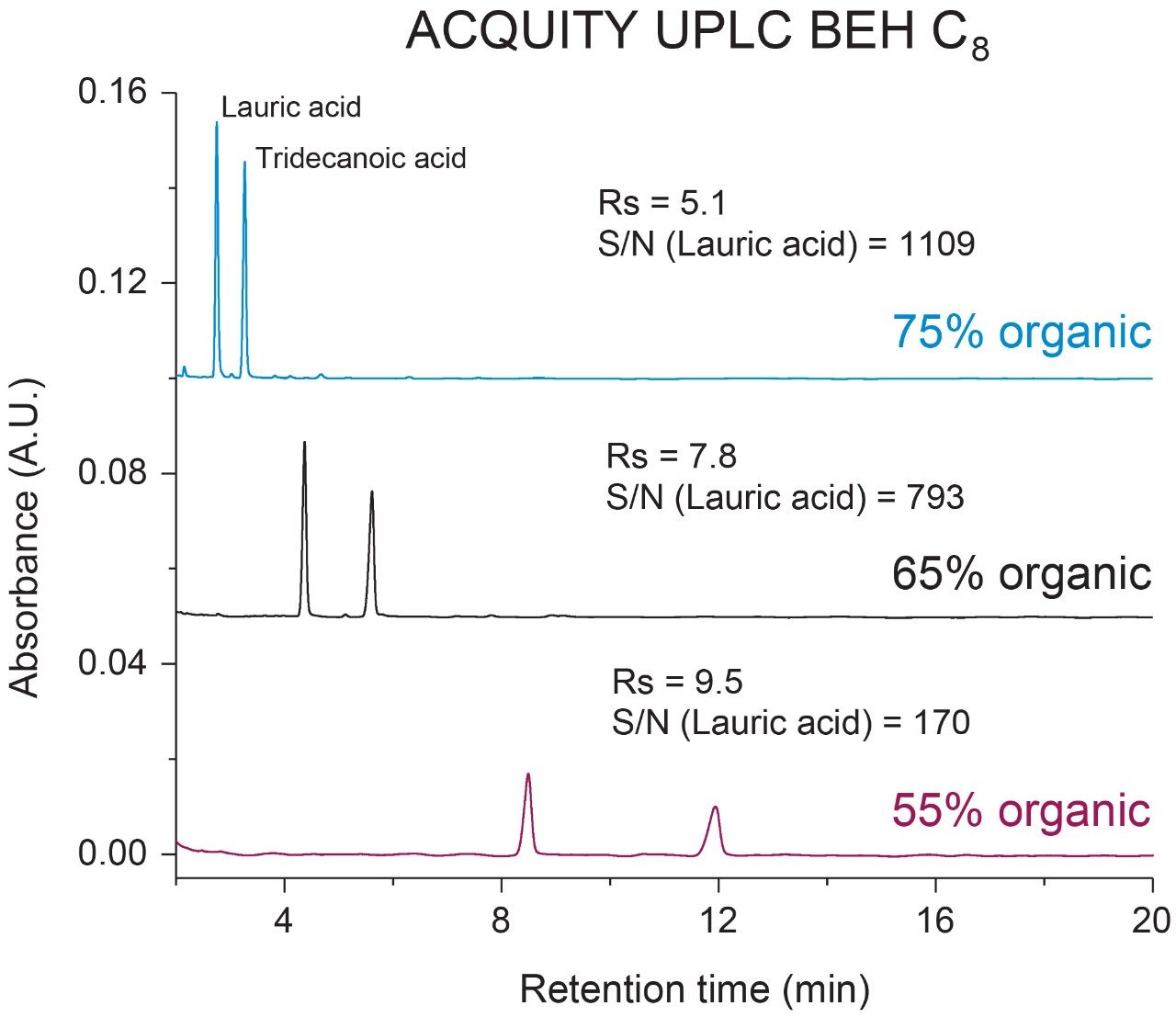
To evaluate the impact of mobile phase composition and column selection in the analysis of FFAs an ACQUITY UPLC BEH C8 and BEH C18 Column of the same dimensions (130Å, 1.7 µm, 2.1 mm × 100 mm) were used for this study. Lauric acid and tridecanoic acid were chosen to assess chromatographic performance as both are fully saturated and differ by one carbon in chain length. Mobile phase A and B were prepared as H2O, 0.1% FA and MeCN, 0.1% FA, respectively, and flowed under isocratic conditions at a %B composition of 75%, 65%, and 55%. Resolution and signal-noise-ratio (S/N) were used as figures of merit in this evaluation. As shown in Figures 1 and 2, samples run at higher organic compositions yielded the highest S/N with the FFAs eluting in the shortest amount of time. As expected, retention time of the FFAs increased as eluting organic composition was decreased due to increased hydrophobic interaction with the stationary phase. This effect was more pronounced in the C18 column, with the tridecanoic acid not eluting within the 20-minute run time. Consequently, lower S/N was observed in the more hydrophobic C18 column due to peak broadening when comparing organic compositions across columns. In addition to peak broadening at lower organic compositions, fronting was observed in the less retentive C8 column whereas tailing was observed in the more retentive C18 column for both fatty acids. This suggests incorporation of gradients may be necessary to resolve mixtures of higher or lower order FFA’s if needed. Both columns were able to adequately resolve the FFAs across all organic compositions with a resolution ≥5. These combined observations illustrate the importance of mobile phase composition and column selection when developing LC-based methods for the analysis of free fatty acids.
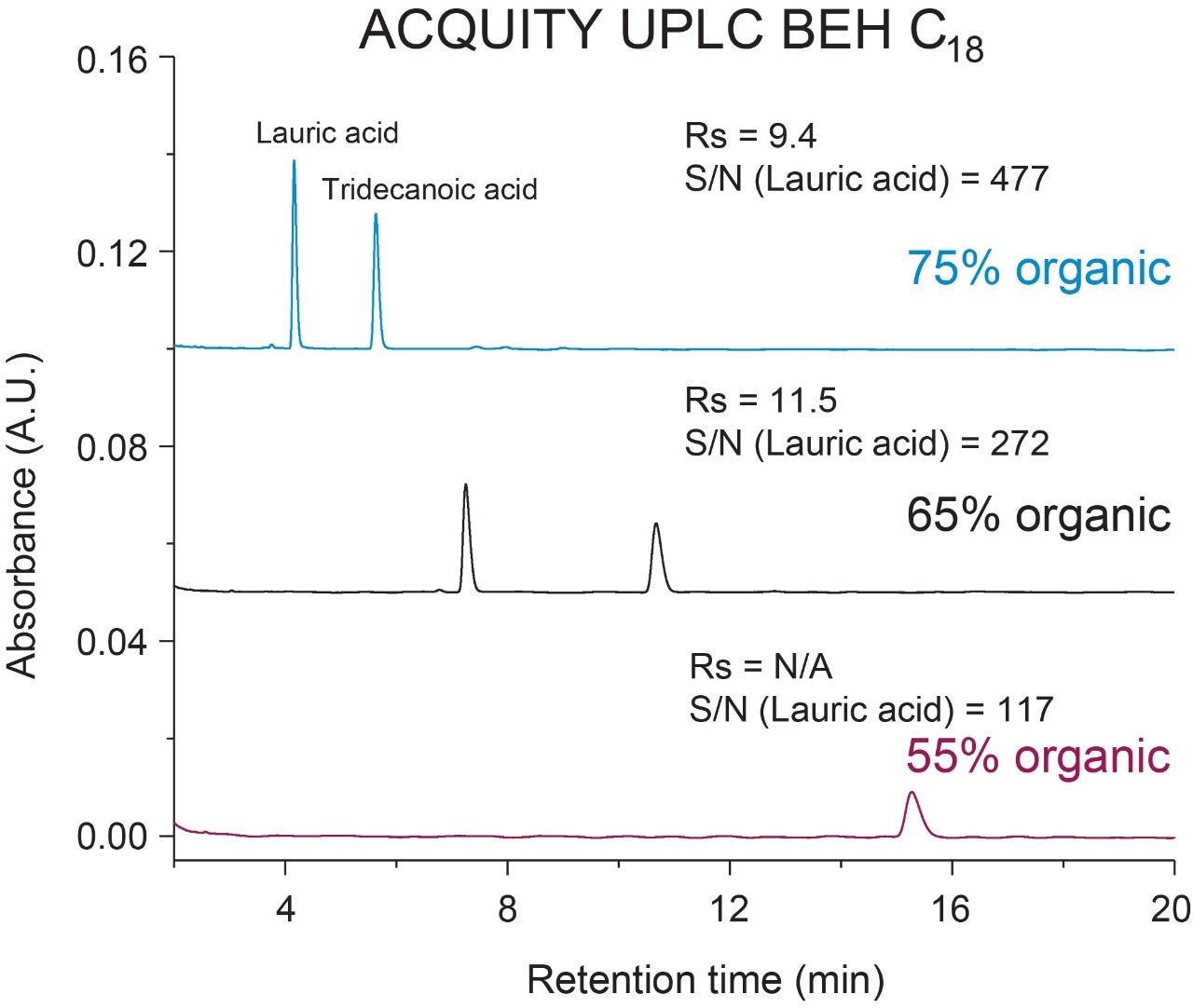
Intrinsic properties of FFAs such as chain length, degree of saturation, and hydrophobicity, can impact chromatographic performance and require optimization prior to analysis. When using RPLC conditions, organic composition and column selection can have significant impact on resolution, peak shape, and overall assay sensitivity. Higher organic composition mobile phases yield shorter elution times with higher S/N and peak resolution when compared to lower organic composition mobile phases. These observations were amplified when using more retentive column chemistries due to increased peak broadening and tailing. This work highlights the importance of mobile phase composition and column selection when performing FFA analysis.
720006611, June 2019