
Chlorate and perchlorate have traditionally been analyzed by ion chromatography, which requires the use of specialized equipment. This application note evaluates the performance of the ACQUITY UPLC I-Class System, coupled with the Xevo TQ-XS Mass Spectrometer, for the determination of chlorate and perchlorate in dairy products.
An alternative analytical method for the determination of chlorate and perchlorate in infant milk and other dairy products. The method is suitable for both official control and food business operators’ due diligence testing. It offers:
Recent reports by the European Food Safety Authority (EFSA), highlight that most of the food consumed within the European Union (EU) is largely free of pesticides.1 However, the latest monitoring figures show an increased number of exceedances compared to previous years. The difference is attributed to chlorate residues, a compound that was included for the first time in the 2016 control programs to support ongoing work in establishing maximum residue levels (MRLs). The report also indicated that a significant proportion of baby foods exceeded the MRL for chlorate. EFSA is currently reviewing the MRLs for chlorate and perchlorate in food.2
Food can be contaminated by chlorate and perchlorate during different stages of production. Perchlorate can be present in food via the use of fertilizers, while chlorate can be present due to the use of chlorinated disinfectants during food production. The dairy industry often uses hypochlorites and chlorine dioxide as sanitizing agents. Studies have confirmed that the use of these types of sanitizing agents could result in elevated chlorate levels in final products.3
Due to the rising health risks that chlorate poses to public safety in the food chain and the wider environment, chlorate is no longer approved as a pesticide in the European Union, therefore the default MRL of 0.01 mg/kg applies.2 However, the levels found in foods are often above this MRL.2
Chlorate and perchlorate have traditionally been analyzed by ion chromatography, which requires the use of specialized equipment. More recent methods involve LC-MS/MS, utilizing analytical columns that are highlighted in the Quick Polar Pesticides Methodology (QuPPe).4
An alternative UPLC-MS/MS method for chlorate and perchlorate, first outlined in an earlier Waters Corporation application note (p/n: 720006421EN), enables excellent chromatographic retention and separation using a novel hydrophilic interaction liquid chromatography (HILIC) column, which is now available as an application specific format, namely Waters' Anionic Polar Pesticide (APP) Column. The method offers a rapid, cost-effective approach to quantifying chlorate and perchlorate in infant milk products, extracted and cleaned up following the Quick Polar Pesticides Method for products of animal origin (QuPPe-AO) method.4
Samples were extracted and cleaned up following a modified version of the QuPPe-AO extraction procedure. Infant formula was rehydrated with LC-MS-grade water, as per labelling instructions, and 10 mL of the sample was extracted using 1% formic acid in methanol. Isotopically labelled internal standards were added to all samples prior to extraction, to correct for any extraction and injection variability. The samples were centrifuged, and an aliquot of the resulting supernatant was mixed with acetonitrile and C18 sorbent. Samples were vortex mixed and centrifuged prior to LC-MS/MS analysis. Figure 1 outlines the sample preparation method used for this analysis.

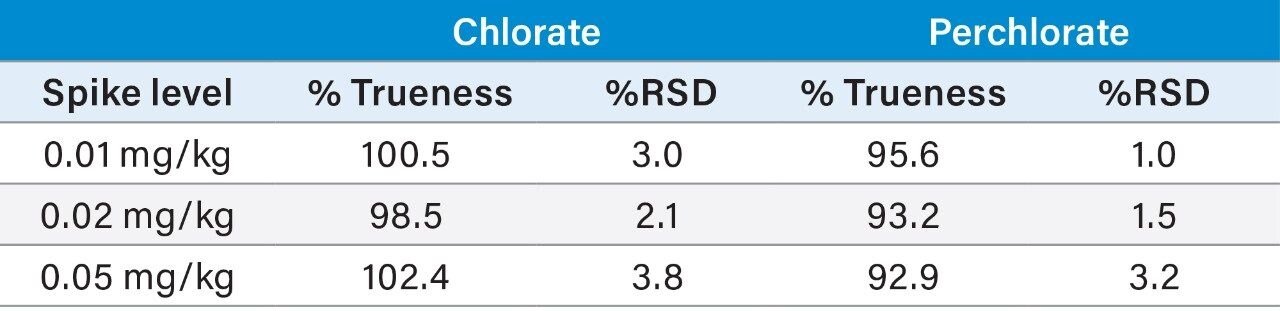
The performance of the method was assessed using the SANTE guidelines 11813/2017.5 To assess the trueness and precision of the method, infant milk samples were spiked at 0.010 mg/kg, 0.020 mg/kg, and 0.050 mg/kg. Solvent calibration standards were prepared in diluted QuPPe solvent, over the range of 0.004–0.800 mg/kg (1.0–200 ng/mL, in vial concentration). Labelled versions of chlorate and perchlorate were also added as internal standards. Bracketed calibration curves were used to determine the concentration of the chlorate and perchlorate spiked samples.
|
LC conditions |
|
|---|---|
|
System: |
ACQUITY UPLC I-Class PLUS |
|
Column: |
Waters Anionic Polar Pesticide (APP) Column 2.1 × 50 mm, 5 μm (P/N: 186009286) |
|
Column temp.: |
50 °C |
|
Sample temp.: |
10 °C |
|
Injection volume: |
2 μL |
|
Flow rate: |
0.5 mL/min |
|
Mobile phase A: |
Water with 0.9% formic acid and 50 mM ammonium formate (LC-MS-grade) |
|
Mobile phase B: |
Acetonitrile with 0.9% formic acid (LC-MS-grade) |
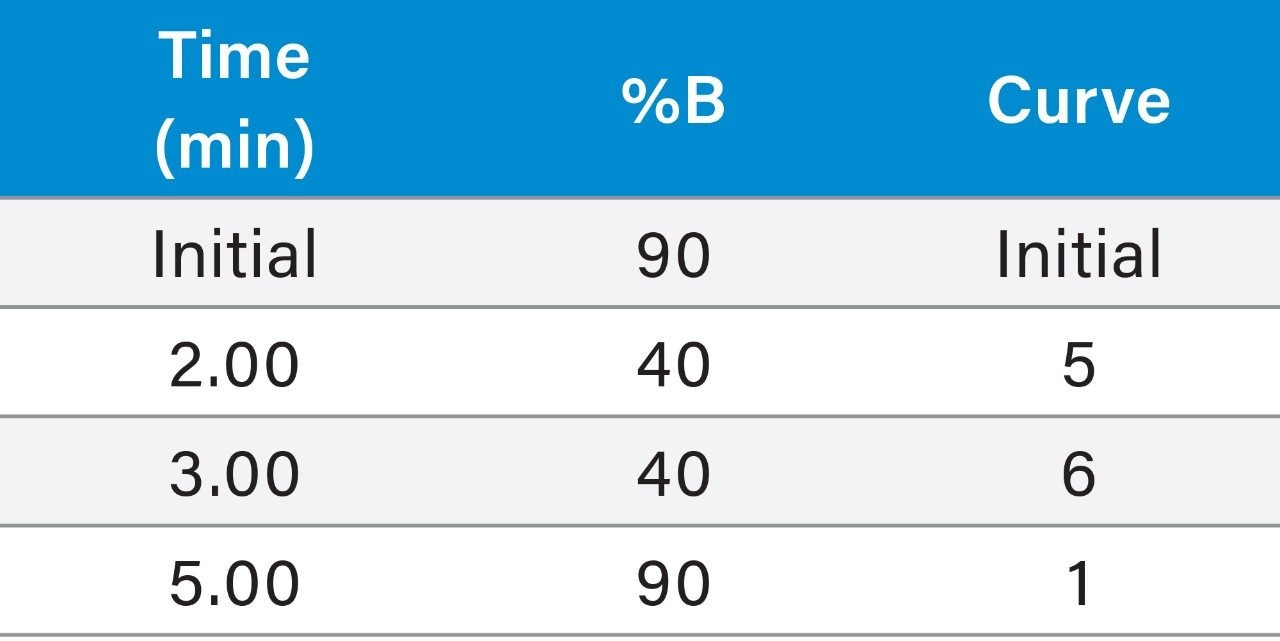
|
System: |
Xevo TQ-XS Triple Quadrupole Mass Spectrometer |
|
Software: |
MassLynx v4.2 |
|
Ionization mode: |
ESI- |
|
Acquisition mode: |
MRM |
|
Capillary voltage: |
0.50 kV |
|
Cone: |
15 V |
|
Cone gas flow: |
150 L/Hr |
|
Desolvation temp.: |
600 °C |
|
Desolvation gas flow: |
800 L/Hr |
|
Source temp.: |
150 °C |
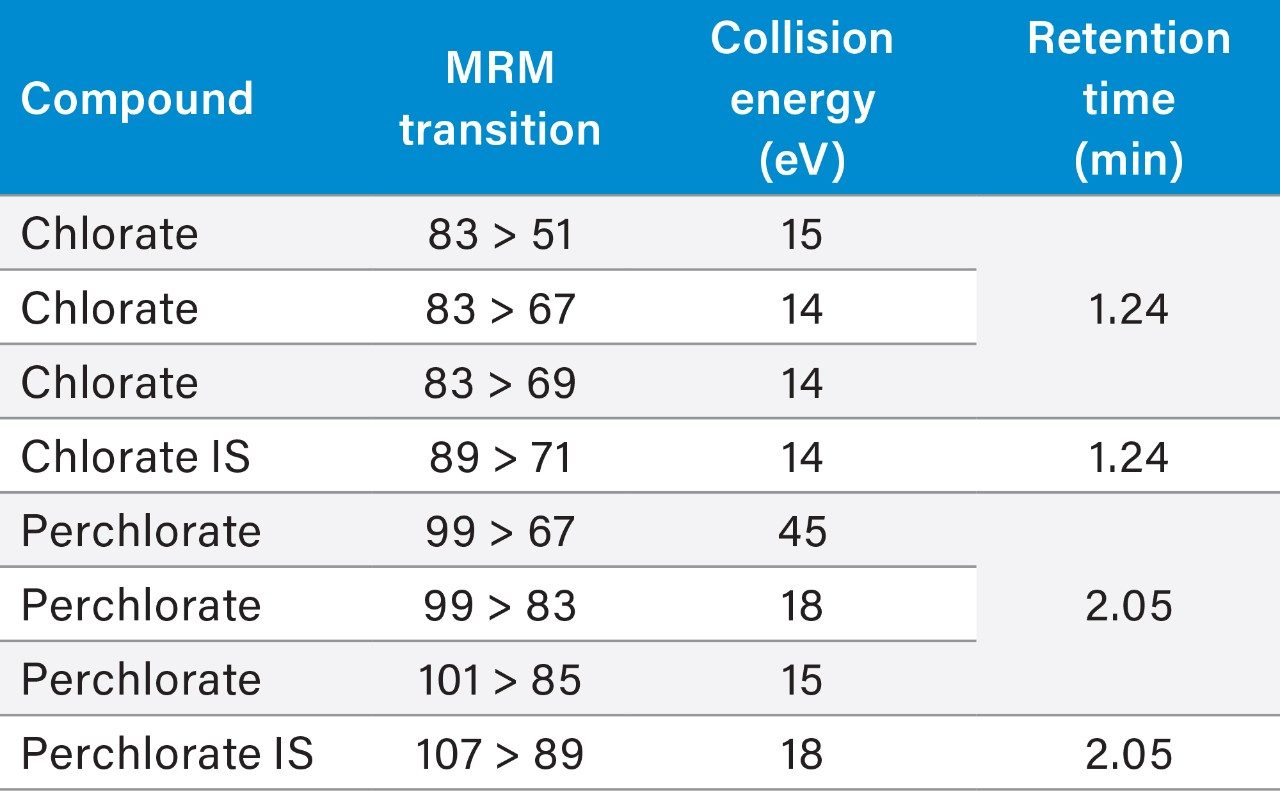
Method optimization was accomplished by evaluating mobile phase composition, buffer concentrations, gradient slopes, and MS transitions. The conditions detailed in the experimental section provided the best overall performance.
Dairy samples contain matrix components that can interfere with various stages of the analysis, such as proteins, fats, and sugars. To minimize the impacts of these matrix components, analytical methods typically include protein precipitation and defatting steps. The QuPPe-AO extraction and clean-up procedure provided adequate removal of proteins and fats from the infant milk samples and improved the robustness of the methodology. Isotopically labelled internal standards of chlorate and perchlorate were able to correct for any extraction and injection variability.
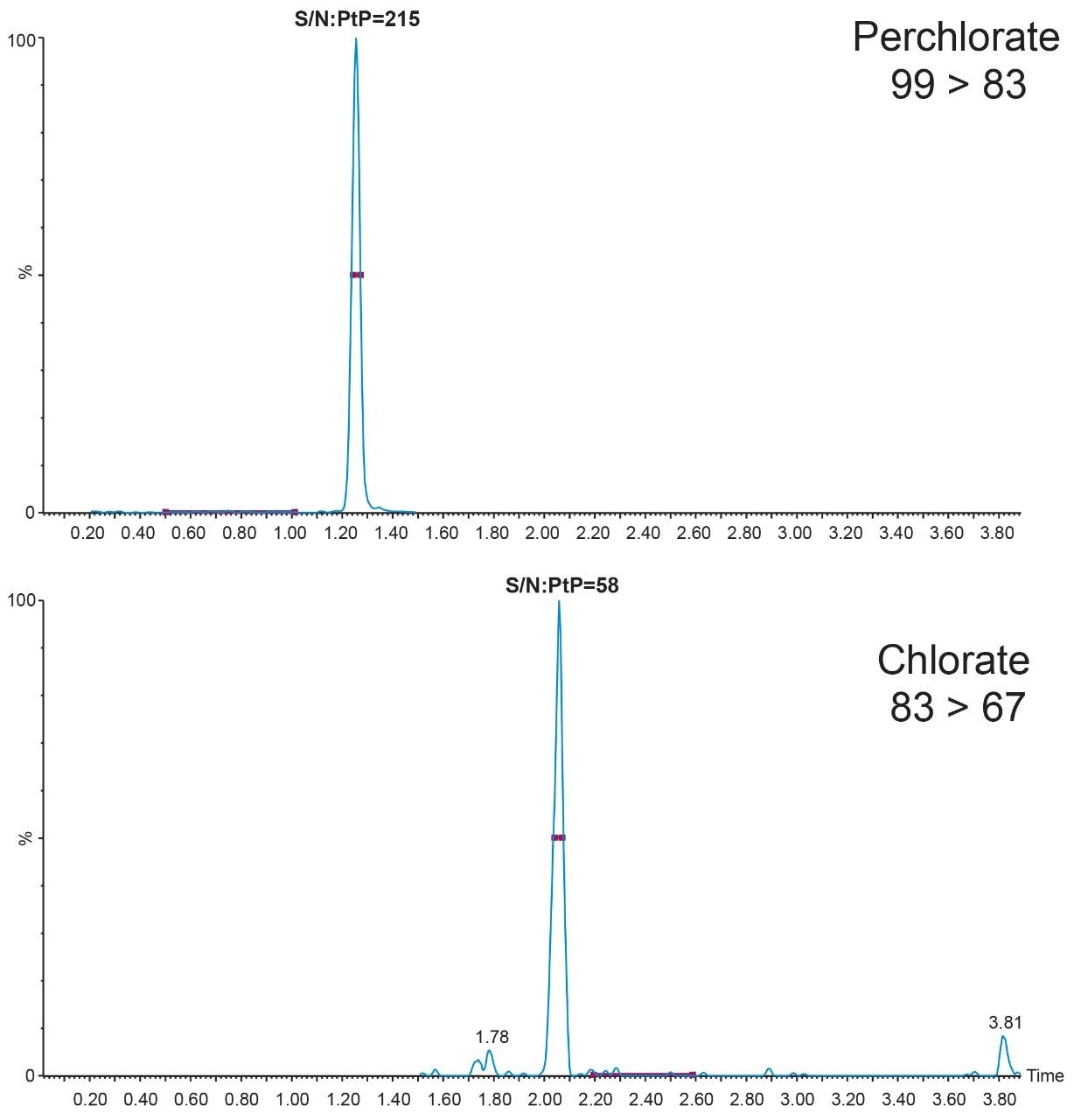
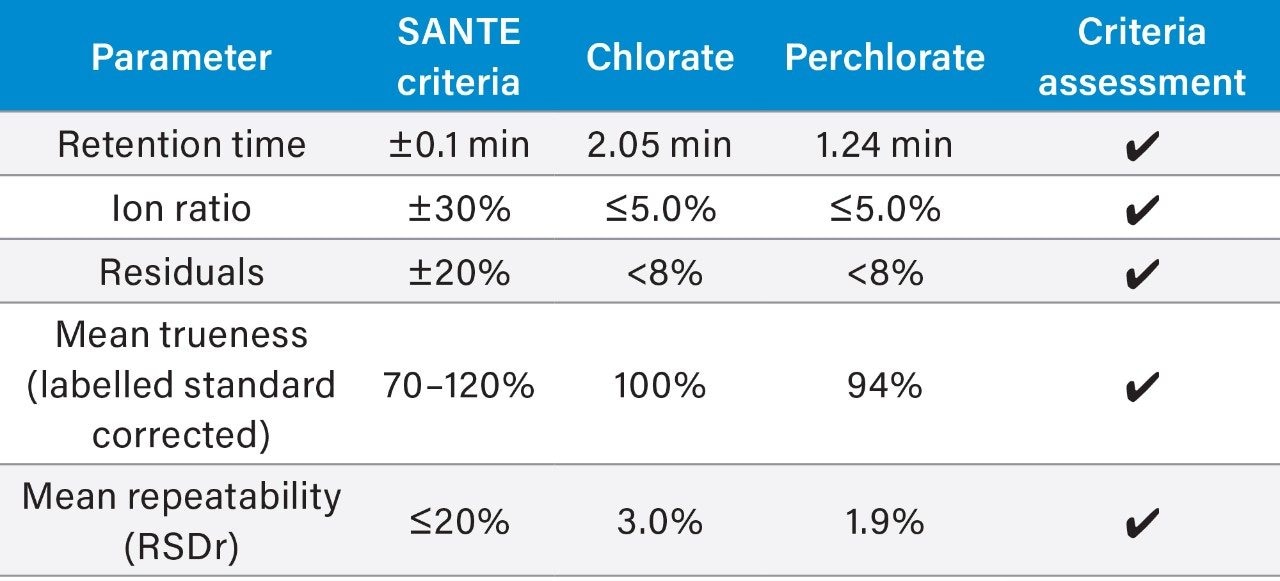
The method has been assessed for performance using the criteria outlined in SANTE guidelines 11813/2017, including repeatability (RSDr) and trueness. The internal verification of the method demonstrated excellent performance for the identification and quantification of chlorate and perchlorate. An example chromatogram for a 0.004 mg/kg solvent standard showing the peak to peak signal to noise is shown in Figure 2.
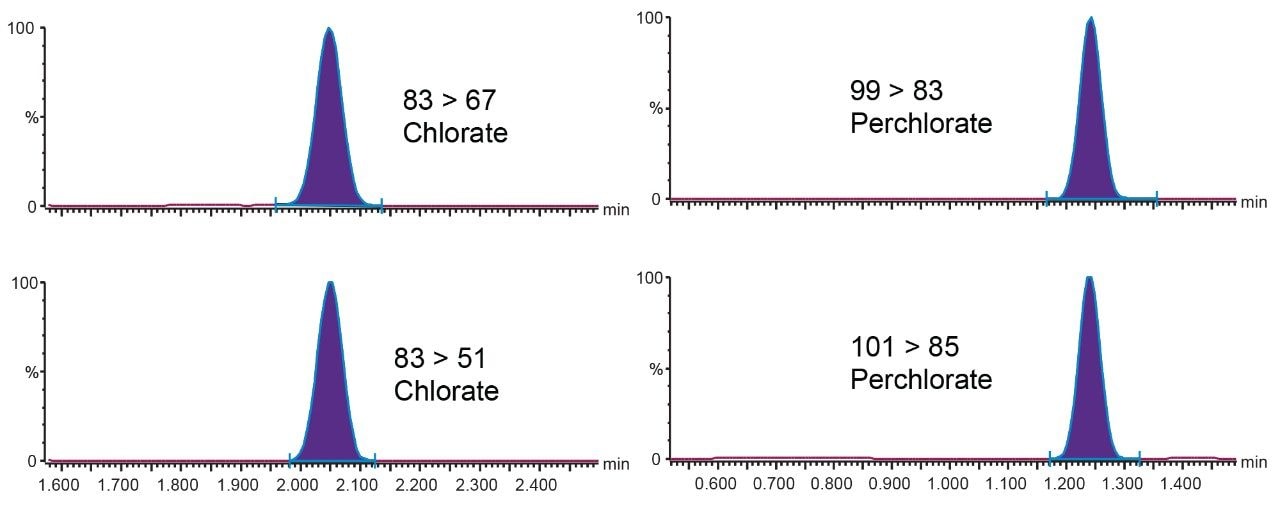
Example chromatograms showing the retention and separation for the two target compounds are shown in Figure 3. The column void volume (t0) is calculated for this APP column at 0.24 min, retention was achieved for all the compounds at greater than the two times the column void volume criteria outlined in the SANTE guidelines. The total run time of the method, including column re-equilibration, was five minutes.
The calibration linearity for both chlorate and perchlorate was evaluated using bracketed calibration curves. The calibration curve was spiked into solvent over the range of 0.004–0.800 mg/kg (1.0–200 ng/mL, in vial concentration) as shown in Figure 4. The correlation coefficient and the residuals were excellent (r2 > 0.999 and residuals <10%).
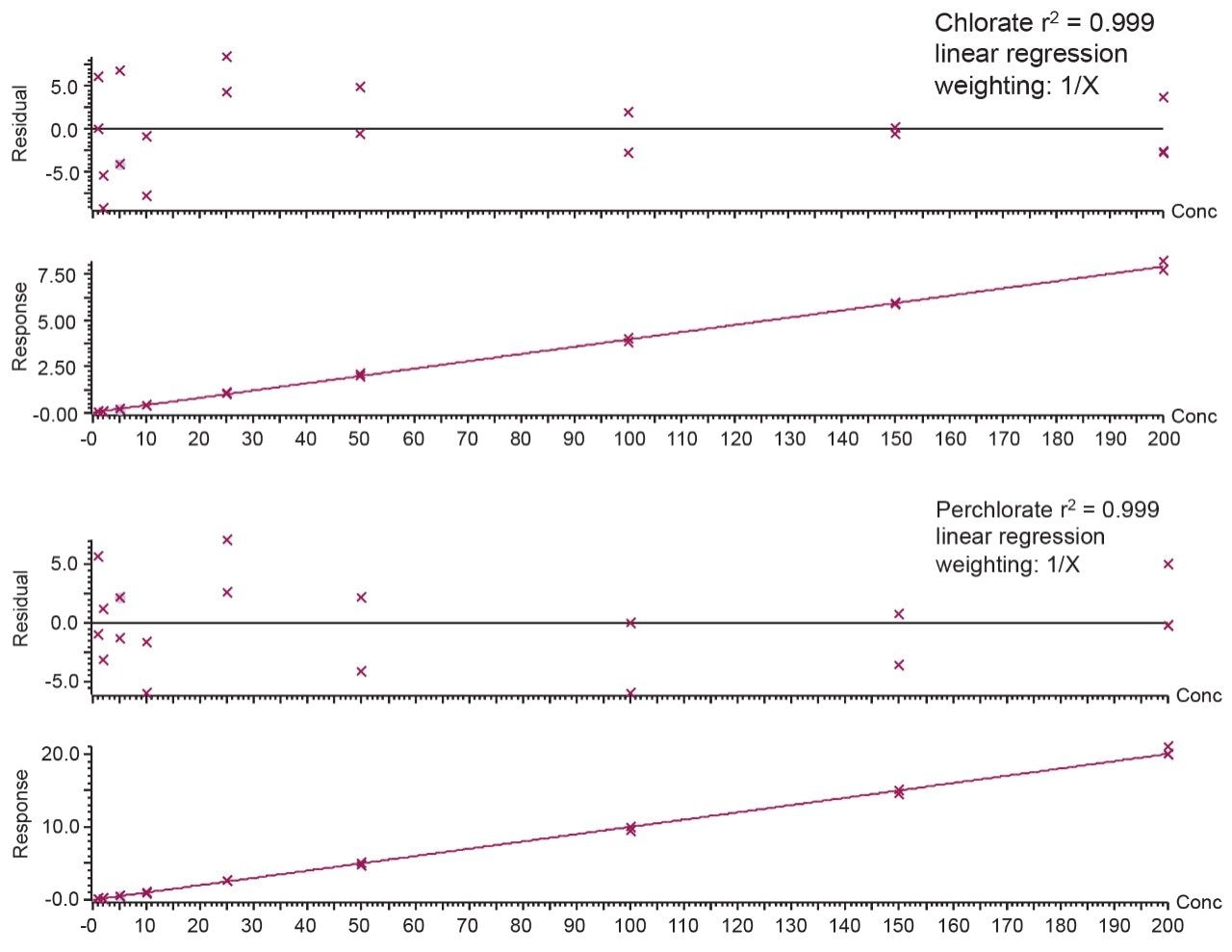
The repeatability and trueness of the methodology were determined by extracting and injecting samples at various spike levels (0.010 mg/kg [n = 5], 0.020 mg/kg [n = 5], and 0.050 mg/kg [n = 5]). The repeatability (%RSD) and recoveries from analysis of infant formula can be seen in Table 3. The retention time and ion ratios were within the tolerances defined in the SANTE guidelines. Table 4 provides a summary of method validation for the determination of chlorate and perchlorate in infant formula against key criteria set out in the SANTE guidelines.
The aim of this study was to evaluate the performance of the ACQUITY UPLC I-Class System and Waters Anionic Polar Pesticide (2.1 x 50 mm) Column coupled with the Xevo TQ-XS Mass Spectrometer, for the determination of chlorate and perchlorate in dairy products. In-house verification of this approach showed excellent sensitivity for the detection, identification, and quantification of chlorate and perchlorate. The method provided effective extraction and clean-up and can be applied to various dairy matrices. The Xevo TQ-XS provided exemplary performance in terms of linearity and calibration range. The trueness and precision of the method determined at various QC levels gave excellent results for bias and %RSD.
720006686, Revised July 2020