For research use only. Not for use in diagnostic procedures.
This is an Application Brief and does not contain a detailed Experimental section.
To demonstrate the analytical sensitivity gains obtained for the analysis of aldosterone on the Xevo TQ-XS Mass Spectrometer compared to the Xevo TQ-S Mass Spectrometer for clinical research.
The Xevo TQ-XS provides significantly improve analytical sensitivity for aldosterone in plasma compared to the Xevo TQ-S.
Aldosterone is a mineralocorticoid steroid hormone produced by the zona glomerulosa of the adrenal cortex. The production of aldosterone is assessed during pharmacological clinical research studies of aldosterone synthase (CYP11B2) inhibitors. It can be present at low concentrations in plasma (<25 pg/mL), making it difficult to measure with an acceptable level of precision and accuracy.

The Xevo TQ-XS (Figure 1) is an advanced benchtop, tandem quadrupole mass spectrometer expanding the scope of ultimate analytical sensitivity analysis. It features StepWave™ XS ion transfer optics – delivering enhanced analytical sensitivity, robustness, and reliability. In addition, the new Xtended Dynamic Range (XDR) Detector allows six orders of linear dynamic range. This ensures that methods are easily transferable onto the instrument and wide concentration ranges can be measured in a single analysis.
In this technology brief, the Xevo TQ-XS was used to significantly improve the analytical sensitivity and imprecision of a LC-MS/MS method for the analysis of aldosterone in plasma.
Plasma samples were precipitated with zinc sulfate/methanol and diluted with phosphoric acid before loading onto the Oasis® MAX μElution Plate. Consecutive washes with phosphoric acid, ammonia in 10% methanol, and water were performed. Samples were eluted with 70% aqueous methanol and diluted with water.
Samples were injected on an ACQUITY UPLC I-Class System utilizing a water/methanol gradient and a CORTECS C18 column. The same UPLC system and solvents were used to evaluate both the Xevo TQ-S and Xevo TQ-XS.
MRM transitions monitored for aldosterone were 359.2 > 189.0 (quantifier) and 297.2 (qualifier). The internal standard – aldosterone-⁴H2 – had a MRM transition of 363.2 > 190.0.
To convert conventional mass units (pg/mL) to SI units (pmol/L) multiply by 2.774.
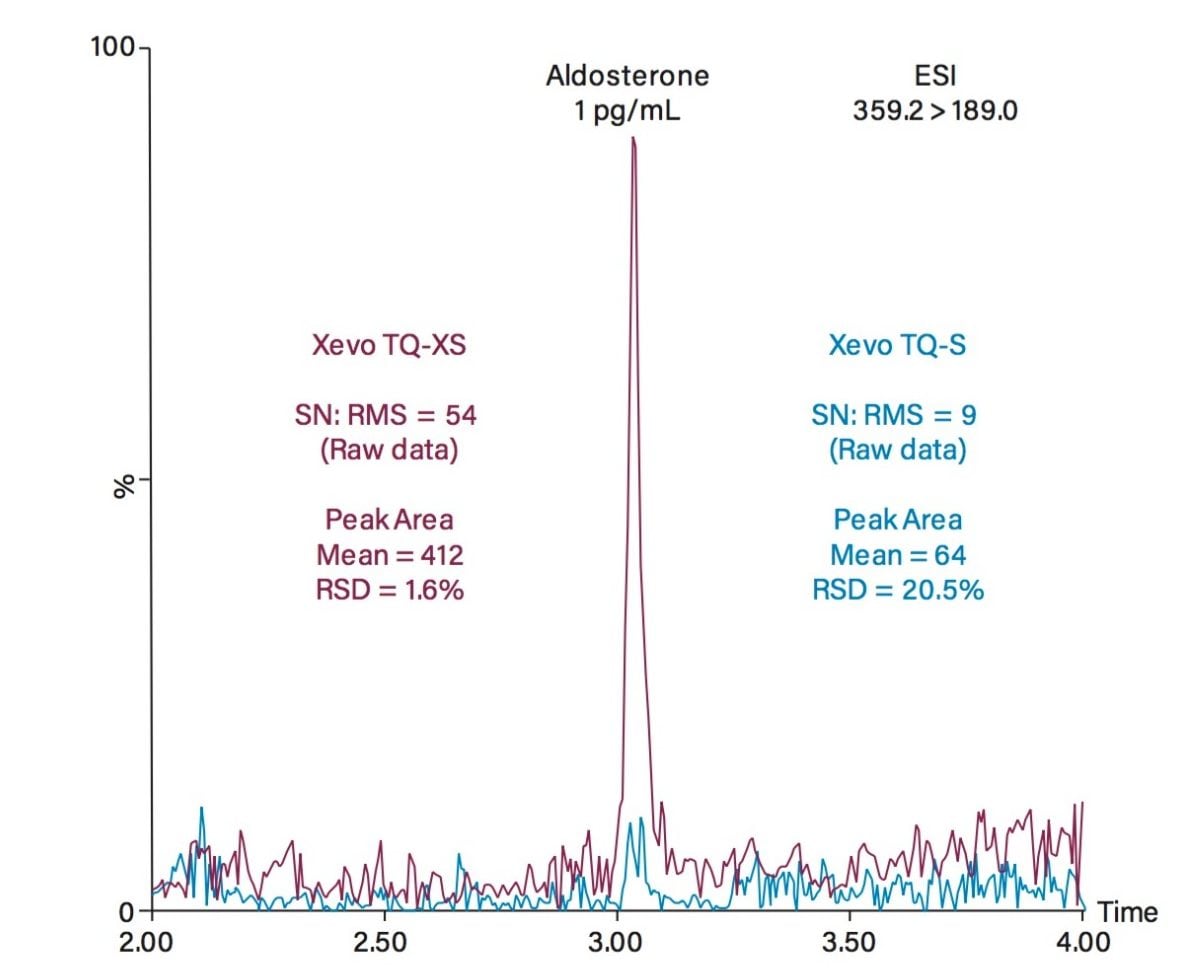
A system comparison using a 1 pg/mL solution of aldosterone was performed to demonstrate the analytical sensitivity improvement observed for aldosterone on the Xevo TQ-XS (Figure 2). This analytical sensitivity improvement also contributes to a significant improvement in imprecision of the peak area at this concentration (<2% RSD, n=3).
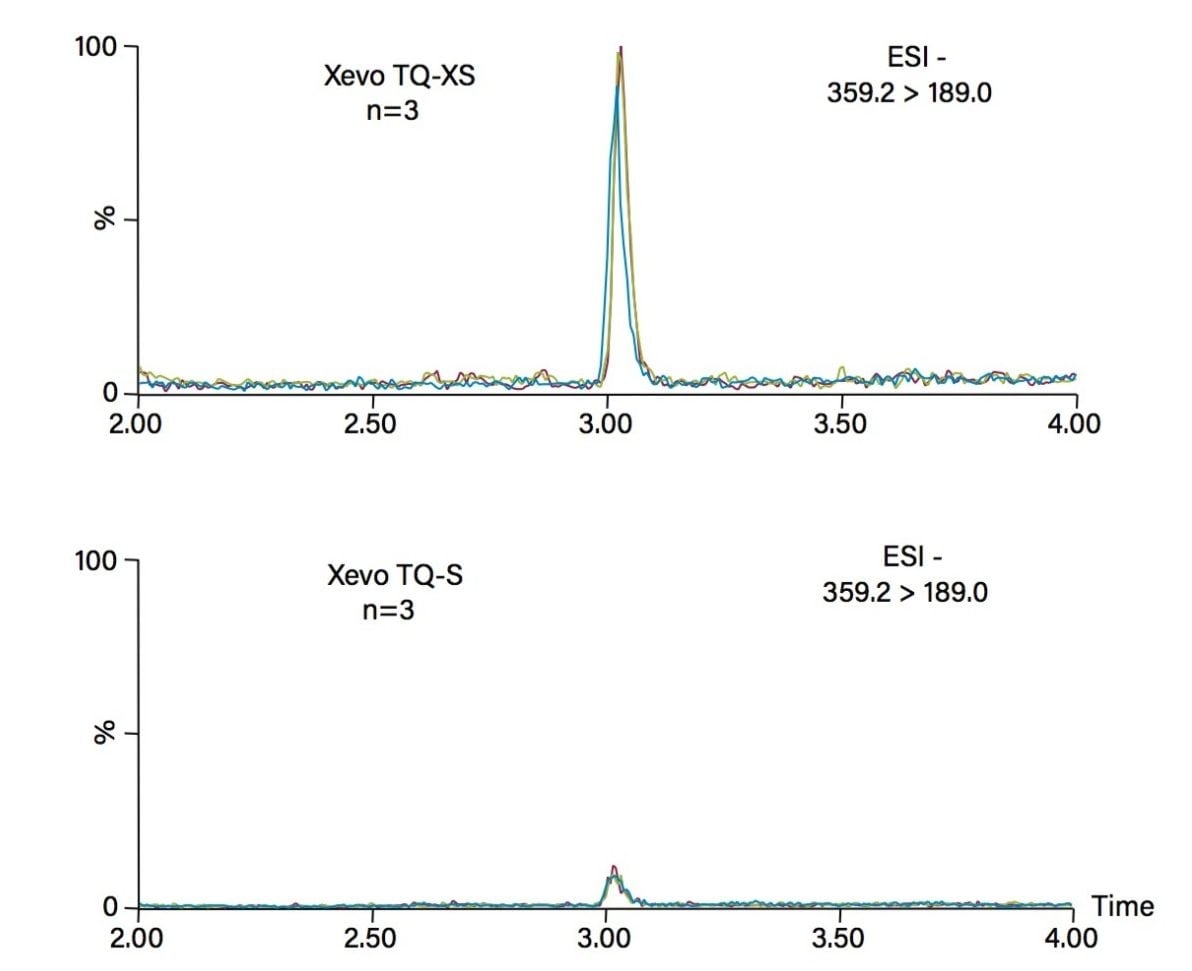
The low calibration serum standard used for the LC-MS/MS method was assessed on each system over three separate days. This evaluation demonstrates reproducibility of a sample extracted and injected on both the Xevo TQ-XS and Xevo TQ-S, on three separate occasions (Figure 3).
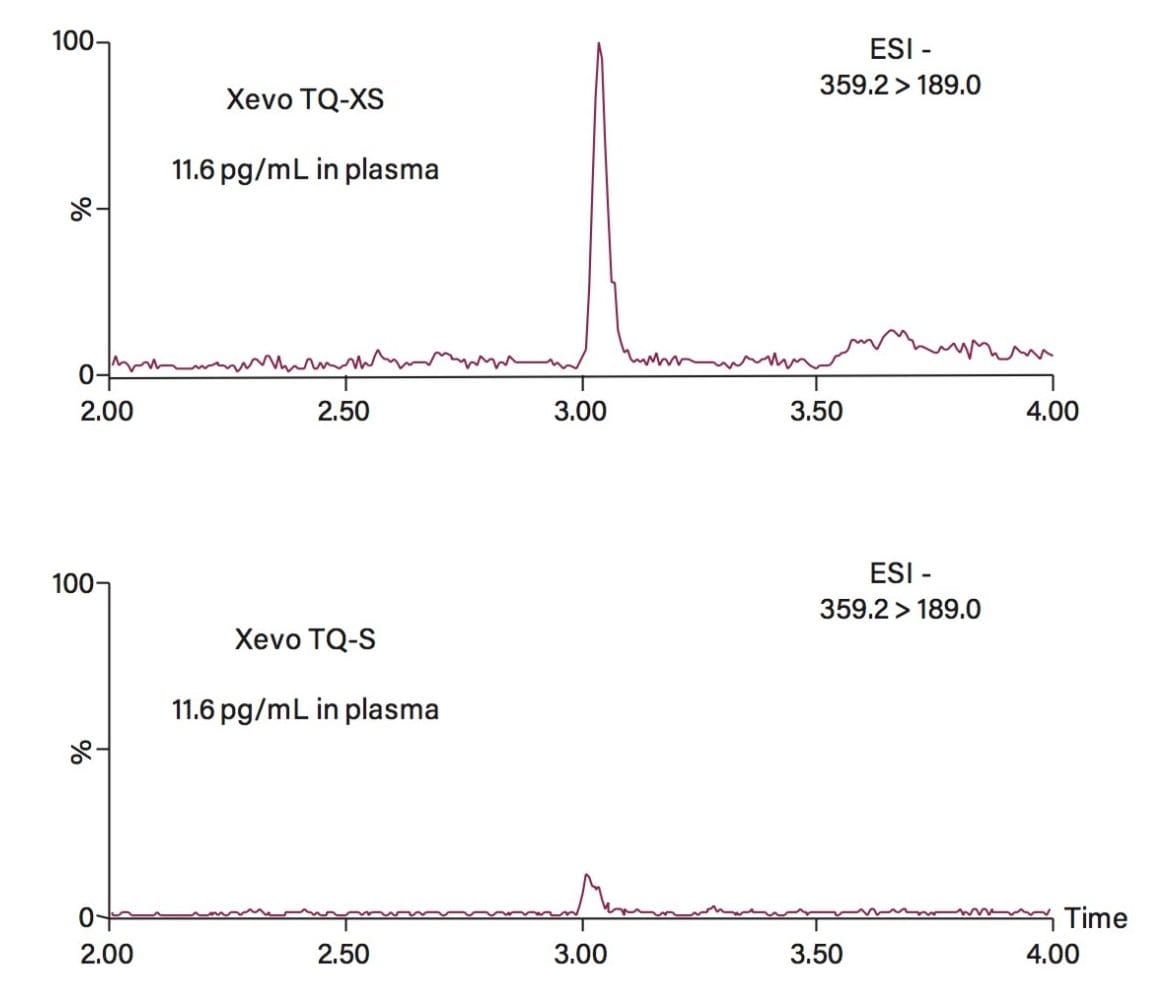
An evaluation using an extracted aldosterone plasma sample was performed, demonstrating the analytical sensitivity improvement on the Xevo TQ-XS that was observed for analyzing endogenous aldosterone in plasma (Figure 4).
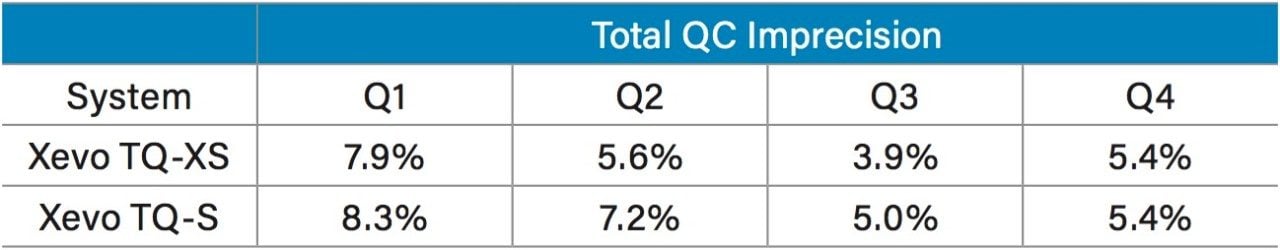
Total imprecision of the LC-MS/MS method was assessed on both the Xevo TQ-XS and Xevo TQ-S over three separate days, with ten replicates on each day at 21, 42, 174, and 669 pg/mL (n=30). Total imprecision was shown to improve on the Xevo TQ-XS (Table 1).
The Xevo TQ-XS has demonstrated significantly improved performance of our LC-MS/MS method for the analysis of aldosterone for clinical research.
The benefits of this system for the analysis of aldosterone include:
720005728, June 2016