This application note demonstrates that an established ion-exchange method for the separation of a monoclonal antibody can be easily replicated and can yield repeatable results. The ACQUITY Arc System offers users the ability to emulate legacy HPLC methods or update methods to UHPLC separations through the use of Arc Multi-flow path technology.
Many of the top grossing biopharmaceuticals today are protein-based drugs. Unlike the synthesis of small molecule drugs which is a controlled chemical process, biologics are produced from live cell cultures, making them more prone to variation. A host of techniques are often required throughout the life cycle process to ensure product quality and safety. Ion-exchange chromatography (IEX) is a common technique used to monitor charge variants, which can reflect changes made in various stages of production such as fermentation, purification, and formulation.
The ACQUITY Arc System, part of the quaternary based LC portfolio, is equipped to address the challenge of maintaining consistent results throughout the life cycle of product testing. Arc Multi-flow path technology allows the ACQUITY Arc System to easily replicate legacy HPLC methods (Path 1) or improve existing methods by transitioning to UHPLC-based separations (Path 2). The focus of this application note is to demonstrate that an established ion-exchange method for the separation of a monoclonal antibody can be easily replicated and can yield repeatable results.
|
LC systems: |
ACQUITY Arc System with 2489 UV/Vis detector, flow path 1 Agilent 1100 Series HPLC System with quaternary pump and DAD detector |
|
Absorption wavelength: |
280 nm |
|
Sampling rate: |
20 Hz |
|
Column: |
Dionex ProPac WCX-10 10 μm,4.0 mm x 250 mm |
|
Column temp.: |
30 °C |
|
Mobile phase A: |
0.02 M MES, pH 6.0 |
|
Mobile phase B: |
0.02 M MES, 0.4 M sodium chloride, pH 6.0 |
|
Sample: |
1 mg/mL rituximab in 0.15 M sodium chloride, 0.025 M sodium citrate, 0.06% (v/v) polysorbate 80, pH 6.5 |
|
Sample temp.: |
5 °C |
|
Injection volume: |
40 μL |
|
Time (min) |
Flow rate (mL/min) |
%A |
%B |
%C |
%D |
|---|---|---|---|---|---|
|
Initial |
0.700 |
100 |
0 |
0 |
0 |
|
0.01 |
0.700 |
100 |
0 |
0 |
0 |
|
5.00 |
0.700 |
100 |
0 |
0 |
0 |
|
10.00 |
0.700 |
83 |
17 |
0 |
0 |
|
95.00 |
0.700 |
55 |
45 |
0 |
0 |
|
100.00 |
0.700 |
0 |
100 |
0 |
0 |
|
102.00 |
0.700 |
0 |
100 |
0 |
0 |
|
102.01 |
0.700 |
100 |
0 |
0 |
0 |
|
115.00 |
0.700 |
100 |
0 |
0 |
0 |
Empower 3 CDS Software, SR2
Charge variants are often monitored throughout the life cycle of a drug product to monitor process control. As such, system-tosystem variability must be determined to ensure product quality, regardless of the instrument used for testing. Ideally, each system used should provide comparable results that fall within the accepted guidelines for that product. To investigate the ability of a cation-exchange method to migrate from a legacy instrument to a modern LC platform, rituximab, a monoclonal antibody, was analyzed on an Agilent 1100 Series HPLC System and compared to results from the ACQUITY Arc System. The method used is from the USP Medicines Compendium,1 and although the method has since been discontinued, it serves as a representative method used in industry.
The method described above was executed on both the Agilent 1100 Series HPLC System and the ACQUITY Arc System without modification (Figure 1). As shown, the chromatographic performance between the two systems is nearly identical. When looking at the insets in Figure 1, it can be seen that selectivity and resolution are preserved across platforms. Treatment of the data to determine relative retention time of the main peak and additional acidic and basic variants is shown in Table 1. Although differences in retention time exist, it is more important to note that the relative retention times of all major peaks are negligible.
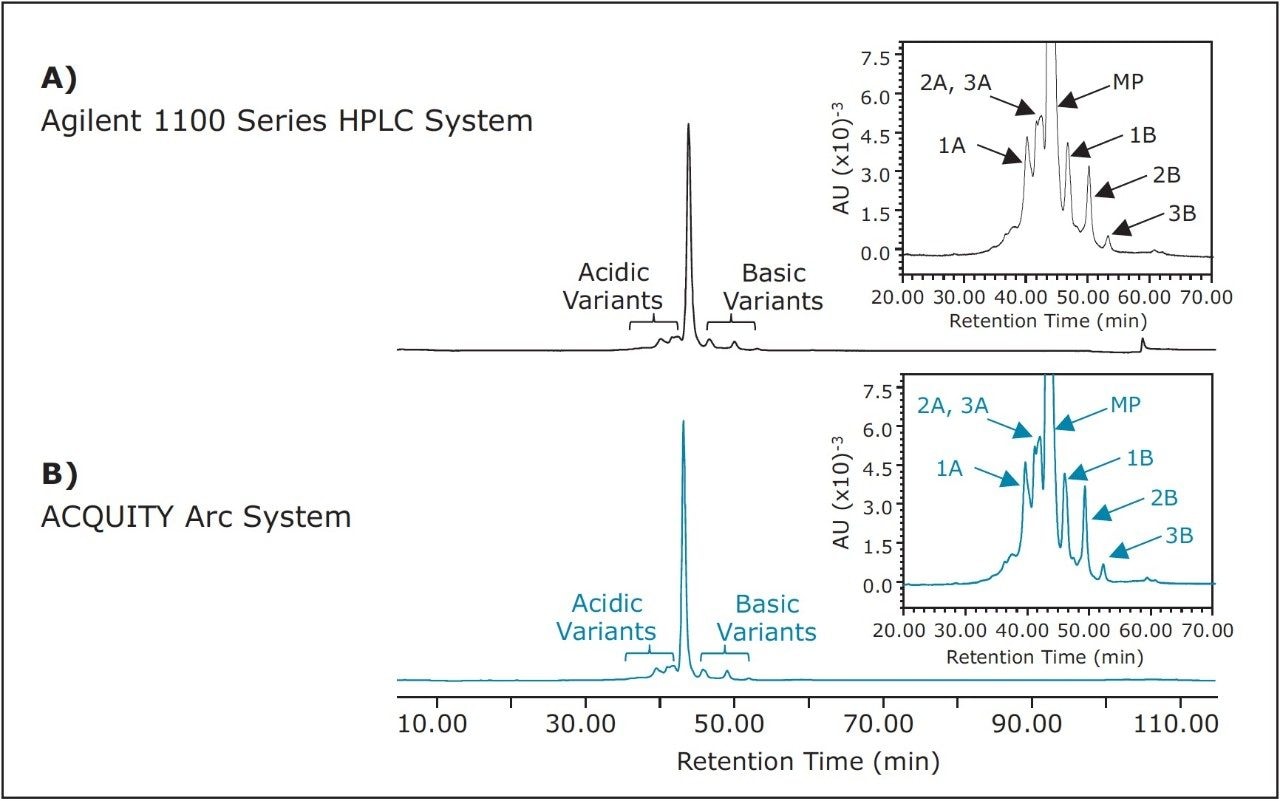
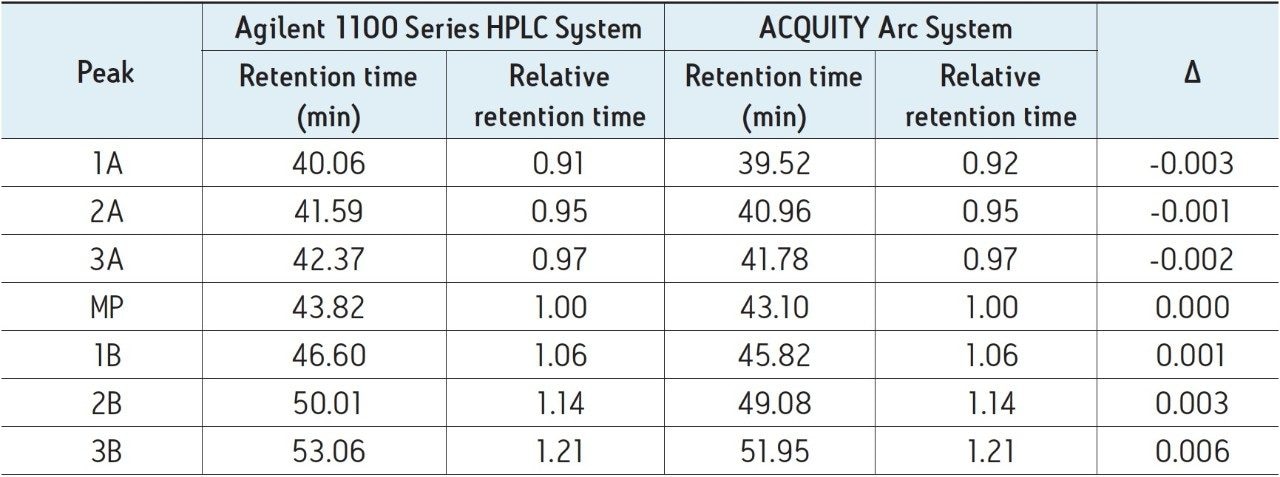
Resolution is an example of one parameter that can be calculated to determine if system suitability criteria is met. Per the USP Medicines Compendium,1 resolution between the main peak of rituximab (MP) and the lysine variant (1B) should be no less than 1.5. Over five injections, the Agilent 1100 Series HPLC System reports an average resolution of 1.95 ± 0.02 compared to 2.09 ± 0.02 reported on the ACQUITY Arc System. Both systems fall within regulations.
To ensure product consistency, reproducibility must not only be demonstrated from system to system, but within an instrument as well. Figure 2 shows an overlay of five rituximab injections on the ACQUITY Arc System run under the same method conditions as described above. Each chromatogram is nearly indistinguishable from the next throughout the length of the run. From the inset, an image of a zoomed in chromatogram further confirms this agreement.
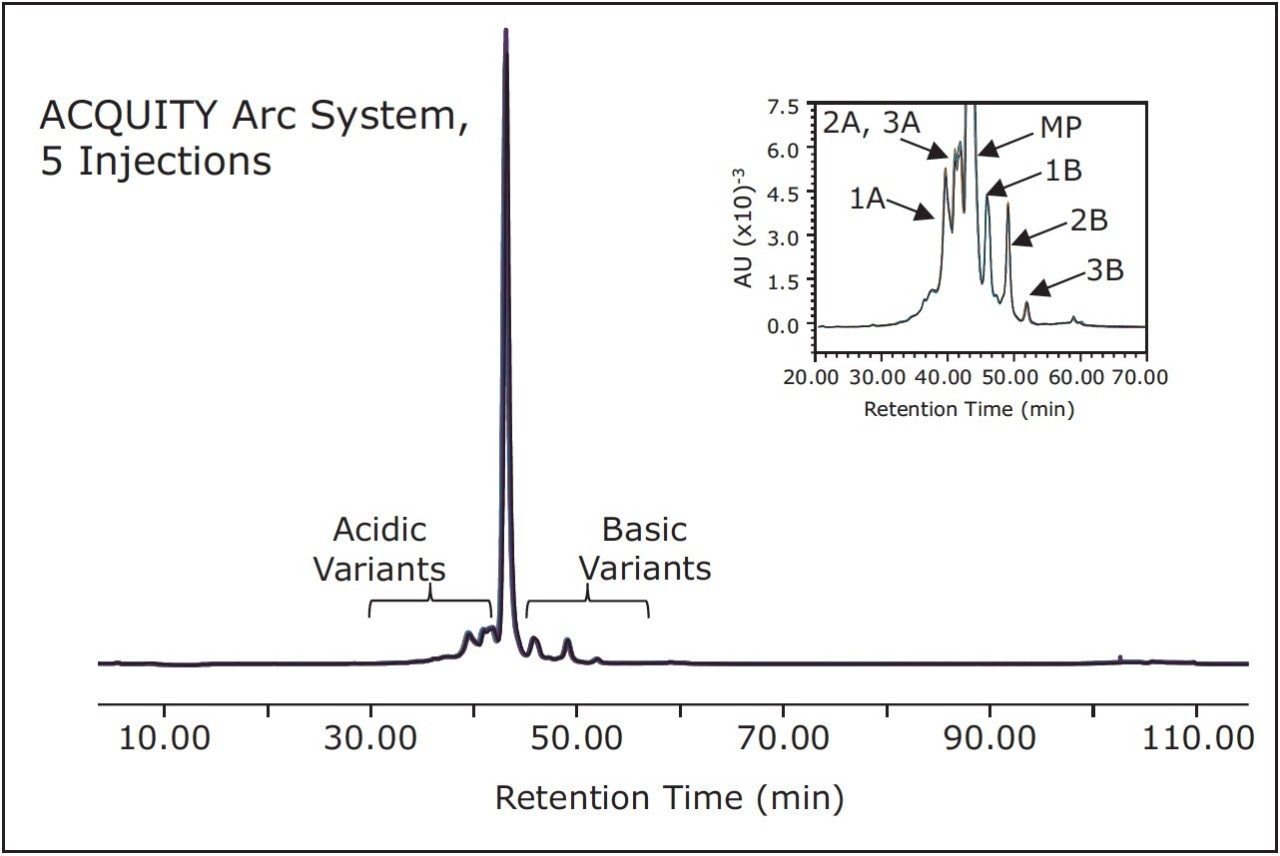
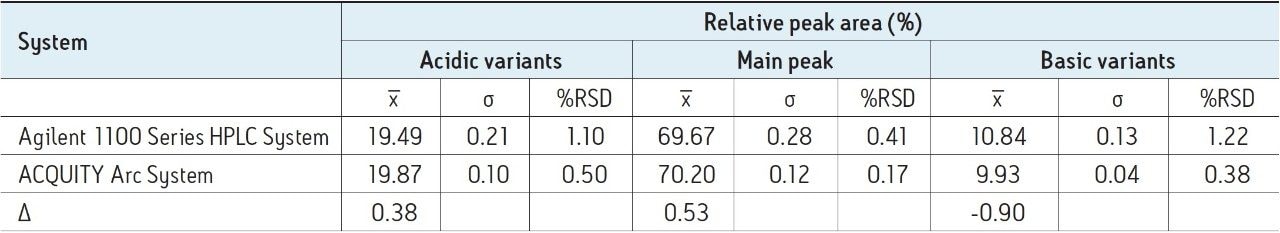
Table 2 provides quantitative analysis of the data displayed in Figure 2 by reporting peak area percent of the main peak, acidic variants, and basic variants and comparing these percentages to five injections made on the Agilent 1100 Series HPLC System. Overall, the values reported between the two systems are comparable. The difference in peak areas between the systems for the main peak, acidic variants, and basic variants are 0.53%, 0.38%, and 0.90%, respectively.
The ACQUITY Arc System offers users the ability to emulate legacy HPLC methods or update methods to UHPLC separations through the use of Arc Multi-flow path technology. This application note demonstrated the ease of transferring an IEX method from an Agilent 1100 Series HPLC System to the ACQUITY Arc System (Path 1). When keeping all method parameters the same, resulting chromatograms had relative retention times that were virtually indistinguishable from one another. Further inspection of five replicate injections on the ACQUITY Arc System shows a high degree of reproducibility.
720005529, November 2015