
Using the Waters AutoPurification System, separation methods can be developed on an analytical scale and transferred to preparatory scale on the same system, reducing a laboratory's overall capital investment. Here we illustrate a systematic approach to scale-up using the separation of Kudzu (Pueraria lobata) root extract to move from a 4.6 mm I.D. analytical column separation through 10, 19, and 30 mm I.D. preparatory column separations. The Waters OBD Prep Calculator is featured.
Chromatographic separation methods can be developed on any scale. To minimize the consumption of sample and solvents, there is a benefit in developing separation methods on a small scale and transferring them to a larger scale. Taking into account the important parameters and applying appropriate scaling factors, in a logical manner, enables users to scale up from analytical chromatography to larger-scale preparative separations easily and successfully. In this application note, the analytical-scale separation of Kudzu (Pueraria lobata) root extract is used to demonstrate the calculations and techniques used to move from a 4.6 mm I.D. analytical column separation through 10, 19, and 30 mm I.D. preparatory column separations.
Kudzu is a climbing, woody or semi-woody, perennial vine with a tuberous root. The roots of Kudzu contain a number of potentially useful isoflavones, including daidzein, daidzin, genistein, genistin, and quercetin. Kudzu is also a unique source of the isoflavone puerarin. Kudzu root extracts are thought to reduce alcohol intake and reduce alcohol withdrawal symptoms. Antibacterial, anti-cancer, anti-inflammatory, and antioxidant effects have also been noted.1

Kudzu root pieces (20 g) were added to 100 mL of 9:1 water/methanol and shaken for one hour, allowed to stand overnight, and shaken for one additional hour. This extract was centrifuged at 3000 RPM for 20 minutes and used without further treatment.
Chromatographic separations, at all scales, were carried out using the Waters AutoPurification System (Figure 1), which consisted of the following components:
|
Pump: |
Waters 2545 Binary Gradient Module |
|
Detectors: |
Waters 2998 Photodiode Array, Waters 3100 Mass Detector |
|
Injector/collector: |
Waters 2767 Sample Manager |
|
Column management: |
Waters System Fluidics Organizer An initial analytical-scale separation was developed on a Waters SunFire C18, 5 μm, 4.6 x 50 mm Column, using the conditions described below. |
|
Column temp.: |
Ambient |
|
Flow rate: |
1.5 mL/min |
|
Mobile phase A: |
Water + 0.1% Formic acid |
|
Mobile phase B: |
Methanol |
|
Gradient: |
5% to 70% B over seven minutes |
|
Injection vol.: |
20 μL |
|
Detection: |
UV (200 to 400 nm) and MS Full Scan 150 to 700 m/z |
The resulting chromatogram (Figure 2) showed a number of resolvedcompounds and was considered an acceptable candidate for scale-up.
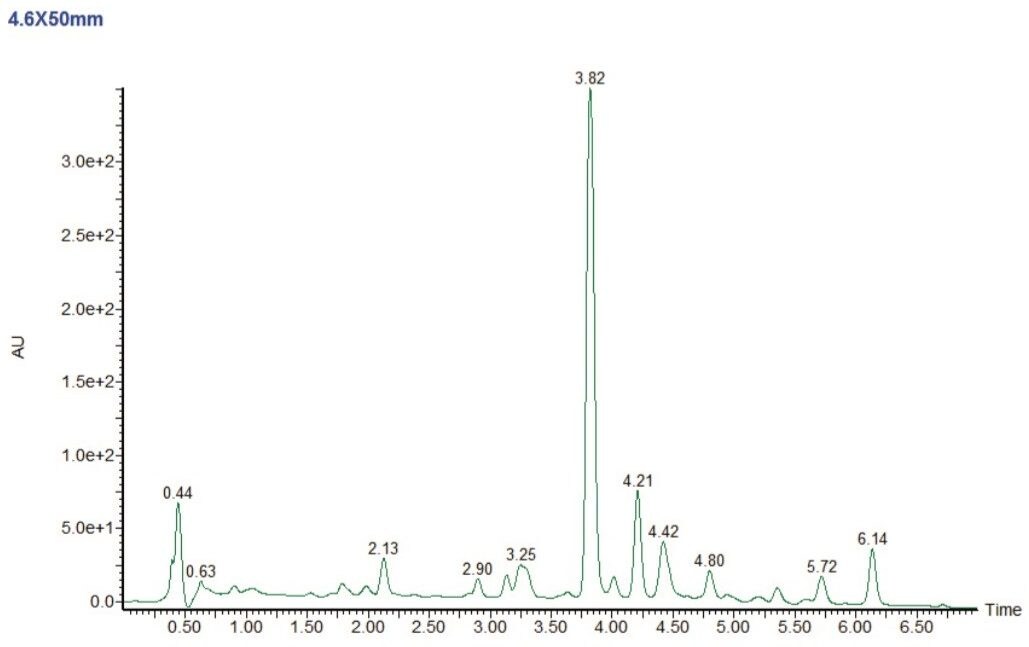
A systematic approach to scale up will provide the best possible result. The ultimate goal is to maintain chromatographic resolution between key components and enable users to better predict chromatographic performance between analytical and preparative chromatography.
There are a number of key factors to consider when approaching this scale-up process.
The heart of the separation is the column. Ideally, you should choose column chemistries that are identical. If the analytical and preparative columns are of different chemistries, it becomes very difficult to predict the preparative separation based on the analytical results. Waters offers a wide range of column chemistry choices available in analytical- and preparative-scale dimensions. As well as the chemistry itself, particle size should also be considered. Columns of the same particle size will provide similar resolution of critical pairs at both separation scales. Column length also influences the separation efficiency; columns of identical length, when scaled, give similar separation power. It is possible to scale to shorter or longer columns, but keep in mind that the separation will change.
To maintain peak shape and loading capacity, the injection volume needs to be suitably scaled using the following equation:
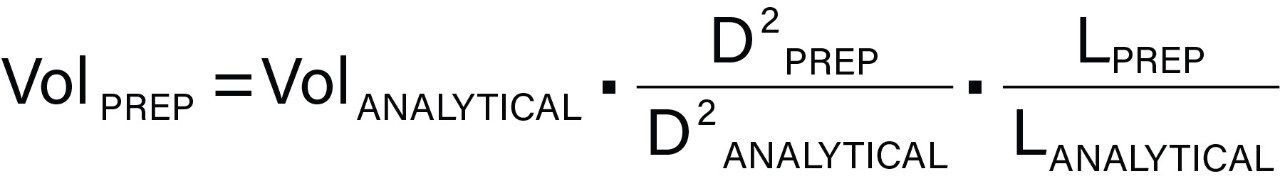
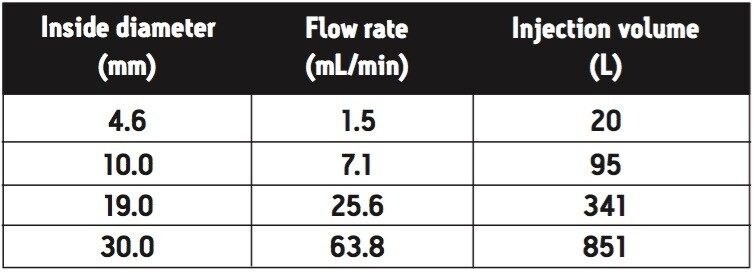
where Vol is the injection volume (μL), D is the inner diameter of the column (mm), and L is the column length (mm). For example, a 20 μL injection on a 4.6 x 50 mm column corresponds to a 341 μL injection on a 19 x 50 mm preparative column.
To maintain separation quality, the flow rate must be scaled based on column dimensions. With columns of identical particle size, the following equation is used to geometrically scale flow rate:

where F is flow rate (mL/min) and D is the inner diameter of the column (mm). For example, a 1.5 mL/min flow rate on a 4.6 mm I.D. column equates to a 25.6 mL/min flow rate on a 19 mm I.D. column.
When columns are of identical length, no changes to the gradient profile are required. If scaling to longer or shorter columns, the gradient segment volume must be maintained to preserve the separation profile.
The Waters Optimum Bed Density (OBD) Prep Calculator, a free download, (Figure 3) is an easy-to-use tool that aids in these analytical-to-preparative scaling calculations (www.waters.com/prepcalculator). The Waters OBD Prep Calculator was used to convert the analytical separation method to the preparatory separation methods described in this application note.
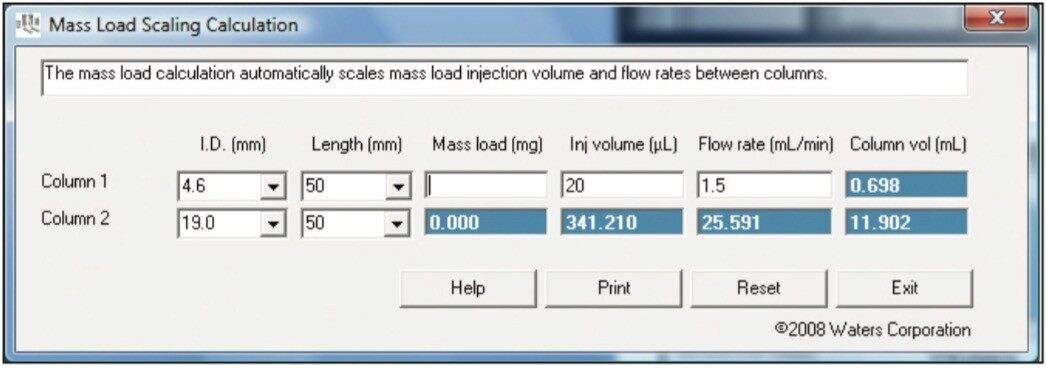
To calculate injection volume and flow rates, select the mass load scaling calculation (Figure 4) from the opening screen. Input your analytical and preparative column dimensions, analytical flow rate, and injection volume and the calculator returns the correct preparative values.
If your column lengths are identical, you can simply input the preparative flow rates into your gradient table using the same gradient segment times as your analytical method. Alternatively, for gradient methods, choose the basic gradient scalar calculation (Figure 5) from the opening screen, select your analytical and preparative column dimensions, input your analytical gradient table, and click the Calculate button. The preparative gradient table is automatically calculated and shown on the bottom half of the page. The Waters OBD Prep Calculator User Guide gives detailed instructions on use of all calculator functions.
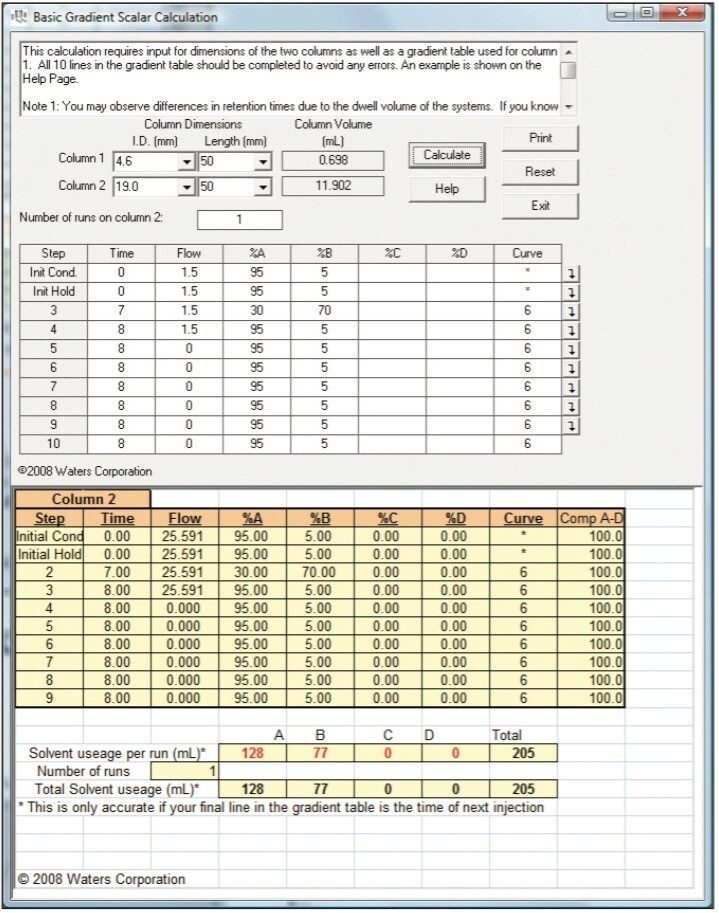
To demonstrate the previously described techniques, the analytical separation method described in the experimental section was scaled to three different preparative dimension columns (10.0, 19.0, and 30.0 mm I.D.). The scaled flow rates and injection volumes (all calculated using the Waters OBD Prep Calculator) are shown in Table 1.
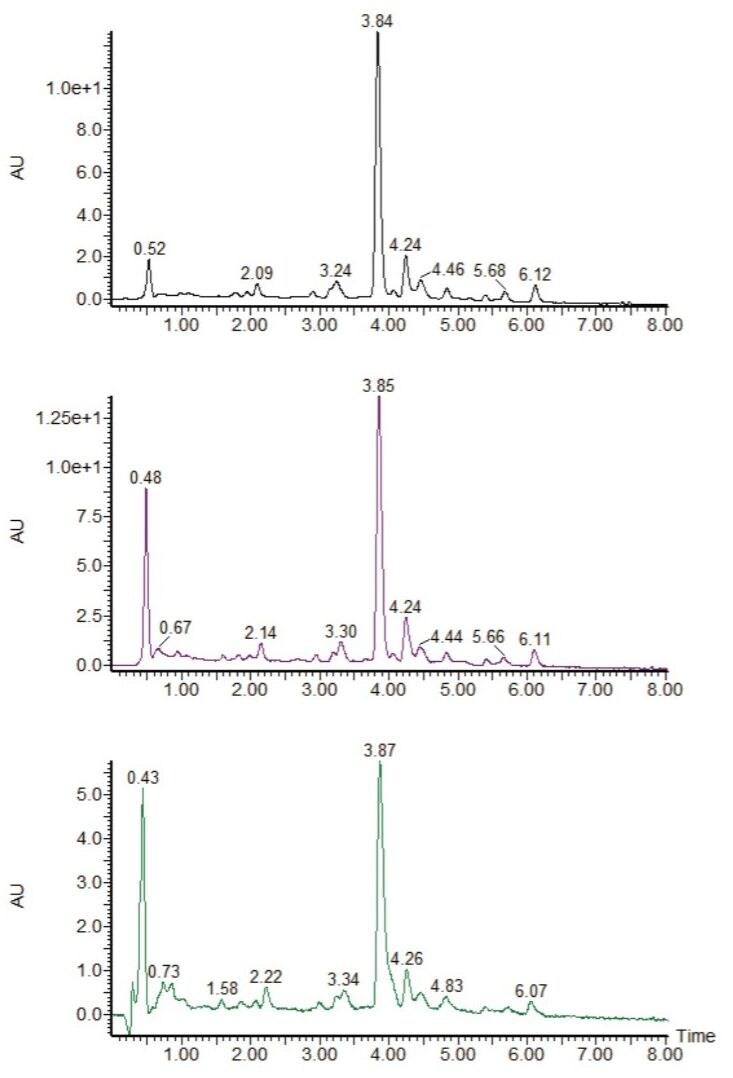
All of the preparative columns are SunFire Prep C18 OBD, 5 μm, 50 mm in length, and all of the separations were performed on the same system as the analytical-scale chromatography. As can been seen in Figure 6, regardless of the scale, the chromatography (UV TIC) is very similar. When compared to the original 4.6-mm I.D. scale (Figure 2), it can be seen that in terms of resolution and retention time the chromatography is again very similar.
This simple experiment demonstrates that a systematic approach to scale up meets the goal of maintaining chromatographic resolution between key components, and enables users to better predict chromatographic performance between analytical and preparative chromatography. This exercise also demonstrates the unique capability of the Waters AutoPurification System, which allows users to perform both analytical and preparatory chromatography on the same system with no performance compromise.
Analytical chromatography can be successfully scaled to preparatory chromatography easily by using a systematic approach.
720003120, June 2009