
The reversed-phase separation of proteins is a useful analytical technique and acetonitrile has been the solvent of choice because of good peak shape and resolution. Alcohol-based mobile phases can be substituted for acetonitrile in reversed phase chromatography of proteins. There is little change in selectivity with the different solvents. These guidelines provide a straightforward approach to mitigating the effects of the acetonitrile shortage on routine reversed-phase chromatography of proteins.
The reversed-phase separation of proteins is a useful analytical technique that is used in a variety of applications and at various stages of the biopharmaceutical development process. Acetonitrile has been the solvent of choice because of good peak shape and resolution. It also minimizes viscosity while providing transparency at low UV wavelengths for sensitive detection. Other organic solvents have been used for either improved recovery or different selectivity, but there has been little need for changing the routine use of acetonitrile.
Recently, however, a combination of economic, social, and environmental factors has created an acute shortage of acetonitrile accompanied by a substantial price increase. It is, therefore, necessary to examine more closely the solvent options for reversed-phase protein separations. It is especially important to quickly make wise choices that meet the financial requirements without sacrificing the quality of the separations.
Several organic solvents were evaluated. The test mixture of proteins used in the evaluation represents a wide range of properties, including molecular weights from 10 kD to 150 kD, isoelectric points from 4.5 to above 10, and eluting anywhere from 20% to well-above 50% acetonitrile. Three monoclonal antibodies were also used as test samples. The effect of temperature and pressure were observed and summarized for each of these solvents.
Protein Mixture (in 0.1% CF3COOH in 5% acetonitrile)
Ribonuclease A, bovine pancreas: 0.08 mg/mL
Cytochrome c, horse heart: 0.11 mg/mL
Albumin, bovine serum: 0.40 mg/mL
Myoglobin, horse heart: 0.25 mg/mL
Enolase, baker’s yeast: 0.43 mg/mL
Phosphorylase b, rabbit muscle: 1.18 mg/mL
Mixture of monoclonal antibodies (murine, chimeric, and fully humanized): ~0.5 mg/mL in 0.1% CF3COOH
|
LC System: |
Waters ACQUITY UPLC System PEEK-Sil Needle (PN/205000507) Flow restrictor (PN/205000547) Peptide Mixer (PN/205000403) |
|
Column: |
ACQUITY UPLC BEH300 C4, 1.7 μm, 2.1 mm x 50 mm |
|
Column Temp.: |
40˚C or 80˚C, as indicated |
|
Sample Temp.: |
4˚C |
|
Flow Rate: |
200 μL/min. |
|
Mobile Phase A: |
0.1% CF3COOH in DI water |
|
Mobile Phase B: |
0.1% CF3COOH in indicated solvent |
|
Weak Needle Wash: |
0.1% CF3COOH in 5% Acetonitrile 600 μl |
|
Strong Needle Wash: |
0.1% CF3COOH in 75% Acetonitrile 200 μl |
|
Injection Volume: |
3.3 μL, Partial Loop |
|
Detection: |
UV (TUV), 220 nm |
|
Time |
%A |
%B |
Curve |
|---|---|---|---|
|
0.00 |
80.0 |
20.0 |
- |
|
29.06 |
28.6 |
71.4 |
6 |
|
31.60 |
28.6 |
71.4 |
1 |
|
49.60 |
80.0 |
20.0 |
1 |
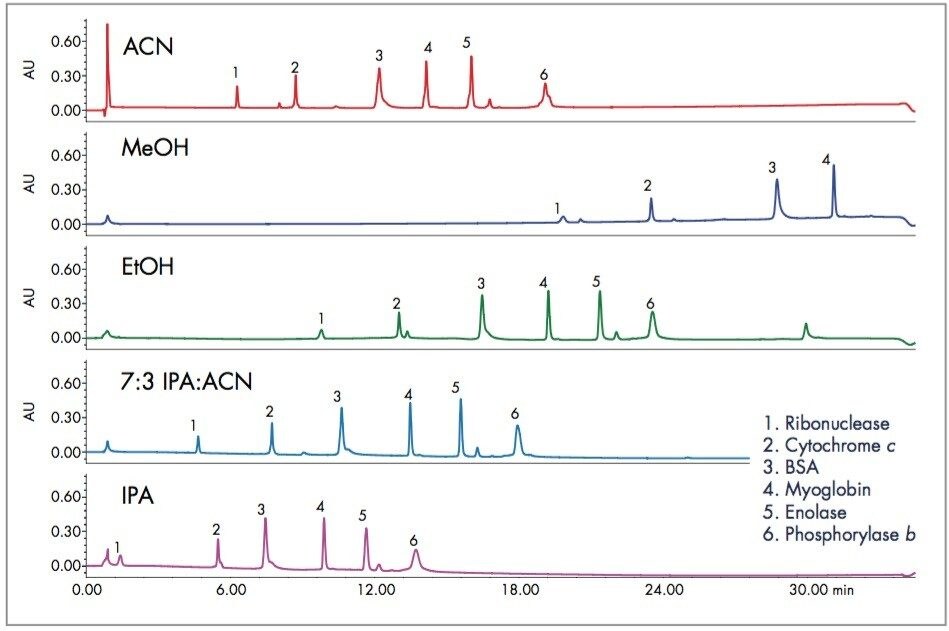
A standard mixture of proteins was separated using a gradient of increasing acetonitrile. The acetonitrile was replaced with alcohols of progressively longer chain length as shown in Figure 1. A blend of acetonitrile and isopropanol was included in the series, as this mixture has often been used in the past. The blend has lower viscosity than isopropanol while retaining improved resolution and recovery. Methanol gave wide, asymmetrical peaks and did not elute all the proteins within the standard gradient. Ethanol did give a reasonable separation with increased retention compared to acetonitrile. Replacing seventy percent of the acetonitrile with IPA gave good separation with slightly less retention for all of the proteins. Using 100% IPA resulted in even less retention for the proteins, and with reduced resolution for some proteins, such as Phosphorylase b.
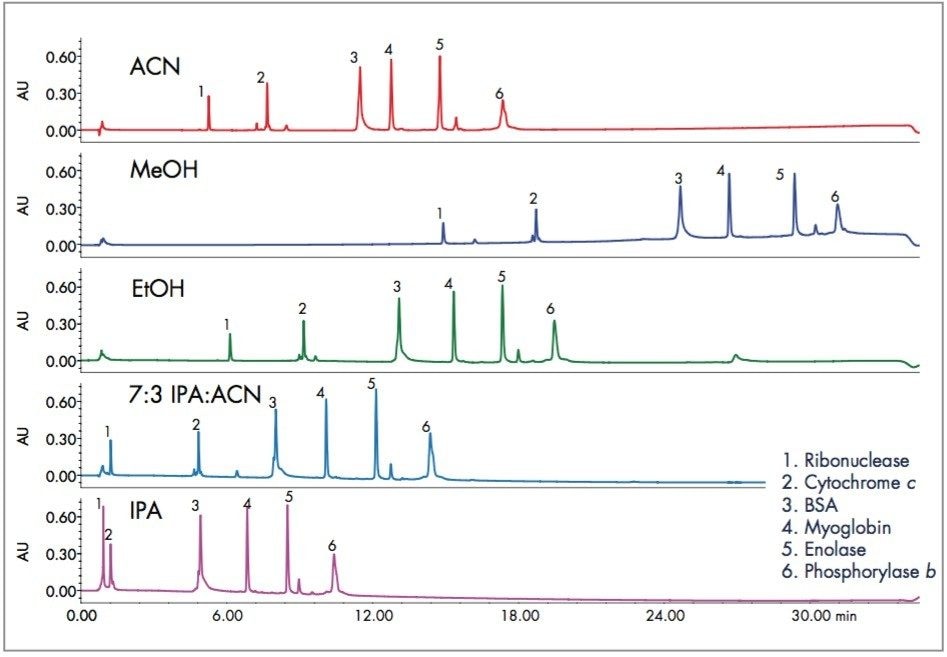
Increasing the temperature for the analysis resulted sharper peaks and reduced retention for all proteins, as shown in Figure 2. While methanol was not a reasonable choice at 40°C, it gave acceptable peak shape within the standard gradient at 80°C. The rest of the standard proteins shifted to lower retention at the higher temperature and generally gave narrower peaks. The shift to lower retention could compromise the analysis of polar proteins, but this gradient uses 20% organic as the initial conditions. The gradient could be modified to lower starting organic concentration as required for the least retained proteins.
The same series of experiments was repeated with a mixture of monoclonal antibodies as show in Figure 3. The same patterns are observed as with the protein standard mixture. Methanol is unacceptable for these proteins, even at the highest temperature. Isopropanol appears to give the best peak shape and resolution.
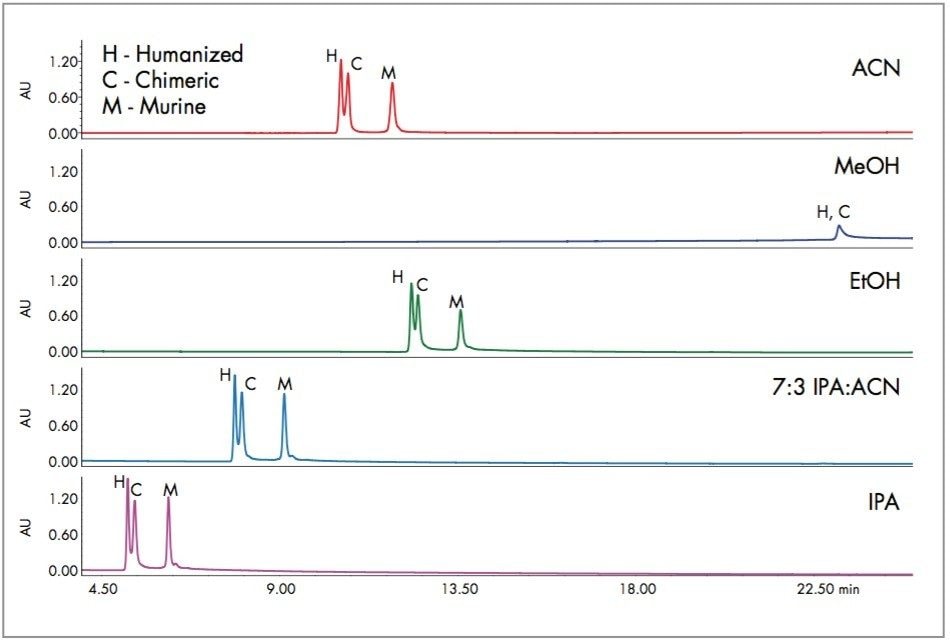
In the experiments comparing different solvent combinations and temperatures, with both the protein standard mixture and the monoclonal IgG’s, there is little or no evidence of selectivity changes. This suggests that the solvent substitutions may be made as convenient without the need for extensive re-development of methods. Re-validation would, of course, be required for regulated methods.
The major challenge in replacing acetonitrile with an alcohol is the relatively high viscosities associated with water alcohol mixtures. Some combination of increased temperature and reduced flow rate can be used to manage the higher pressures. Table 1 shows the approximate system pressures for an ACQUITY UPLC system, fitted with a pressure restrictor and peptide mixer, for the different solvent and temperature combinations, all tested at 0.2 mL/min. The relative pressures will be the same on all systems so this table is a useful guide for making substitutions. Isopropanol gives almost three times the back pressure of acetonitrile. Raising the temperature from 40-80°C reduces the pressure for all solvents by about 20%. Regardless of the solvents and temperatures tested, the ACQUITY UPLC system has sufficient pressure capacity to handle the separation. While the flow restrictor was installed and used for single variable testing of all solvents, its use above 10k PSI will likely result in leaks at the fittings of the restrictor, and is therefore not recommended for use with isopropanol or ethanol.
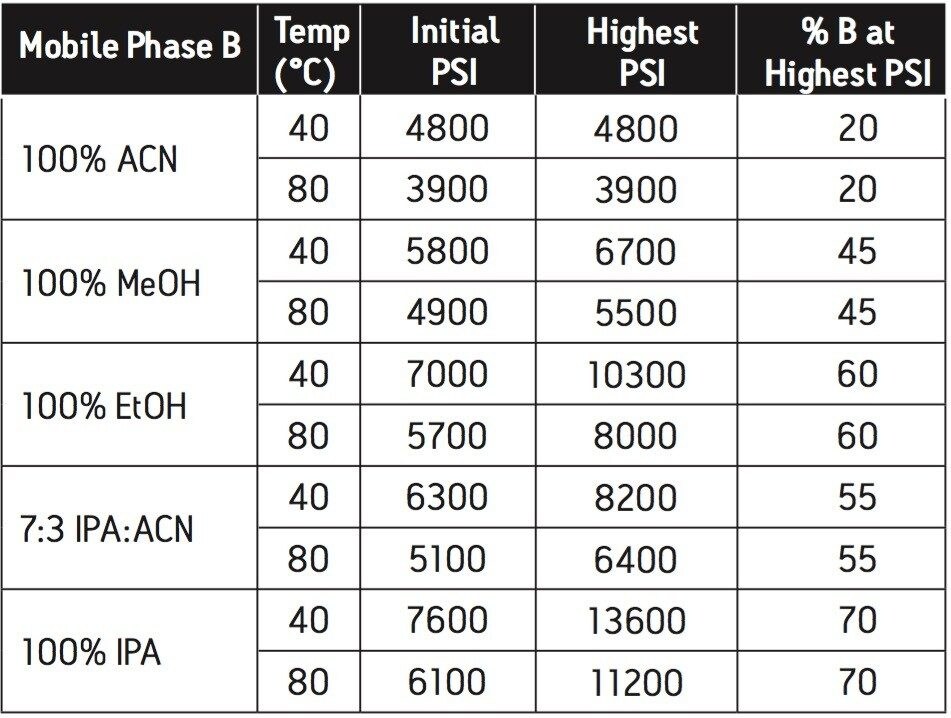
Alcohol-based mobile phases can be substituted for acetonitrile in reversed phase chromatography of proteins. Isopropanol is much superior to methanol for this purpose. Elevating the temperature of the separation improves peak shape in all the solvents and for all the samples tested. There is little change in selectivity with the different solvents. These guidelines provide a straightforward approach to mitigating the effects of the acetonitrile shortage on routine reversed-phase chromatography of proteins.
720003056, June 2009