This application note describes the evaluation of UniSpray ionization for analysis of PFAS in various environmental samples, as well as showing a direct comparison to typical electrospray ionization (ESI) for the same set of samples.
Perfluorinated alkyl substances (PFAS) are ubiquitous environmental pollutants garnering widespread attention around the globe. They are most commonly found in water and soils but are contaminants of concern in many sample types. PFAS are used in a range of consumer goods and industrial processes due to their chemical properties. They are commonly used in coatings to impart water resistance and are even used in firefighting foams. Occurrence of PFAS in the environment can come from release from industrial facilities creating or using PFAS, normal use and disposal of consumer products containing PFAS, and from use of firefighting foams. PFAS are classified as persistent organic pollutants (POPs) within the Stockholm Convention and their impact on humans and the environment are being closely monitored by organizations globally.
Detection requirements for PFAS in environmental samples are typically in the ng/L or parts per trillion (ppt) range, requiring sensitive analysis methods, most typically LC-MS/MS. PFAS respond well using negative electrospray ionization (ESI-) and this has become the preferred method of analysis. Although this ionization technique works well, the increasingly stringent requirements for detection can benefit from any boost in signal that a technique like UniSpray could provide.
UniSpray is a novel atmospheric ionization technique that allows for multimode ionization of both polar and non-polar analytes in a single injection. The column effluent is nebulized as it exits a grounded, heated probe. The spray is directed onto a stainless-steel pin that is held at high voltage. This creates smaller droplets than ESI, leading to increased desolvation of ions. The nebulized flow bends around the surface of the impactor pin into the sample cone due to the Coanda effect. This mechanism allows for increased ionization and sampling efficiency.1,2
A picture of the Waters UniSpray ion source is shown in Figure 1. Several comparative studies between UniSpray and electrospray on various classes of compounds have shown an enhancement in response using the UniSpray ionization technique.3,4,5

In this application note, we describe the evaluation of UniSpray ionization for analysis of PFAS in various environmental samples, as well as showing a direct comparison to typical electrospray ionization (ESI) for the same set of samples.
Since required detection limits are in the low ng/L range and as a result of the widespread use of PFASs, specific challenges must be addressed for sample collection, preparation, and analysis. There are many common sources of PFAS contamination in the field and laboratory. In the field, caution should be taken to avoid Teflon-containing materials including waterproof clothing/jackets, plastic clipboards, waterproof notebooks, and chemical ice packs, for example. In the lab, items to avoid include sticky notes, certain disposable glass pipettes, aluminum foil, vial caps with Teflon seals, and LDPE containers to name a few. In fact, as is practical, it is recommended that any laboratory supplies be checked for PFAS contamination before use. PFAS contamination can also result from the chromatographic system due to the use of Teflon-coated parts. Therefore, steps should be taken to minimize any system contribution, and as such, the Waters PFAS Analysis Kit (p/n: 176004548) for the UPLC system was utilized in the experiment. The kit is comprised of PFAS-free components (such as PEEK tubing to replace the conventional Teflon-coated solvent lines) and an isolator column that helps to delay any residual background interferences from co-eluting with the analytical peak. Installation of the PFAS Analysis Kit is straightforward and quick.6
Water and soil samples were provided by US EPA Region 5 through a Cooperative Research and Development Agreement (EPA CRADA #884-16). Water samples included reagent water, surface (river) water, ground water, influent waste water, and effluent waste water. Soil samples included sand, silt, lean clay, and fat clay. All samples were spiked with a selection of PFAS compounds at varying concentrations prior to being received in the lab.
Water samples were prepared in accordance with the ASTM 7979 method.7,8 The entirety of each 5-mL water sample was used to avoid any compound loss. Each sample was spiked with 160 ng/L of isotopically labeled surrogates. Five milliliters of methanol was then added to each water sample and vortexed. The entire 10-mL sample was filtered using a disposable polypropylene syringe with a glass filter (25 mm diameter, 1.0 μm pore size) stacked on top of a polypropylene GHP filter (25 mm diameter, 0.2 μm pore size). Following filtration, 10 μL of acetic acid was added to each sample. An aliquot of each sample was transferred to a polypropylene autosampler vial and sealed with a polyethylene cap (p/n: 186005230). Soil samples were prepared in the following manner in accordance with the ASTM 7968 method.9 Two grams of soil was diluted with 10 mL of 1:1 water:methanol. Sample pH was adjusted to 9–10 using 20 μL of ammonium hydroxide. Samples were shaken using a mechanical shaker for one hour, followed by centrifugation at 1900 rpm for 10 minutes. The entire supernatant was filtered using a disposable polypropylene syringe with a dual glass fiber and GHP membrane filter. Following filtration, 50 μL of acetic acid was added to each sample. An aliquot of each sample was transferred to a polypropylene autosampler vial and sealed with a polyethylene cap.
Any sample preparation technique suitable for PFAS analysis could be used prior to UniSpray analysis. Please see application notes 720006471EN and 720006695EN for alternate SPE preparation techniques.10,11
|
LC system: |
ACQUITY UPLC H-Class PLUS fitted with PFAS kit (p/n: 176004548) |
|
Column: |
ACQUITY UPLC BEH C18 2.1 × 100 mm, 1.7 μm (p/n: 186005407) |
|
Column temp.: |
35 °C |
|
Sample temp.: |
10 °C |
|
Injection volume: |
30 μL |
|
Mobile phase A: |
95:5 Water:methanol + 2 mM ammonium acetate Mobile phase B: Methanol + 2 mM ammonium acetate |

|
MS system: |
Xevo TQ-S micro |
|
Ionization mode: |
ESI- |
|
Capillary voltage: |
0.5 kV |
|
Desolvation temp.: |
350 °C |
|
Desolvation gas flow: |
900 L/hr |
|
Cone gas flow: |
50 L/hr |
|
Source temp.: |
110 °C |
|
MS system: |
Xevo TQ-S micro |
|
Ionization mode: |
UniSpray |
|
Impactor voltage: |
1.0 kV |
|
Desolvation temp.: |
400 °C |
|
Desolvation gas flow: |
900 L/hr |
|
Cone gas flow: |
100 L/hr |
|
Source temp.: |
110 °C |
MRM parameters for each compound were optimized in electrospray mode using the QuanOptimize tool in MassLynx and are listed in Appendix A. These parameters were also used for the UniSpray analysis.
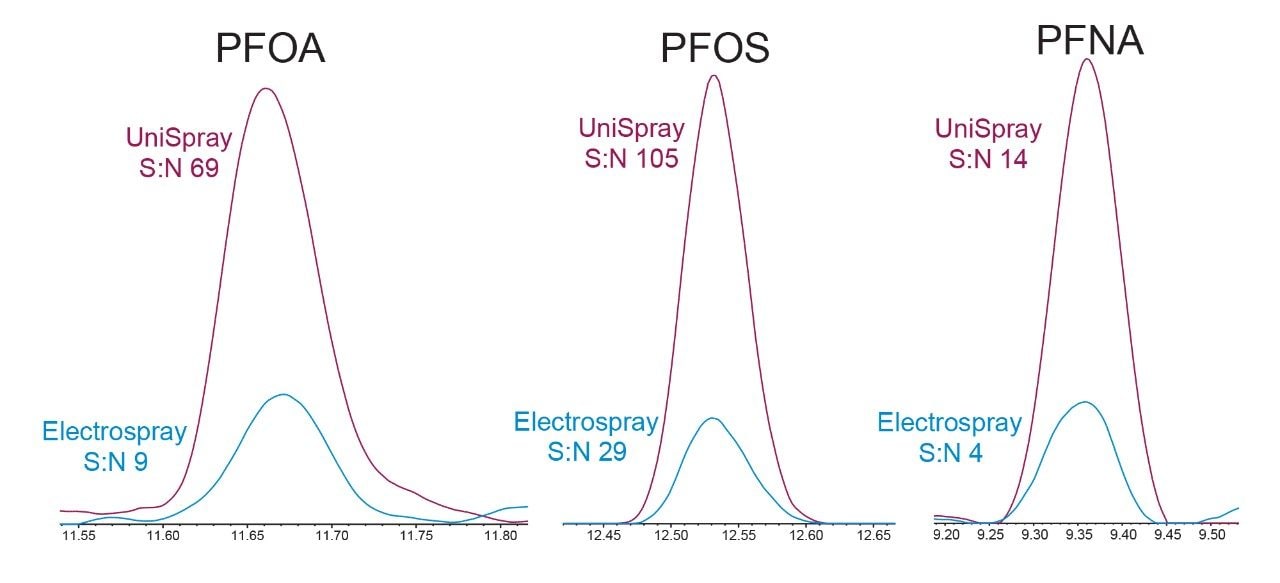
The performance of UniSpray ionization for PFAS was compared to the well-characterized technique of electrospray by running the same samples using both source options. Source conditions were optimized for each ionization technique to ensure that optimal conditions were being compared. Peak response, peak area, and signal:noise (S:N) were all evaluated for comparison of the two techniques. A wide variety of PFAS were evaluated, including legacy carboxylates and sulfonates, precursor compounds, and emerging PFAS. When using UniSpray ionization, a majority of the PFAS evaluated resulted in increased intensity and peak area as compared to electrospray ionization. All remaining compounds had very similar peak response and area from both ionization techniques. S:N, when using UniSpray as the ionization technique, was equal to or better for all PFAS evaluated when compared to electrospray. Figure 2 demonstrates three compounds (PFOA, PFOS, and PFNA) with enhanced peak response, area, and S:N when using UniSpray ionization as compared to electrospray.
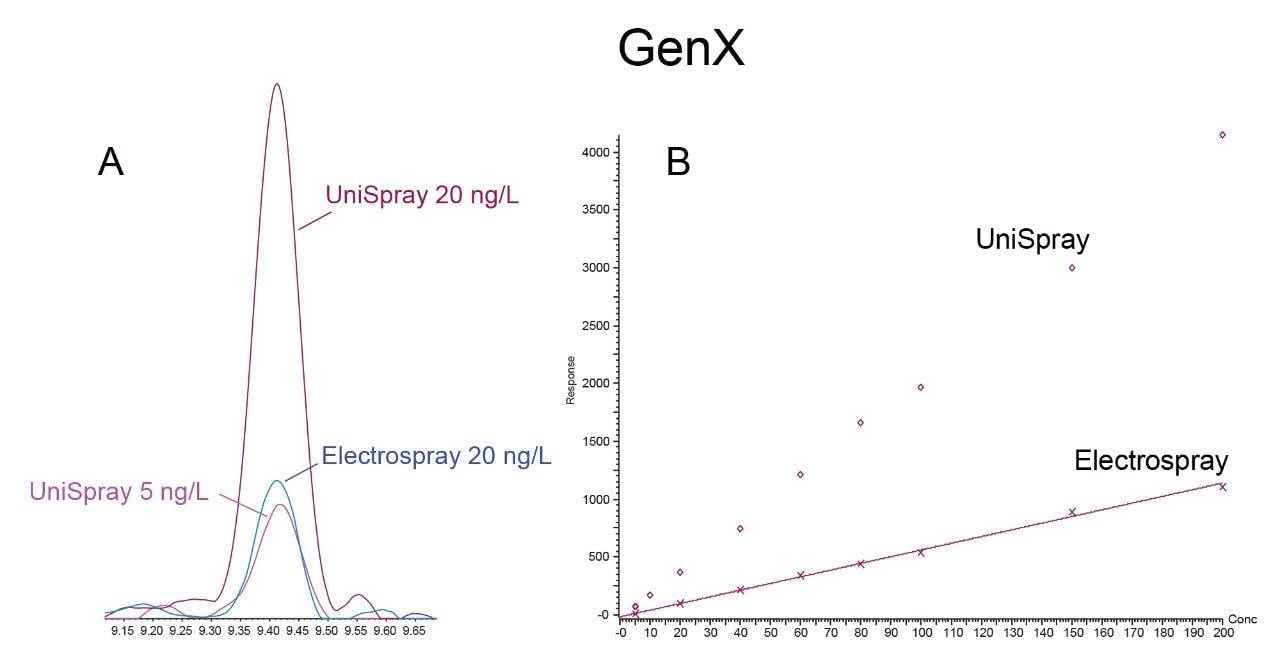
The increase in both peak area and S:N allows for lower detection limits to be achieved. This is demonstrated by the data highlighted in Figure 3A that shows GenX as an example. The limit of detection generated by electrospray ionization based on peak response and S:N generated was about 20 ng/L, whereas UniSpray produced a similar peak response and S:N at 5 ng/L. This would allow a reduction in the limit of detection by approximately at least 4x when using UniSpray for GenX.
Compounds that only had similar S:N but also had an increase in peak area would still benefit from analysis using UniSpray, as this would allow for easier automated integration and more accurate quantitation at lower concentrations. This idea is demonstrated in Figure 3B by plotting both the UniSpray and electrospray calibration curves together.
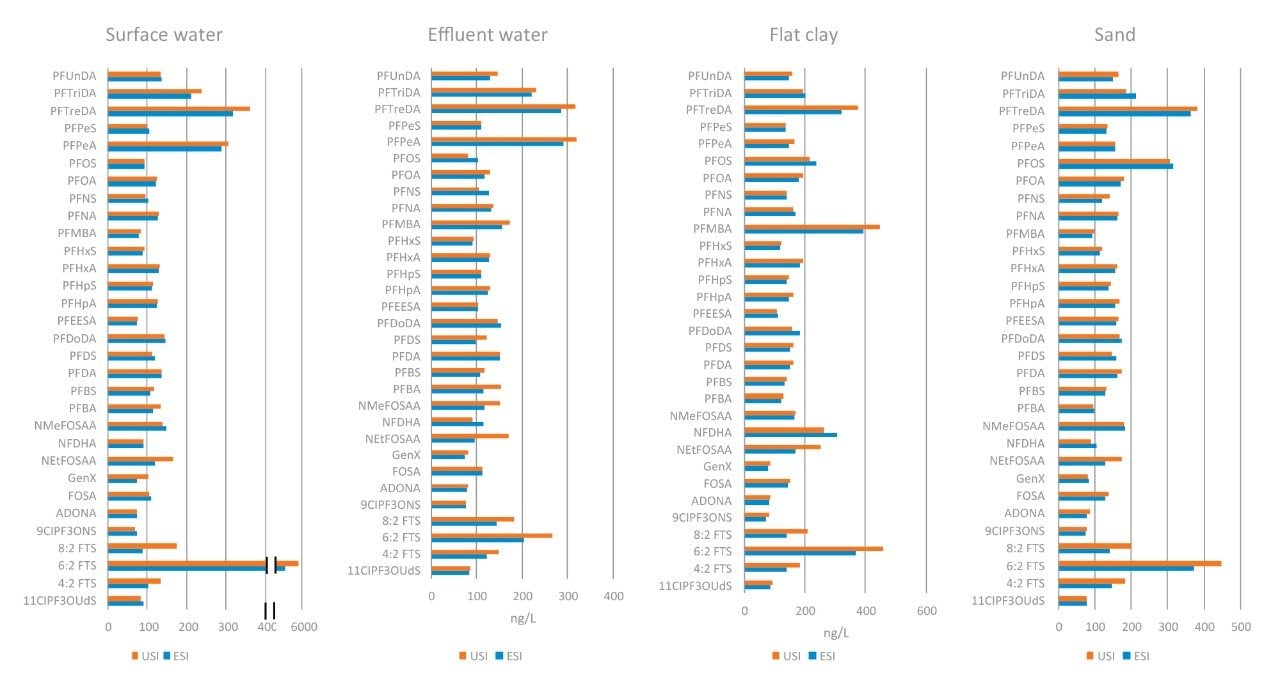
A variety of water (surface, ground, and waste water) and soil (sand, silt, and clay) samples were evaluated using both ionization techniques to understand the effect of sample matrix. Concentrations of each compound in matrix were calculated and compared to ensure UniSpray did not bias the accuracy due to increased peak response. Figure 4 shows the calculated concentrations determined in four of the samples (surface water, effluent water, fat clay, and sand) using both ionization techniques. As demonstrated in the figure, the calculated concentrations from UniSpray and electrospray were very comparable, having a mean percent difference of approximately 6%, indicating UniSpray is a suitable and accurate alternative ionization technique for routine sample analysis and quantitation. This also demonstrated the increase of ionization efficiency experienced in UniSpray does not create any change in matrix interference or matrix effects.
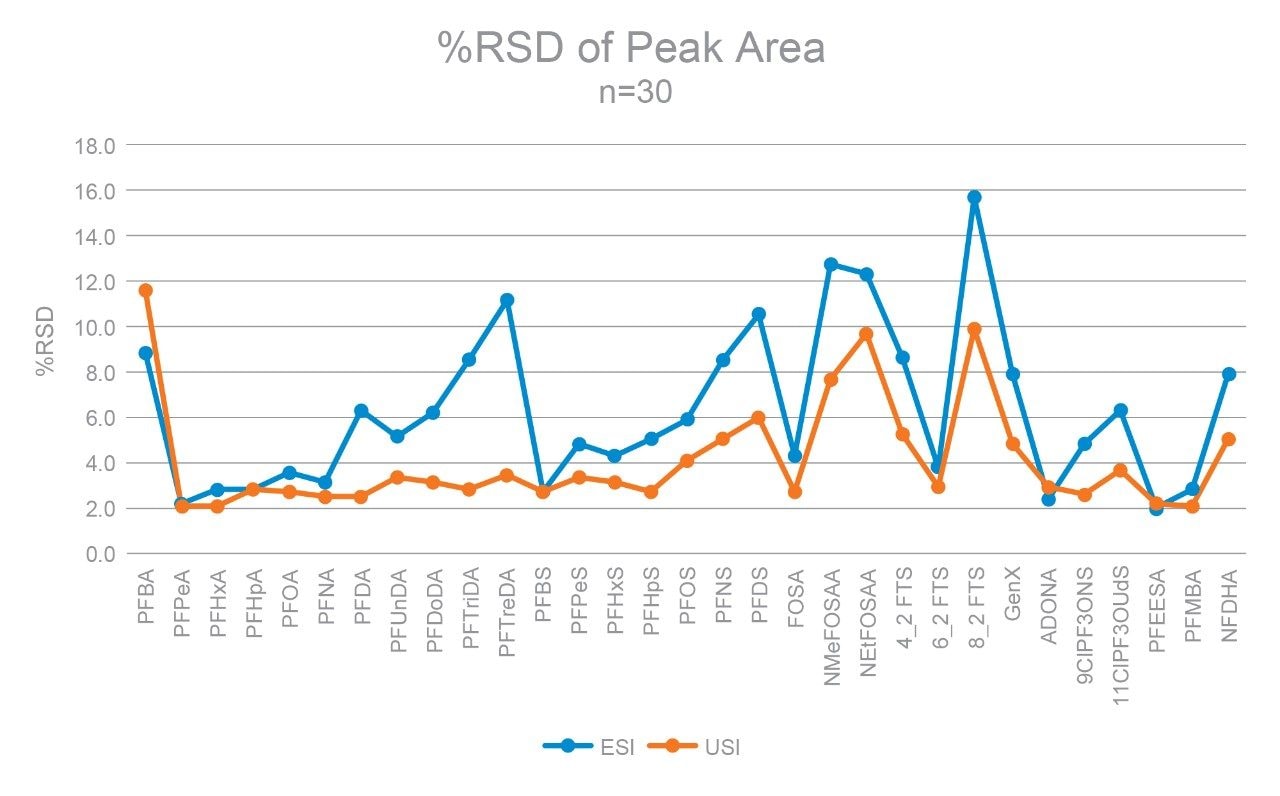
Robustness was also evaluated and compared for each ionization technique by performing 30 replicate injections of a surface water sample and comparing the %RSD of the replicate injections. The %RSD of the peak areas across the 30 injections were all within 15% (Figure 5). A majority of the peak area RSDs from the electrospray injections were under 10%, while a majority were under 5% for UniSpray. The slight reduction of RSD experienced by UniSpray could potentially be attributed to the increased peak areas, which inherently reduce uncertainty in these values. The robustness data establishes that both techniques are valid options for routine PFAS analysis.
720006760, February 2020