
In this application note, the key factors that affect the separation of lycopene, beta-carotene, lutein, and coenzyme Q10 by UPC2 were investigated.
Carotenoids form an important class of natural pigments that are found in microorganisms, plants, and animals. They are fat-soluble compounds and play an important role in physiological and biological processes. In addition to the pro-vitamin A activity of some carotenoids, these compounds have been associated with a number of health benefits. Several studies have shown that their antioxidant capacity help prevent cancer, diabetes, and cardiovascular diseases.1-2 Besides their medicinal use, the importance of carotenoids has also increased due to their use in the food, cosmetic, and feed industries.3
Coenzyme Q10 (CoQ10, ubiquinone-10) is a fat-soluble, vitamin-like nutrient that is involved in the mitochondrial respiratory chain. Like carotenoids, coenzyme Q10 is also a powerful antioxidant. Its antioxidant capacity is ten times greater than vitamin E. Many therapeutic values of coenzyme Q10 have been reported, such as prevention of lipid peroxidation and oxidative damage of hemoglobin. Furthermore, coenzyme Q10 shows promising results when administered to hypertensive patients.4-5
Supercritical fluid chromatography (SFC) has been intensively studied due to its versatility in chiral and achiral separations. The technique is suitable to the analysis of thermally unstable compounds like carotenoids and the separation kinetics is faster than in High Performance Liquid Chromatography (HPLC) due to the lower viscosity of the mobile phase.5 Many fluids employed as mobile phases in SFC are environmentally friendly, nontoxic and non-flammable. Carbon dioxide (CO2) is the most used compound for several reasons: it is not expensive; it has good miscibility with many polar and non-polar organic solvents and is easily available with analytical quality. Furthermore, its critical properties (Pc = 7.38 MPa, Tc = 304.13 K) are easily attainable.6-8 SFC experiments were initially performed with pure CO2 as the mobile phase. Nowadays, a mixture of CO2 and organic modifier is very often used in order to increase the solubility of polar compounds and allow their elution.9-11
UltraPerformance Convergence Chromatography (UPC2) is a separation technique that combines the unique properties of supercritical CO2 with the use of sub-2-µm particle columns to improve the separation efficiency, speed and selectivity. UPC2 employs CO2 as the main component of the mobile phase which reduces organic solvent consumption.12
In UPC2 method development, the stationary phase, mobile phase composition (the type and percentage of the organic modifier) and operating conditions (pressure and temperature) should be evaluated. Since CO2 is a non-polar compound, the addition of organic modifier (for example methanol, ethanol, acetonitrile, etc.) is necessary to improve the solubility of the analytes in the mobile phase and therefore extend the domain for chromatographic applications. Studies have demonstrated that retention of polar solutes is more sensitive to the percentage of the organic modifier, which is the most important parameter after the selection of the stationary phase.11, 13 Temperature and pressure also play an important role in UPC2, since they control the density of the fluid. Changes in the mobile phase density induce variable retention times which can affect the repeatability and reproducibility of the chromatographic separation. Additionally, temperature also affects the vapor pressure of the solute and influences the solubility parameters of both solute and supercritical fluid. On the other hand, when pressure is increased, a reduction is observed in retention due to an increase in the density of the supercritical fluid. Since temperature, pressure and mobile phase composition influence the chromatographic parameters, these parameters should be evaluated during method development.11, 14 In this application note, the key factors that affect the separation of lycopene, beta-carotene, lutein, and coenzyme Q10 by UPC2 were investigated. Design of Experiments (DOE) was employed to optimize the operating conditions (ethanol percentage, temperature and pressure). This study was divided into two steps: 1) selection of the stationary phase and range of organic modifier (ethanol) that provided suitable retention for all the compounds and 2) study of the density of the supercritical fluid considering pressure, temperature and the percentage of organic modifier in the mobile phase using Design of Experiments (DOE).
|
System: |
ACQUITY UPC2 |
|
Detection: |
PDA Detector (detection at 275 nm) |
|
Column: |
ACQUITY UPC2 HSS C18 SB 3.0 × 150 mm, 1.8 μm |
|
Mobile phase A: |
CO2 (tank, medical grade) |
|
Mobile phase B: |
Ethanol (5–30% (v/v)) |
|
Column temp.: |
40 °C |
|
ABPR: |
2190 psi |
|
SM temp.: |
5 °C |
|
Sample diluent: |
MTBE |
|
Injection volume: |
1 μL |
|
Flow rate: |
1.5 mL/min |
|
Vials: |
Waters Amber Glass 12 × 32 mm Screw Neck Vial, 2 mL |
|
PDA scan range: |
210 to 600 nm |
|
Strong needle wash: |
Methanol |
|
Weak needle wash: |
2-Propanol |
|
Seal wash: |
2-Propanol |
|
System: |
ACQUITY UPC2 |
|
Detection: |
PDA Detector (detection at 275 nm) |
|
Column: |
ACQUITY UPC2 HSS C18 SB 3.0 × 150 mm, 1.8 μm |
|
Mobile phase A: |
CO2 (tank, medical grade) |
|
Mobile phase B: |
Ethanol (15–24% (v/v)) |
|
Column temp.: |
25–50 °C (defined based on the maximum allowed temperature for the HSS C18 SB stationary phase) |
|
ABPR: |
1500–2200 psi |
|
SM temperature: |
5 °C |
|
Sample diluent: |
MTBE |
|
Injection volume: |
1 μL |
|
Flow rate: |
1.5 mL/min |
|
Vials: |
Waters Amber Glass 12 × 32 mm Screw Neck Vial, 2 mL PDA scan range: 210 to 600 nm |
|
Strong needle wash: |
Methanol |
|
Weak needle wash: |
2-Propanol |
|
Seal wash: |
2-Propanol |
The maximum analysis time was set at 10 min to enable the separation to be performed at a reasonable time.
Lycopene, beta-carotene, lutein, and coenzyme Q10 standards were purchased from Sigma-Aldrich. For all experiments, a mixture of the standards was prepared in MTBE in a concentration of 0.2 mg/mL and filtered through a 0.45 µm syringe filter prior to the injection. All injections were carried out in duplicate and the average was calculated.
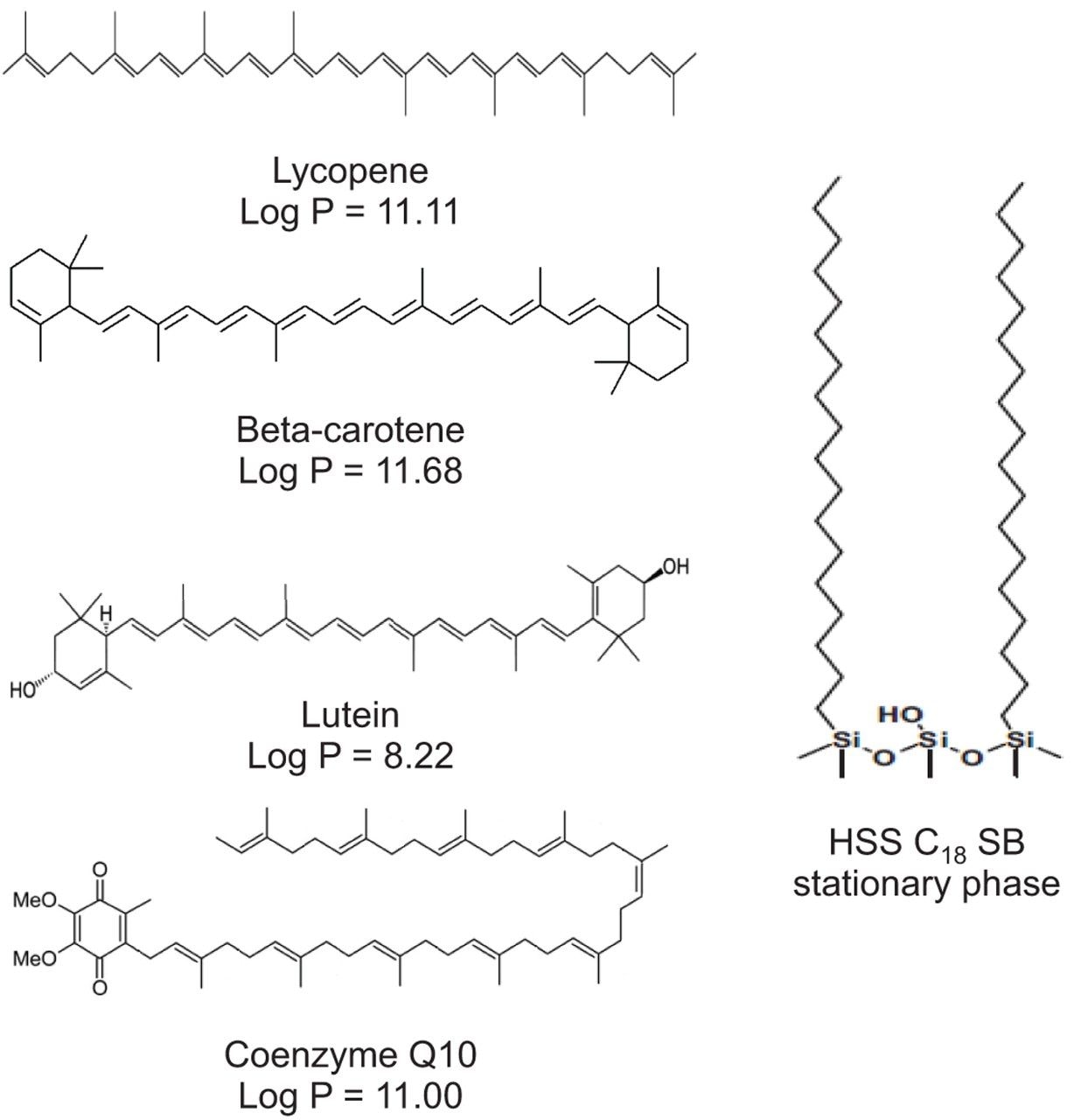
The first step of the method development consisted in the selection of a suitable stationary phase. An ACQUITY UPC2 HSS C18 SB Column was selected due to its non-polar character, which ensures the proper interaction of the non-polar bioactive compounds with the hydrophobic particle surface. The structure of the compounds and the stationary phase are illustrated in Figure 1. This stationary phase has already been described for the separation of the two geometrical isomers, lycopene and beta-carotene, using UPC.2-15
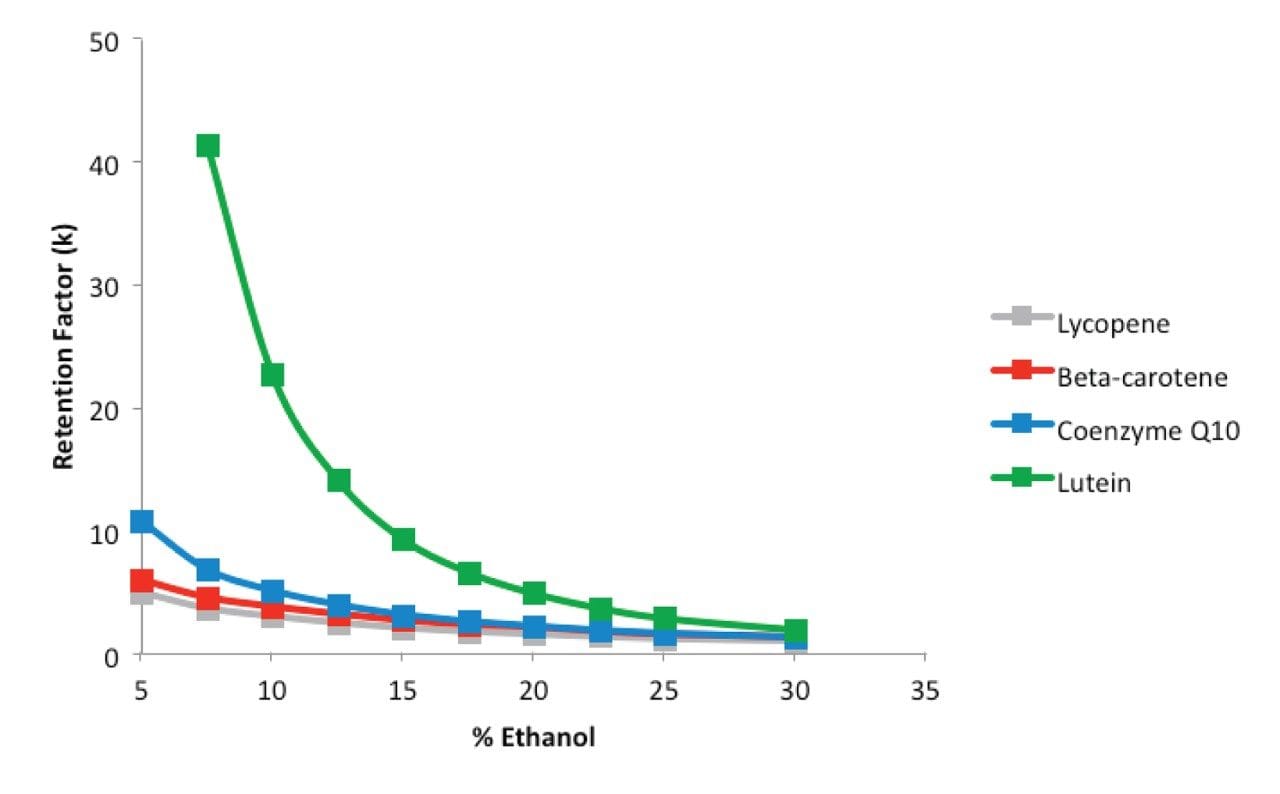
Then the range of ethanol percentage was studied to understand the retention profile of the compounds. Suitable retention factors (2<k<10) for all compounds were achieved within the range of 15 to 24% of ethanol, according to the retention curves shown in Figure 2. The use of lower amounts of ethanol could be beneficial for the separation of the critical pair, beta-carotene and coenzyme Q10. However, unacceptable retention of lutein, the highly retained compound, would occur. A DOE approach was then used to simultaneously evaluate pressure, temperature and ethanol percentage in the range of 15–24% in order to accomplish the best possible separation in isocractic mode.
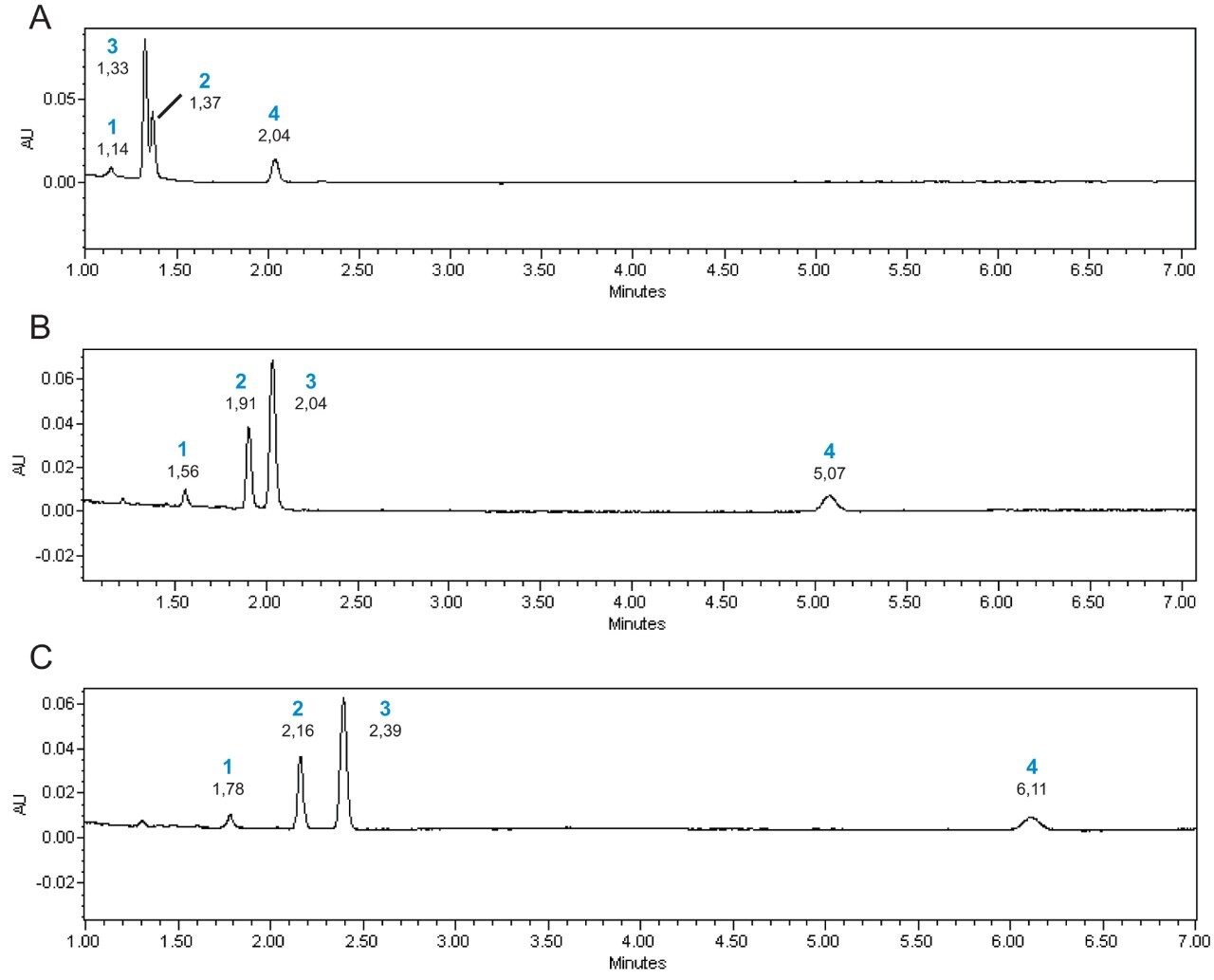
Design of experiments was employed to evaluate the influence of temperature, pressure and ethanol percentage and their interactions on the retention and separation factors of the compounds.16 Pressure and ethanol percentage were the main parameters that affected the retention factors. In the chromatogram shown in Figure 3A (2200 psi, 25 °C, 24% of ethanol), no separation between beta-carotene and coenzyme Q10 was observed. Changes in the operating conditions, decreasing the ethanol percentage to 15%, keeping constant temperature and pressure, provided a better resolution between beta-carotene and coenzyme Q10 (Figure 3B). In this chromatographic condition, a different peak elution order was observed. The decrease of pressure to 1500 psi (Figure 3C) improved the separation of beta-carotene and coenzyme Q10 even more, providing baseline resolution of the four compounds. Even though in this chromatographic condition the analysis time increased to more than 6 minutes, the separation was still performed in a reasonable time. It should be noticed that a gradient of elution could be used to decrease the retention time of lutein, however since this method is intended to be scaled-up, this approach was not used. Furthermore, enough time should be provided for the interferents to elute in real samples.
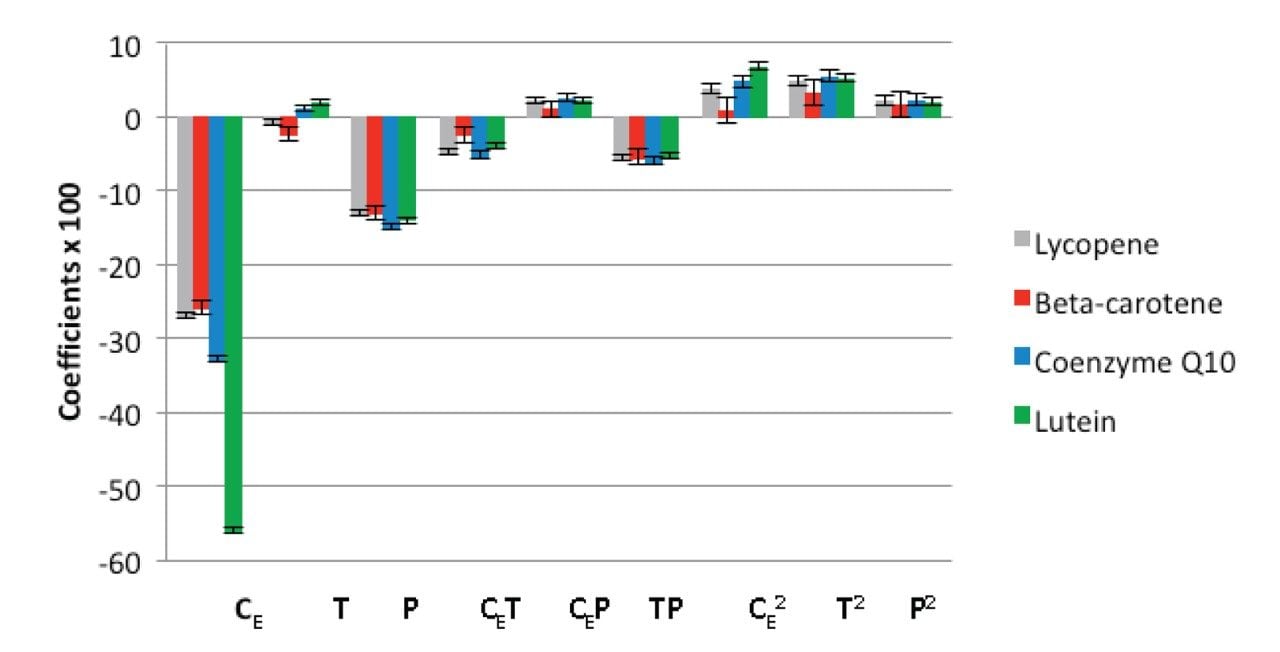
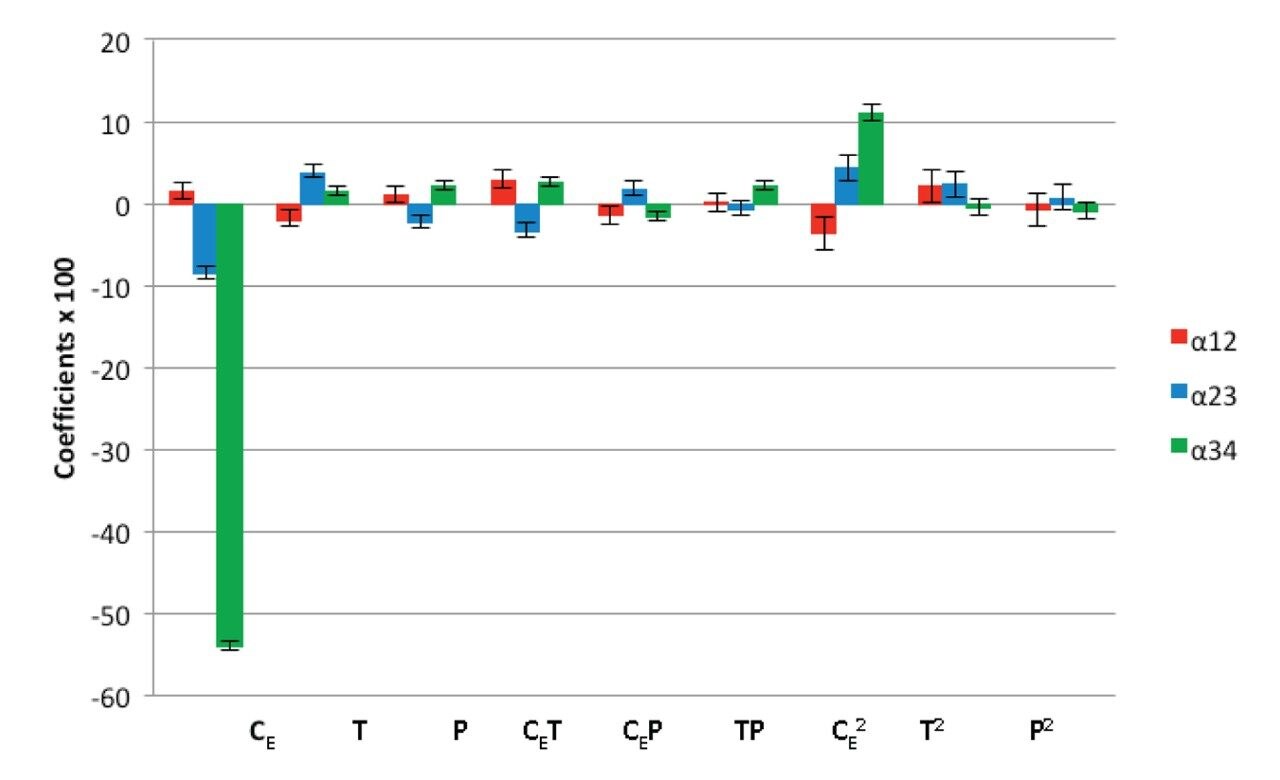
Data analysis showed that the retention strongly depends on the operational conditions within the studied range of the factors. The graphical representation of the coefficients for the regression models of the retention factors is presented in Figure 4. The ethanol percentage (CE) was the most significant effect for the retention of all analytes, with a negative effect, i.e. an increase in ethanol percentage in the mobile phase lead to a decrease of the retention factor. Pressure (P) was the second most significant factor studied and it had nearly the same influence on the retention factors of all compounds. Temperature (T) had a minor effect on the retention factors which might be attributed to the small temperature range employed in the experiments. At low pressure and low percentage of ethanol, the retention factors of all analytes increased continuously with increasing temperature. In contrast, the retention factors were unaffected by changes in temperature at higher ethanol percentage. At high pressure and high ethanol percentage, a small decrease in the retention factors were observed with an increase in temperature. It is interesting to notice that the coefficients of the models for lycopene and beta-carotene were negative whereas the coefficients for coenzyme Q10 and lutein models were positive. These results show that the effect of temperature on the retention factors is more complex than those observed for pressure and ethanol percentage.14, 17-18 Regarding the interaction effects, it can be seen that several of them are significant, which justifies the use of the multivariate approach, since the traditional univariate approach (the change of one factor at a time) would not be able to identify these interactions.
The separation factor (α) describes the separation of two components calculated by the ratio between their retention factors, and therefore, it represents the selectivity of the analysis.13 Beta-carotene and coenzyme Q10 were the analytes that presented separation issues inside the experimental domain and therefore this pair of peaks was considered critical in order to obtain adequate overall separation. The separation of lycopene and beta-carotene was not significantly affected by the experimental variables ethanol percentage and pressure, the only significant factor was temperature. This was due to the fact that the range of the separation factor for this pair was too small (1.2–1.3). For the other two pairs, the range of the separation factors was broader.
Figure 5 shows the coefficients for the regression models of the separation factors. In the case of the separation factor α12 (lycopene and beta-carotene), changes in ethanol percentage and pressure provided minor effects on selectivity. On the other hand, an increase in separation was achieved by lowering temperature. For the separation factor α23, which is related to the critical pair beta-carotene and coenzyme Q10, an increase in temperature provided a better separation of the two analytes. It is important to note that an increase in ethanol percentage resulted in a decrease of the separation factor. The results for the separation factor α34 (coenzyme Q10 and lutein) showed that an increase in separation follows a decrease in ethanol percentage while temperature presented a minor effect.
In order to find the optimal conditions for retention and separation of all analytes, a multiple response optimization procedure was carried out employing to restrictions: the retention factor (k) should be higher than 2 for all analytes (especially lycopene, which was the first eluted) and a separation criterion measured by α should be higher than 1.15 for the critical pair beta-carotene and coenzyme Q10. The experimental region that encompasses both criteria is shown in Figure 6, for 1500 psi (A) and 2200 psi (B).
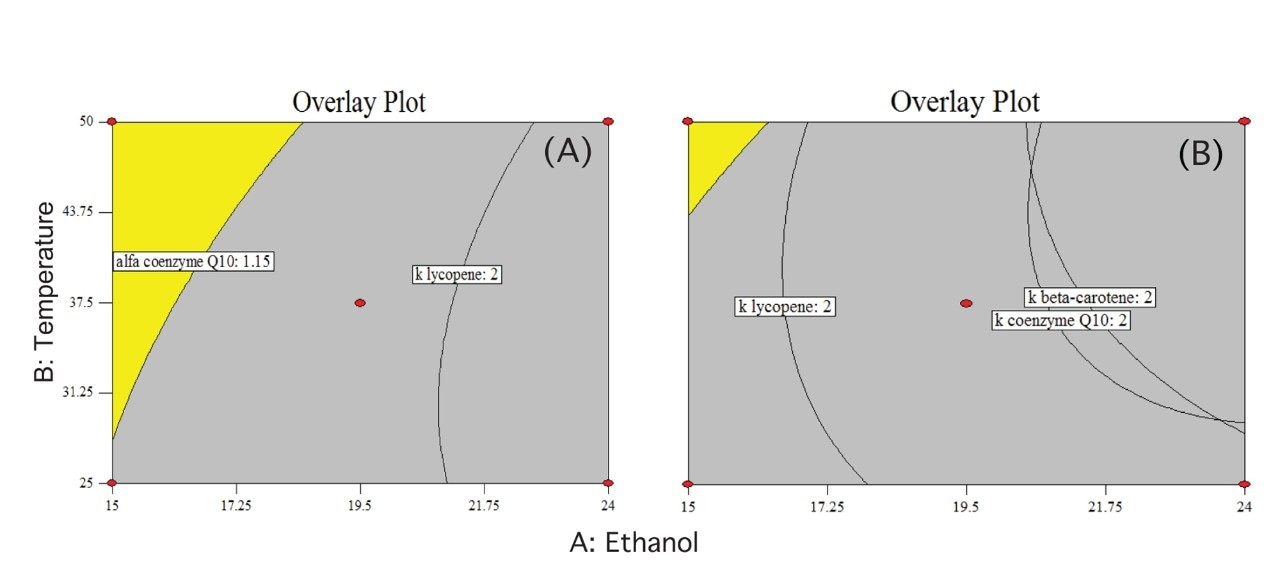
The selected operating conditions for further experiments were: 15.5% of ethanol at 40 °C and 1500 psi, which is located inside the experimental domain for both retention and separation factors (Figure 7).

UPC2 is suitable for the analysis of carotenoids and coenzyme Q10 due to the superior solubility of these non-polar compounds in the supercritical CO2 mobile phase. A fast separation of these bioactive compounds on an ACQUITY UPC2 HSS C18 SB Column was achieved in less than 6 minutes. Design of experiments was employed to investigate the individual and combined effects of ethanol percentage, temperature and pressure on the retention and separation factors. Retention factors were found to be strongly dependent on the amount of ethanol and pressure, followed by temperature whereas the three factors affected the separation. In this study, it was demonstrated that UPC2 enables a fast method development that can be used in routine analysis of carotenoids and coenzyme Q10.
720006559, June 2019