For research use only. Not for use in diagnostic procedures.
This application note demonstrates the use of novel ionKey/MS System, which utilizes the iKey Separation Device packed with 1.7 μm particles for fast and robust chromatographic separation. By integrating microscale LC components into a single platform design, the device avoids problems associated with capillary connections, including manual variability, leaks, and excessive dead volume. This integrated microfluidic device is suitable for lipidomics analyses with considerable advantages when compared to analytical scale LC/MS analysis.
The ionKey/MS System allows for fast and robust LC-MS lipidomics analyses with considerable reduction in solvent consumption and increase in sensitivity when compared to 2.1 mm I.D. chromatography. Potential applications include large-scale lipid profiling and low-abundance lipids analyses in biological materials.
Lipidomics is the comprehensive analysis of hundreds of lipid species in biological samples. Lipids play prominent roles in the physiological regulation of many key biological processes such as inflammation and neurotransmission. Alterations in lipid pathways have been associated with many diseases including cardiovascular diseases, obesity, and neurodegenerative disorders.
The ability to measure the wide array of lipid species in biological samples could help our understanding of their roles in health and disease. The need for a fast, comprehensive, and sensitive analysis of the hundreds of lipid species challenges both the chromatographic separation and mass spectrometry.
Here we used the novel ionKey/MS System, which utilizes the iKey Separation Device packed with 1.7 µm particles for fast and robust chromatographic separation. By integrating microscale LC components into a single platform design, the device avoids problems associated with capillary connections, including manual variability, leaks, and excessive dead volume. This integrated microfluidic device is suitable for lipidomics analyses with considerable advantages when compared to analytical scale LC-MS analysis.
|
LC system: |
ACQUITY UPLC M-Class System |
|
Sample loop: |
1 μL |
|
Separation device: |
iKey CSH C18 Separation Device, 1.7 μm, 150 μm x 100 mm (p/n 186007245) |
|
iKey temp.: |
55 °C |
|
Flow rate: |
2 μL/min |
|
Mobile phase A: |
Acetonitrile/water (60/40) with 10 mM ammonium formate + 0.1% formic acid |
|
Mobile phase B: |
Isopropanol/acetonitrile (90/10) with 10 mM ammonium formate + 0.1% formic acid |
|
Volume injected: |
0.2–0.5 μL |
|
Time (min) |
%A |
%B |
Curve |
|---|---|---|---|
|
Initial |
55.0 |
45.0 |
Initial |
|
1.00 |
40.0 |
60.0 |
6 |
|
10.00 |
1.0 |
99.0 |
6 |
|
16.00 |
1.0 |
99.0 |
6 |
|
16.01 |
55.0 |
45.0 |
6 |
|
18.00 |
55.0 |
45.0 |
6 |
|
Mass spectrometer: |
Xevo TQ-S |
|
Acquisition mode: |
MRM |
|
Ionization mode: |
ESI positive |
|
Capillary voltage: |
3.0 KV |
|
Source temp.: |
120 °C |
Lipid standards were purchased from Avanti Polar Lipids (Alabaster, AL) and Nu-Chek Prep (Elysian, MN). Total lipid extract from bovine brain was purchased from Avanti Polar Lipids. Mouse plasma (10 µL) was extracted with isopropanol (490 µL). The solution was then allowed to stand for 30 minutes in ice, vortexed, and then centrifuged (10,000 x g, at 4 °C for 10 min). The supernatant was collected in a new vial, evaporated to dryness under vacuum, and kept at -80 °C until further analysis. Immediately prior to analysis, all lipid extracts were re-suspended in isopropanol/acetonitrile/water (50/25/25, 250 µL).
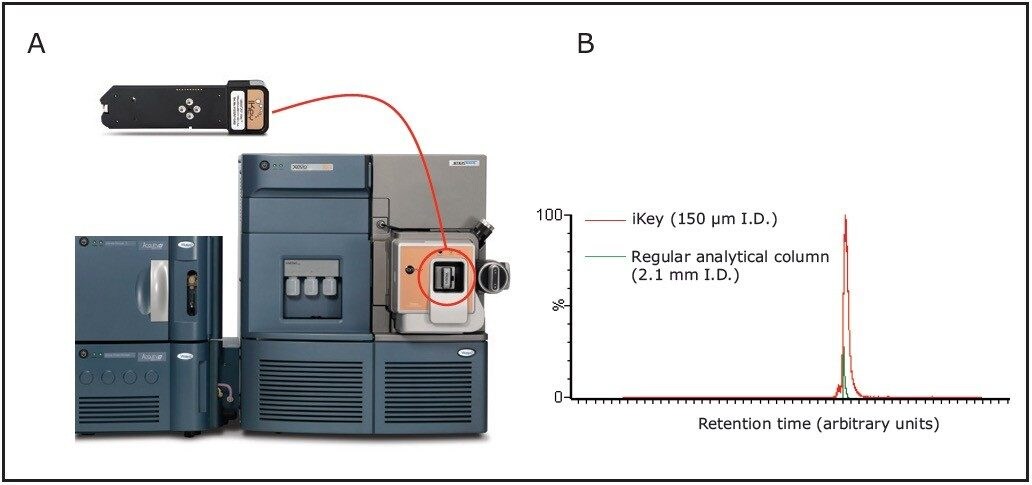
For the analysis of lipids, we used the ionKey/MS System, comprised of the Xevo TQ-S Mass Spectrometer, the ACQUITY UPLC M-Class System, the ionKey Source, and the iKey Separation Device. The iKey Separation Device contains the fluidic connections, electronics, ESI interface, heater, e-Cord, and the chemistry, permitting operation at high pressure with sub-2-micron particles, leading to highly efficient LC separations of lipid molecules. By integrating microscale LC components into a single system design, we avoided problems associated with capillary connections, including manual variability, leaks, and excessive dead volume. Lipidomics analyses were conducted using small volumes of lipid standards and lipid extracts from typical biological samples including plasma and brain tissues (0.2 µL). We separated lipids at flow rates of 2 µL/min using a ACQUITY UPLC M-Class engineered with 150 µm I.D. x 100 mm ceramic channel packed with CSH C18, 130 Å, 1.7 µm particles size (Fig. 1). The small column diameter (150 μm) of the iKey Separation Device allows low injection volumes (0.5 µL) and low flow rates (2 μL/min) increasing up to 10x the sensitivity compared to regular analytical columns (e.g. 2.1 mm I.D.) (Fig.1). Mobile phase consumption was reduced compared to 2.1 mm I.D. chromatography albeit maintaining comparable chromatographic resolution and analysis times (Fig. 2)1.
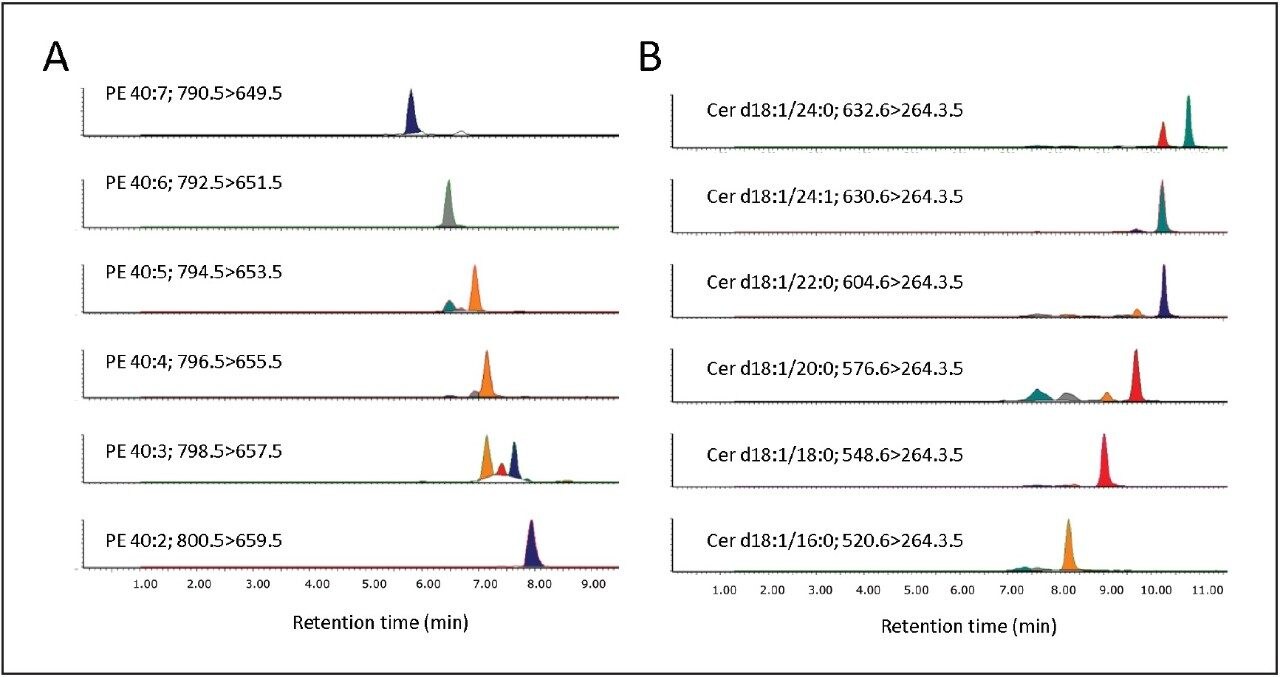
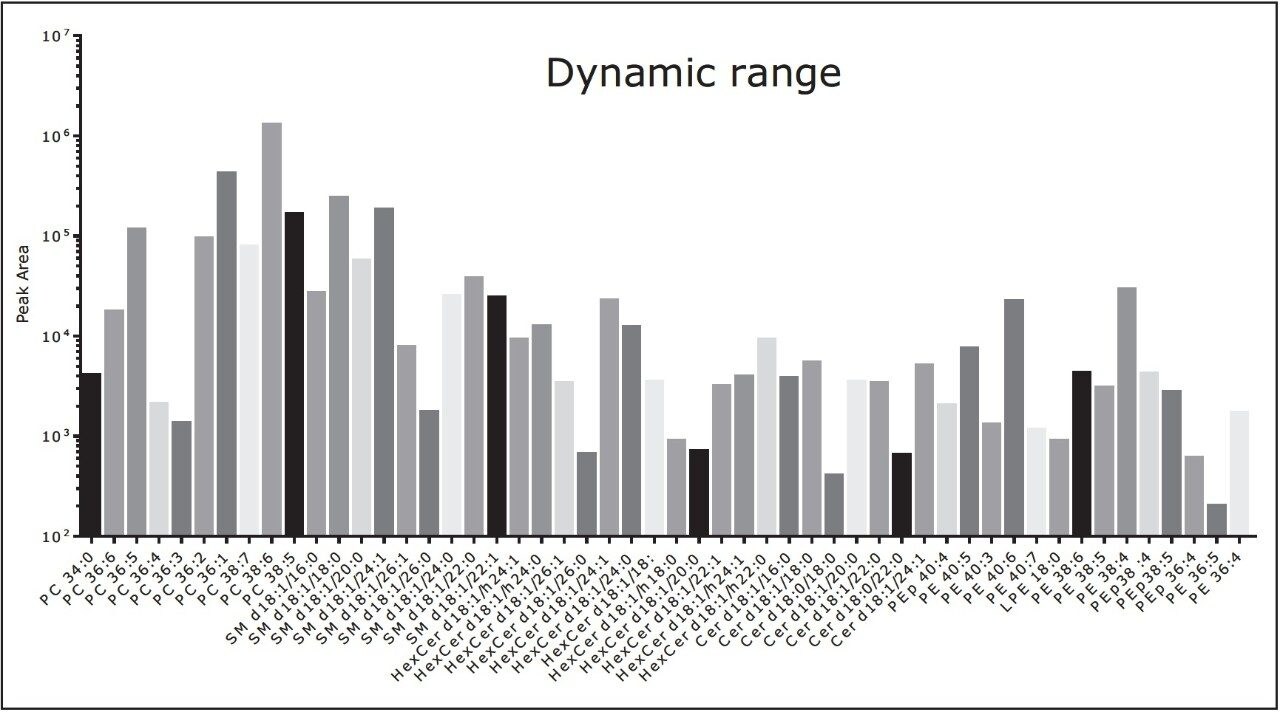
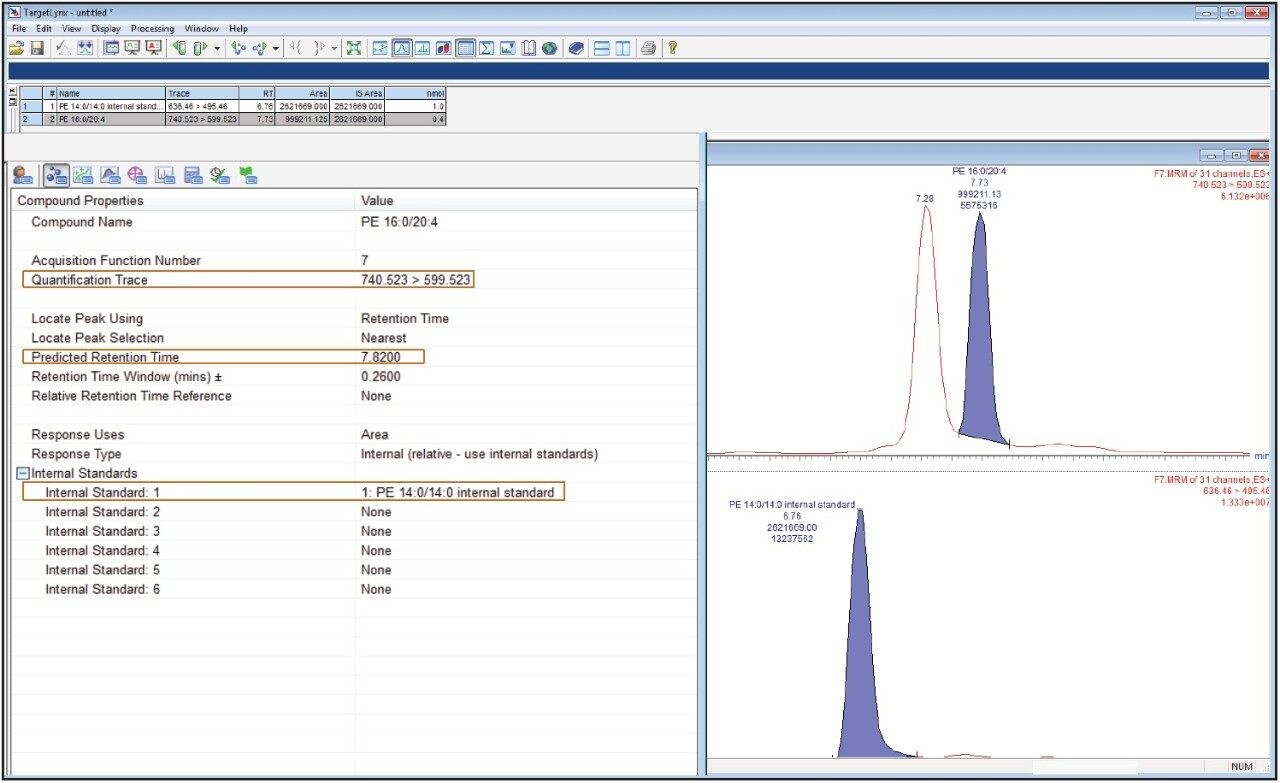
We conducted targeted lipidomic analyses using Xevo TQ-S in MRM mode and monitored 215 lipid species belonging to various lipid classes including phosphatidylethanolamines (PE), lyso PE, phosphatidylcholines (PC), lyso PC, ceramides (Cer), sphingomyelins, hexosylceramides, lactosylceramides, and cholesteryl esters (Table 1). Targeted lipids were measured over approximately five orders of dynamic range (Fig. 3 and 4). Lipids were separated according to acyl chain length and number of double bonds. Quantification was performed using TargetLynx Application Manager (Fig. 5). Initial reports in peer reviewed journals showed the advantages of using the ionKey/MS System in real world applications dealing with the analysis of low abundance lipids.2,3

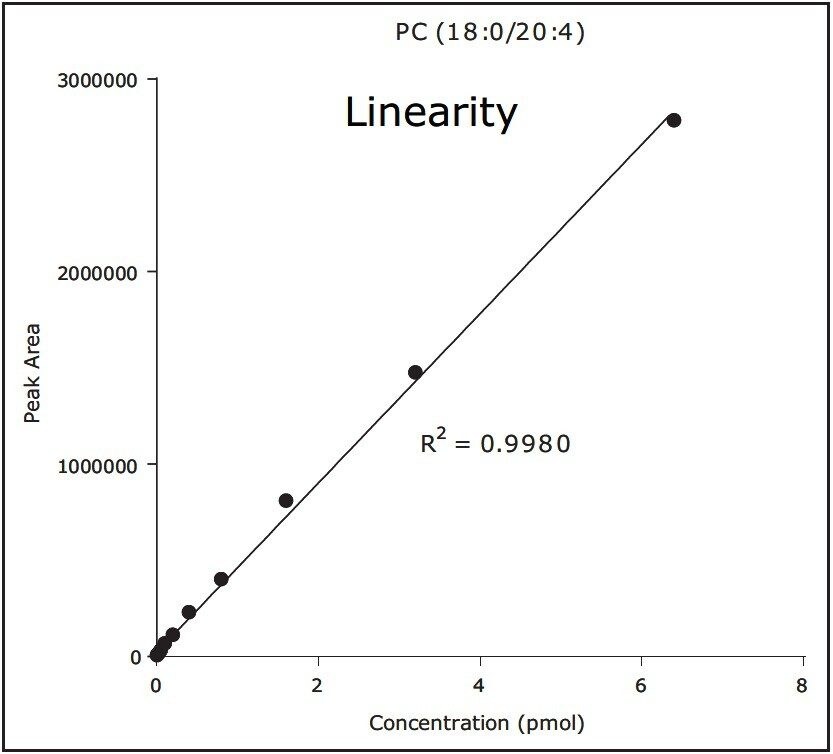
The ionKey/MS System is a novel microfluidics-MS platform that leads to highly efficient LC separation of lipids with comparable resolution to analytical scale LC-MS analysis.1 The use of the 150 µm iKey Separation Device enables the development of low flow MRM methods, bringing three major advantages over standard flow rate analysis1:
720004968, January 2016