
The combination of ionKey/MS, mixed-mode μElution SPE, and higher m/z b or y ion MS fragments provided the level of selectivity and sensitivity necessary to accurately quantify low pg/mL concentrations of glucagon in extracted plasma. Use of μElution format SPE eliminated the need for evaporation, reducing glucagon losses due to adsorption and non-specific binding. The 150 μm iKey enabled the development of a highly sensitive, low flow quantitative MRM method for glucagon with an LOD of 12.5 pg/mL and a dynamic range from 12.5–1,000 pg/mL. The current analysis uses 200 μL of plasma and provides a significant improvement in sensitivity and S:N over the analytical scale (2.1 mm I.D.) analysis using 1/5th the sample injection volume. Furthermore, an injection of the same volume (5 μL) of sample corresponded to a 10X increase in on-column sensitivity allowing for greater confidence in results, as compared to the traditional analytical method for this peptide. In addition, ionKey/MS reduces solvent and sample consumption, thereby reducing cost and allowing for multiple injections of samples for improved accuracy or to meet the guidelines for incurred sample reanalysis (ISR).
Glucagon for Injection (rDNA origin) is a polypeptide hormone identical to human glucagon and is used to treat severe hypoglycemia (low blood sugar).1 It is a single chain polypeptide that contains 29 amino acids residues with a molecular weight of 3483 (Figure 1). As a research tool, accurate quantification of glucagon from biological matrices can help us to better understand diabetes as a function of disease progression and/or drug treatment. Many assays, using different methodologies exist for glucagon analysis in biological samples.2-7 Glucagon, like other biologics, has historically been quantified using ligand binding assays (LBAs).2-5 With advances in MS and chromatography technologies over the past few years there has been a trend toward the analysis of large molecules by LC-MS/MS. This is, in part, driven by the fact that LBAs can suffer from significant cross-reactivity issues and lack of standardization. Additionally, LC-MS/MS also has the advantage of shorter development times, greater accuracy and precision, the ability to multiplex, and can readily distinguish between closely related analogues, metabolites or endogenous interferences. Large peptides, such as glucagon, are particularly difficult to analyze by LC-MS/MS as MS sensitivity is low due to poor transfer into the gas phase and poor fragmentation. In addition, glucagon suffers from significant non-specific binding, poor solubility, and must be properly stabilized in biological matrices during collection and sample preparation,6-8 making LC and sample preparation method development challenging.
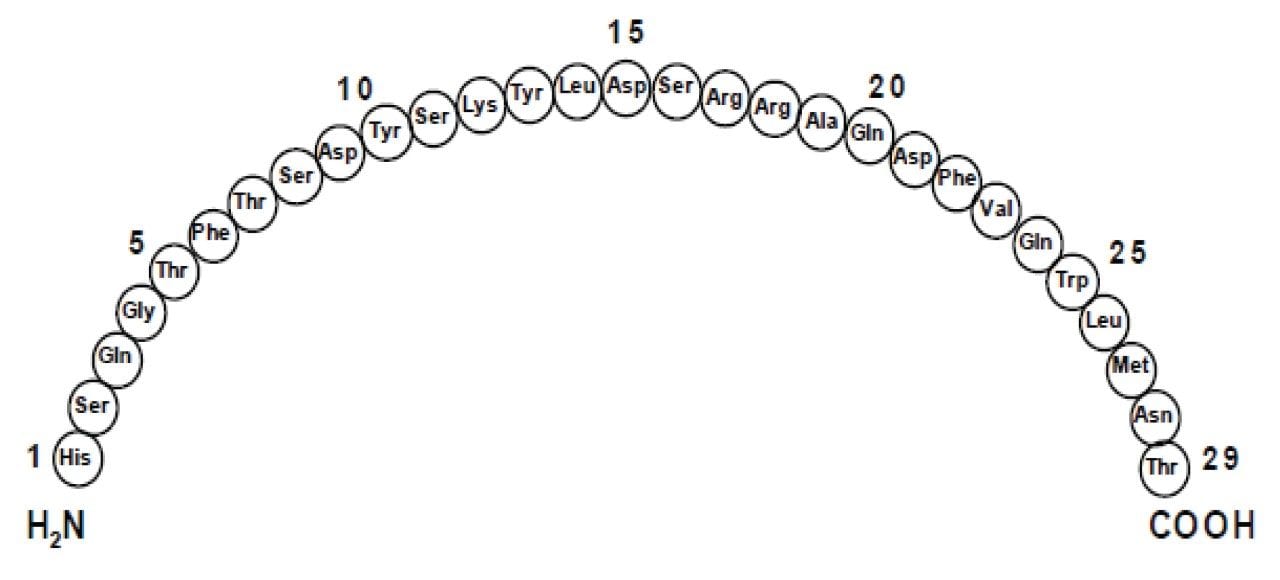
The pharmacokinetic profile of administered exogenous glucagon is characterized by a rapid absorption and elimination with a half-life of <20 minutes, resulting in a total duration of exposure to the peptide of ~2 hours. At the practical clinical dose, 0.25–2.0 ng/mL, maximum glucagon levels of ~8 ng/mL are reached in 20 minutes. Endogenous glucagon in plasma is present in low pg/mL levels (<100 pg/mL), which makes detection by LC-MS/MS even more difficult.
The work described herein uses a combination of selective μElution mixed-mode SPE sample preparation, optimal MS precursor and fragment choice, and the ionKey/MS System (Figure 2) for the highly selective and sensitive quantification of glucagon in human plasma. Detection limits of 12.5 pg/mL using only 200 μL of plasma were achieved with a linear dynamic range from 12.5 to 1,000 pg/mL. This work also capitalizes on the attributes of the ionKey/MS System enabling a 5X reduction in injection volume, a 10X increase in sensitivity, and 4X increase in signal-to-noise (S:N) compared to 2.1 mm I.D. analytical scale method.

Commercially available plasma was treated with a protease inhibitor cocktail (1:100). Plasma was then spiked with various concentrations of glucagon and mixed. These samples (200 μL) were acidified with acetic acid (0.5% final concentration) and vortexed, followed by dilution with 200 μL 5% ammonium hydroxide in water.

|
LC System: |
ACQUITY UPLC M-Class, configured for trap and back-flush elution |
|
Separation device: |
iKey Peptide BEH C18, 130Å, 1.7 μm, 150 μm x 100 mm (p/n 186006766) |
|
Trap column: |
ACQUITY UPLC M-Class Symmetry C18 Trap Column, 100Å, 5 μm, 300 μm x 50 mm (p/n 186007498) |
|
Mobile phase A: |
0.1% formic acid in water |
|
Mobile phase B: |
0.1% formic acid in acetonitrile |
|
Loading solvent: |
85:15 mobile phase A:B, 25 μL/min for first two minutes, reverse valve |
|
Valve position: |
Initial position one (forward loading of trap), switch to position two at two minutes (back flush elute of trap onto the analytical column) |
|
Analytical gradient: |
see Table 1 |
|
Elution flow rate: |
2.0 μL/min |
|
Column temp.: |
75 °C |
|
Sample temp.: |
15 °C |
|
Injection volume: |
5 μL |
|
Total run time: |
14.5 min |
|
Collection plates: |
Waters 1 mL Collection Plates |
|
Time(min) |
Profile |
Curve |
|
|
%A |
%B |
||
|
0 |
85 |
15 |
6 |
|
6 |
55 |
45 |
6 |
|
6.5 |
15 |
85 |
6 |
|
8.5 |
15 |
85 |
6 |
|
9.5 |
85 |
15 |
6 |
Table 1. LC gradient conditions.
|
MS system: |
Xevo TQ-S |
|
Ionization mode: |
ESI positive |
|
Capillary voltage: |
3.8 kV |
|
Source temp.: |
120 °C |
|
Cone gas flow: |
100 L/hr |
|
Collision cell pressure: |
5.5 x 10(-3) mbar |
|
Collision energy: |
Optimized by component, see Table 2 |
|
Cone voltage: |
Optimized by component, see Table 2 |
|
Chromatography software: |
MassLynx 4.1 |
|
Quantification software: |
TargetLynx |
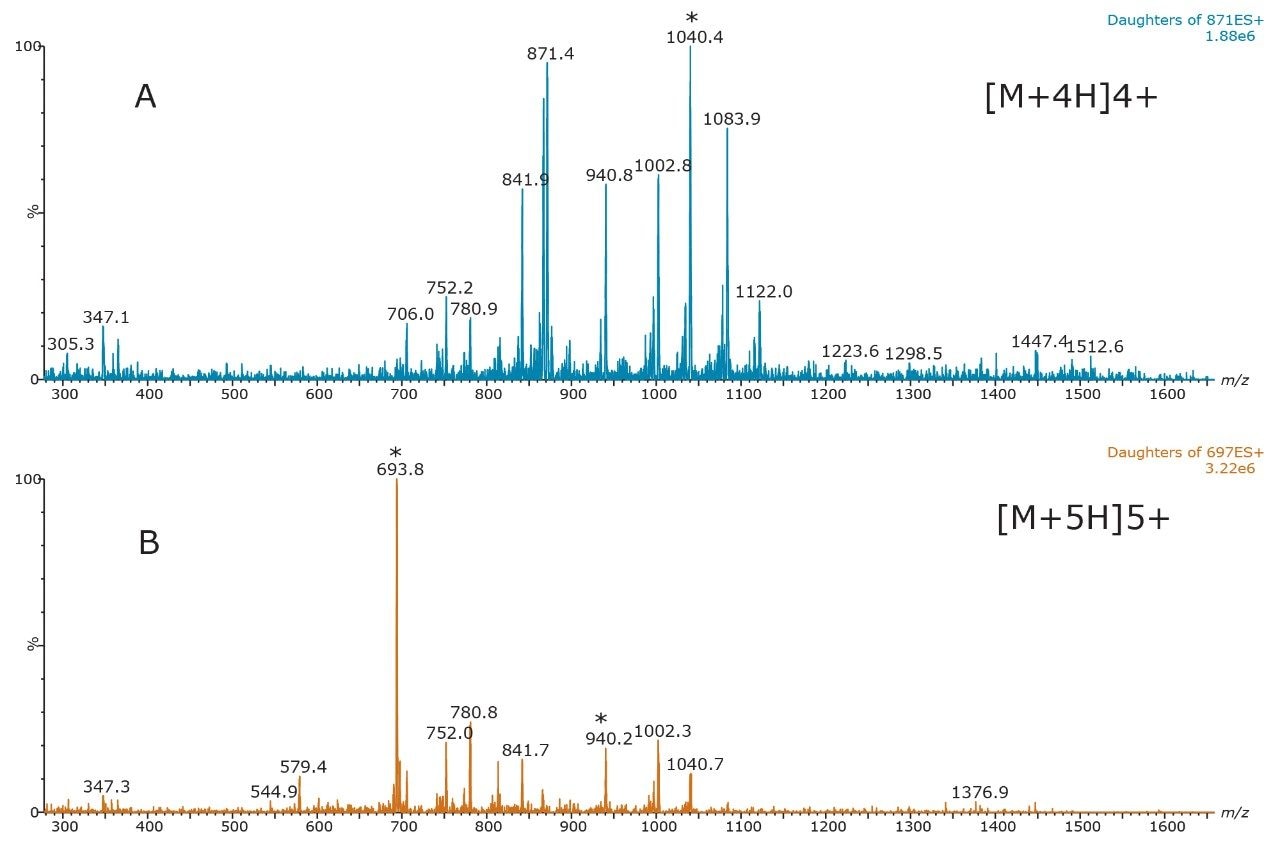
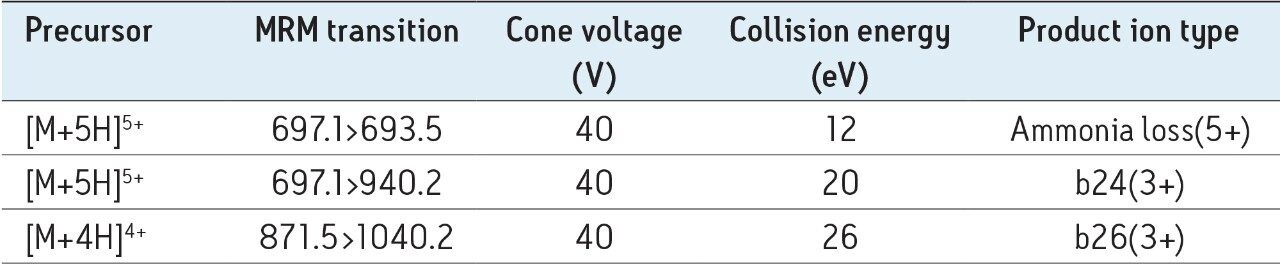
The 4+(m/z 871.5) and 5+(m/z 697.1) multiply charged precursors were observed for glucagon; MSMS spectra for these precursors, obtained at their optimal collision energies, are shown in Figure 3. The fragments at m/z 693.5 and 940.2 of the 5+ precursor, and 1040.2 of the 4+ precursor were chosen for quantification (Table 2). Although many peptides produce intense fragments below m/z 200, these ions (often immonium ions) result in high background in extracted samples due to their lack of specificity. In this assay, the use of highly specific b/y fragments yielded significantly improved specificity, facilitating the use of simpler LC and SPE methodologies.
Chromatographic separation of glucagon was achieved using the novel microfluidic chromatographic iKey Separation Device. The iKey Separation Device (Figure 4) is packed with UPLC-grade sub-2-µm particles that permits operation at high pressure and results in highly efficient LC separations. By integrating microscale LC components into a single platform design, problems associated with capillary connections, including manual variability, leaks, and excessive dead volume, are avoided. Use of the iKey Separation Device provided excellent peak shape, narrow peak widths (<4.0 secs at base), and resolution from endogenous matrix interferences.

Glucagon was eluted using a linear gradient from 15–45% B over 6 minutes. Representative chromatograms are shown in Figure 5. The use of multidimensional chromatography, specifically a trap and back-flush elution strategy, provided further sample cleanup and facilitated the loading of 5 μL of the high organic SPE eluate (required to maintain solubility of the peptides) without experiencing analyte breakthrough. Additionally, the ability to inject sample volumes typical for analytical scale LC analysis on the iKey Separation Device can provide the substantial gains in sensitivity that are often required to accurately and reliably detect low pg/mL levels of peptides and proteins in complex matrices.
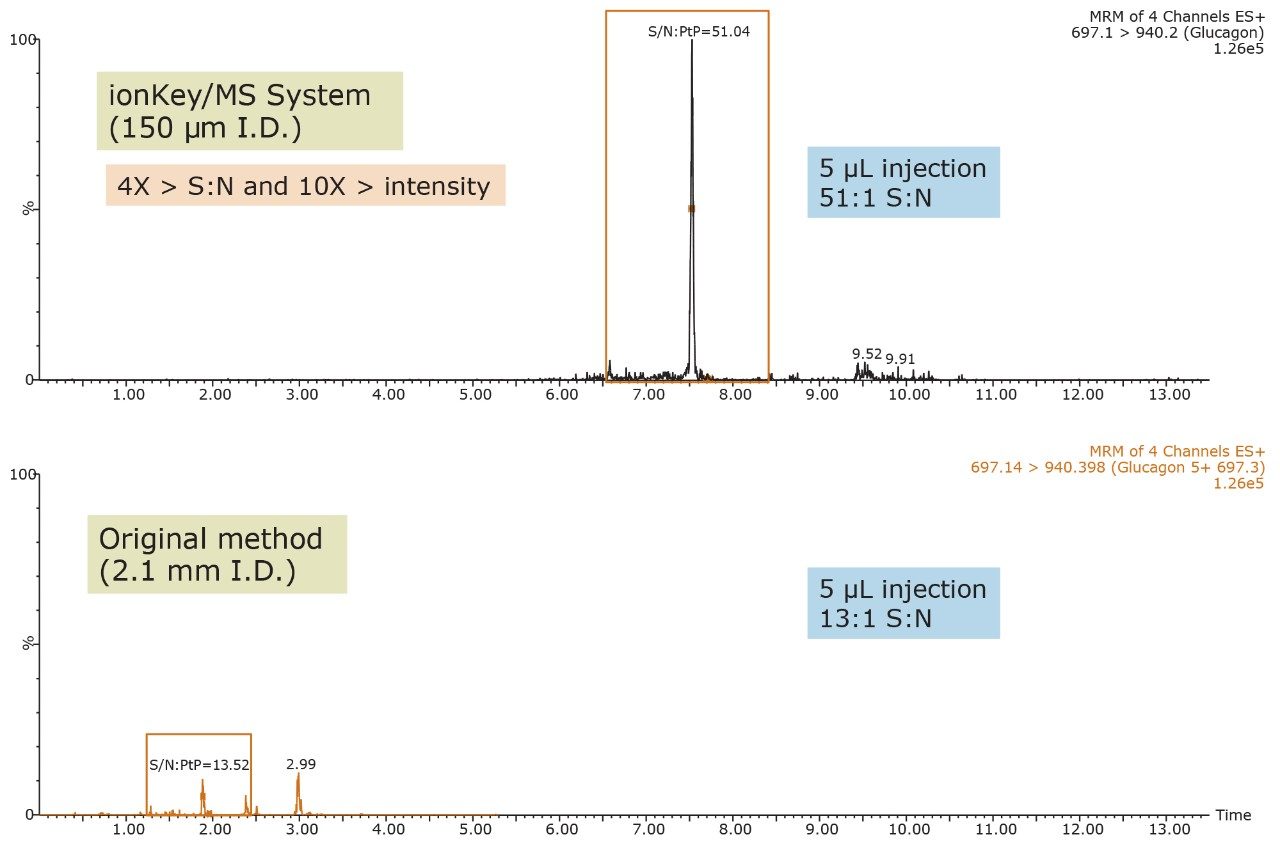
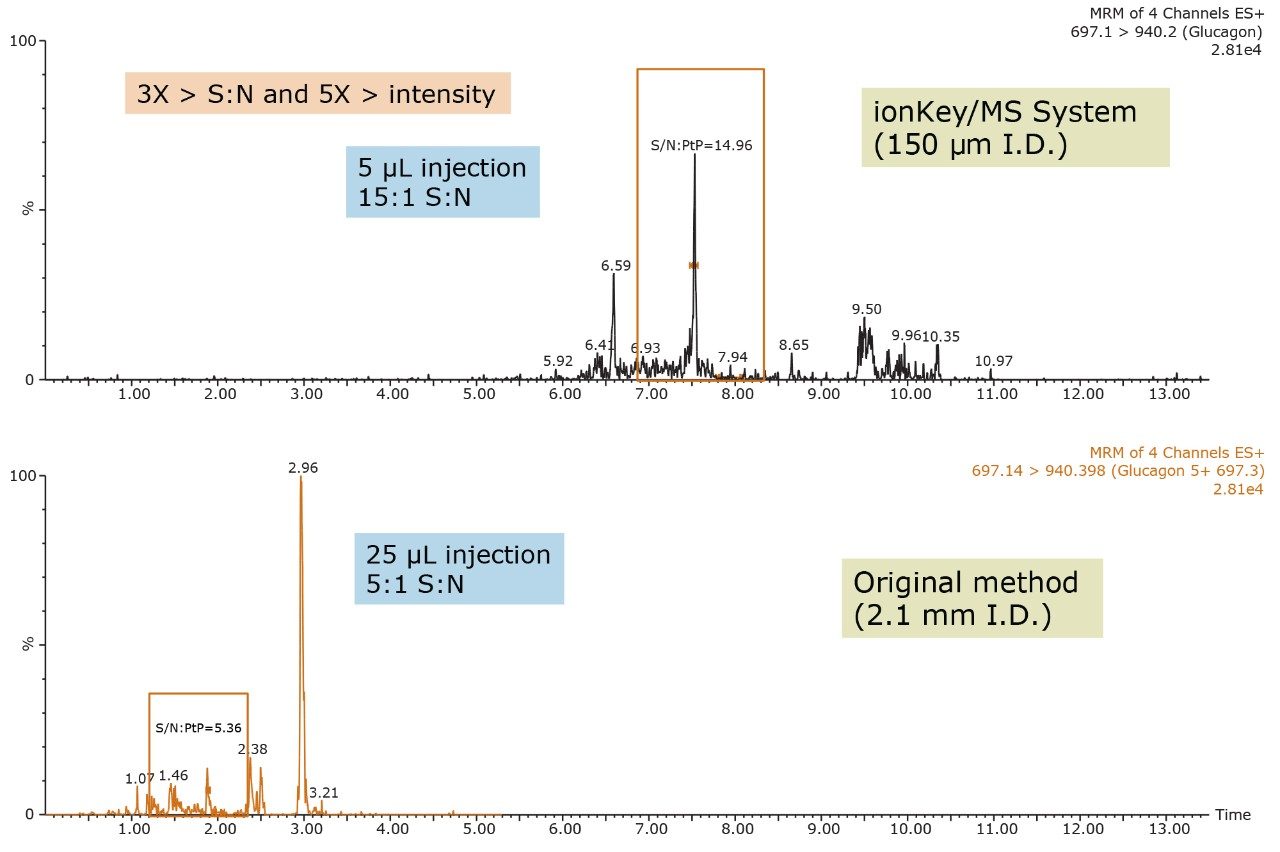
Versus analytical scale (2.1 mm I.D.), the ionKey/MS System generally offers increased sensitivity, making it ideal for glucagon analysis. This also facilitates the use of smaller sample volumes whilst maintaining or improving sensitivity. For a 250 pg/mL plasma extracted sample, the same injection volume (5 μL) on the ionKey/MS System yielded 4X greater S:N and a 10X improvement in sensitivity versus 2.1 mm scale (Figure 6). Using the 150 µm iKey Separation Device, low pg/mL levels of glucagon were detected in extracted plasma. Figure 7 demonstrates the improvements the ionKey/MS System provides for a 25 pg/mL extracted plasma sample. Versus 2.1 mm scale (25 μL injection), a 5 μL injection on the ionKey/MS System yields 5X greater sensitivity and a 3X improvement in S:N. Ultimately, the use of the ionKey/MS System enabled the development of a low flow quantitative MRM method for glucagon that achieved detection limits of 12.5 pg/mL from only 200 μL of plasma.
Development of this assay was challenging due to a high degree of non-specific binding (NSB) and difficulty maintaining peptide solubility throughout the SPE extraction and elution process. SPE was performed using Oasis MAX, a mixed-mode sorbent, to enhance selectivity. To ensure glucagon stability during sample preparation and extraction, human plasma was treated with protease inhibitor cocktail. Glucagon was then spiked at various concentrations into the plasma and mixed. These samples were then acidified with acetic acid (0.5% final concentration). Acidification helped disrupt protein binding. Plasma samples were then pre-treated with 5% NH4OH in water to adjust pH prior to SPE. The diluted plasma (pH >10) samples were then applied to conditioned SPE plates. Glucagon was well retained on the SPE sorbent during the load step, with no break-through occurring. At this basic pH, glucagon will carry a net negative charge, putting it in the proper charge state to bind to Oasis MAX (quaternary amine) by ion exchange. Optimization of the elution solution was critical to maximize recovery, maintain its solubility, and minimize interferences from the plasma matrix. The optimum elution solution was 65% organic, 25% water, with 10% acetic acid.
The enhanced selectivity of the Oasis MAX SPE extraction was imperative to accurately detect and quantify low pg/mL levels of glucagon in plasma. This is especially important where the use of the less specific 5+ precursor and ammonia loss fragment MRM transition might be necessary to achieve low limits of detection. During method development, use of reversed-phase (RP) only SPE was assessed. RP SPE yielded 10–15% greater recovery than the strong anion-exchange mixed-mode SPE (MAX). However, endogenous background was higher and yielded greater matrix effects (data not shown). In particular, matrix effects were >30% for the ammonia loss MRM transition when RP only extraction was employed. This greatly limited its use for robust quantification. Alternatively, the enhanced selectivity of the Oasis MAX SPE device greatly reduced matrix effects (<15%) and facilitated use of the less specific ammonia loss fragment for accurate quantification. In addition, the 96-well Oasis µElution Plate can be processed manually in under 30 minutes and is compatible with most liquid-handling robotic systems for automation to meet sample throughput requirements. This format also provides the ability to elute in very small sample volumes, minimizes the potential for adsorptive peptide losses and chemical instability, as well as concentrates the sample for increased sensitivity.
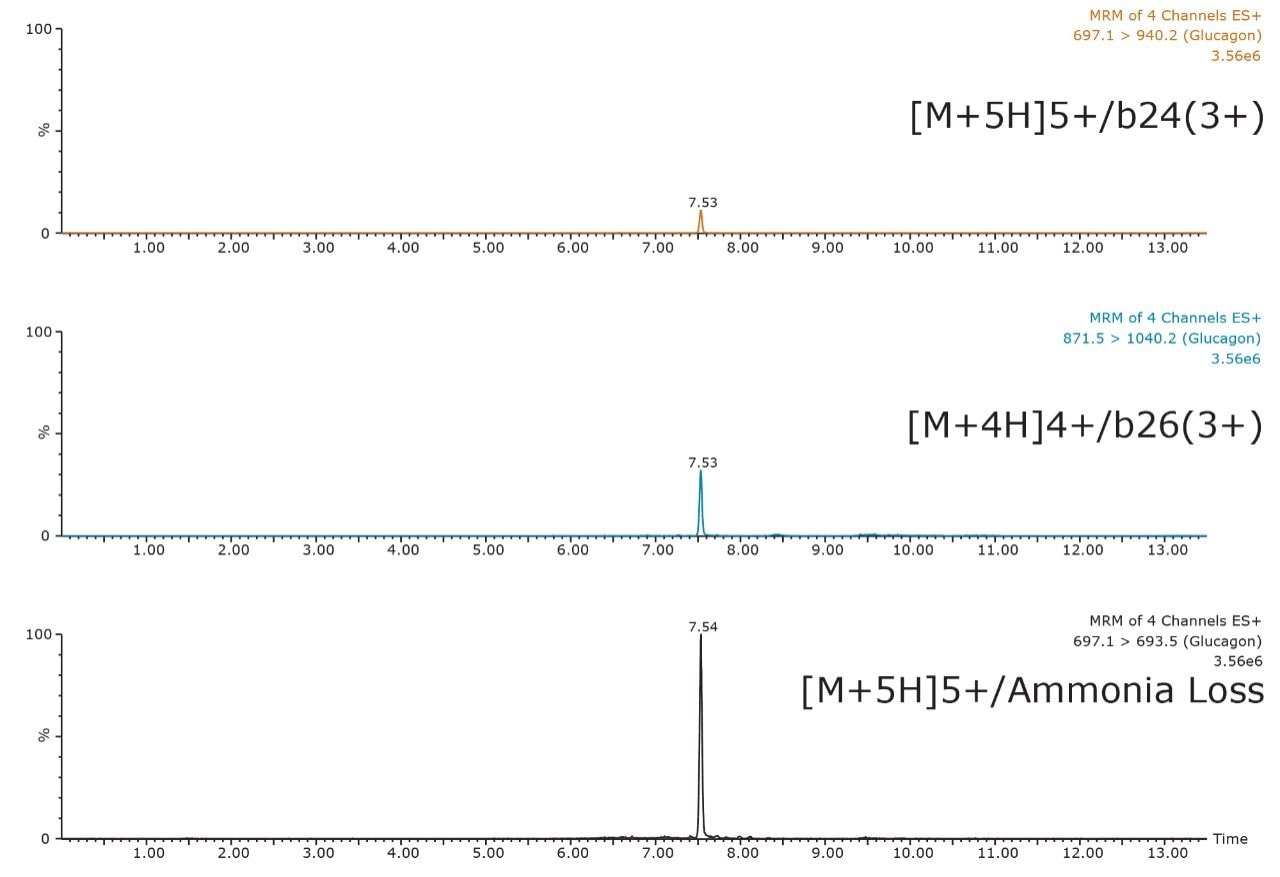
Triple quadrupole mass spectrometers, when operated in MRM mode, offer a unique combination of sensitivity, specificity, and dynamic range. However, in biological matrices, the task of achieving analyte specificity can be difficult, particularly with large peptides due to the high abundance of other endogenous proteins and peptides. Thus, the challenge of improving specificity lies not only in the sample preparation, and chromatography, but also selective choice of MRM transition for analysis. In the case of glucagon, the 697.1/693.5 MRM transition, corresponding to the 5+ precursor and ammonia loss fragment, resulted in a 10X greater signal than any of the other selective precursor/fragment transitions when tested in neat standard solution (data not shown) and in extracted plasma. However, in extracted plasma samples it was not as specific and resulted in higher background noise. Figure 8 shows a 500 pg/mL extracted plasma sample, and demonstrates the increased sensitivity and lack of specificity of the ammonia loss transition compared to the selective b-ion transitions.
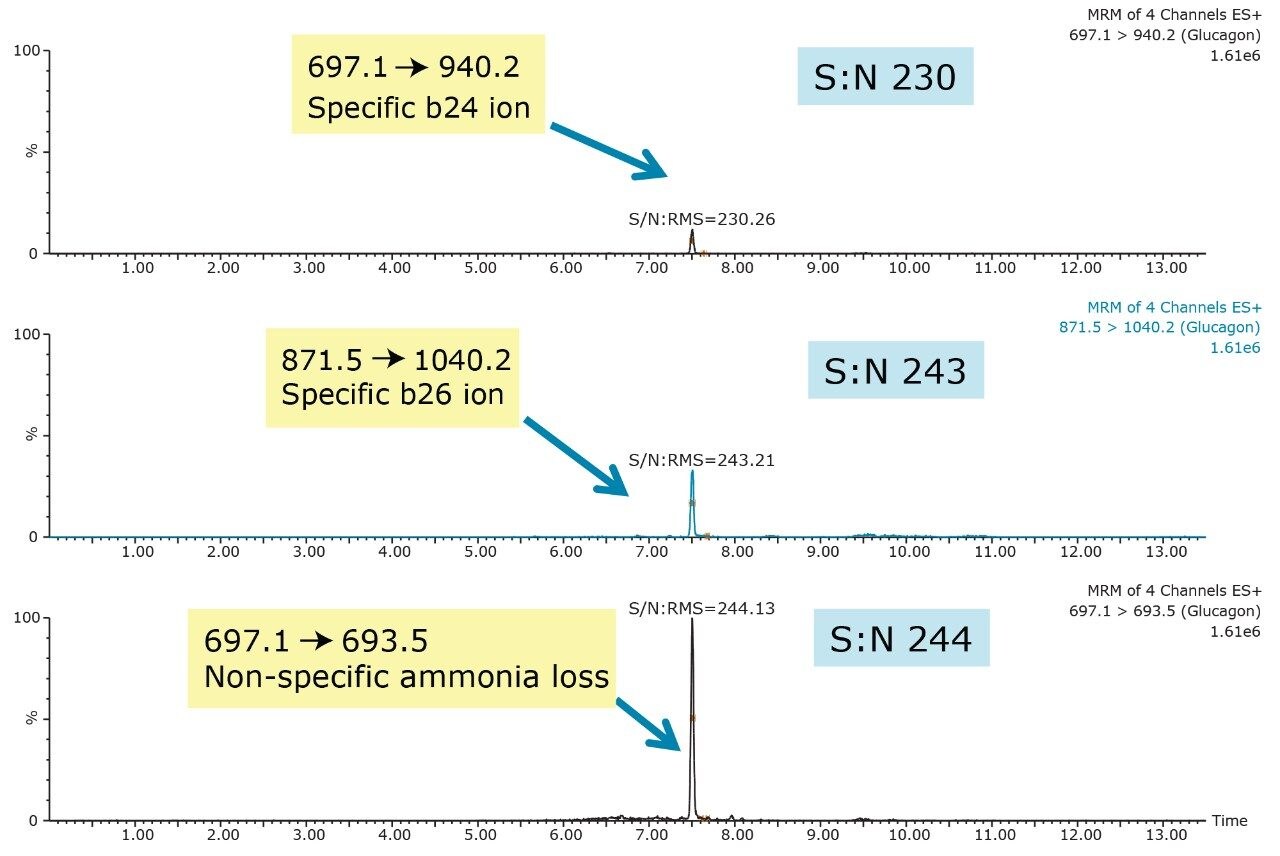
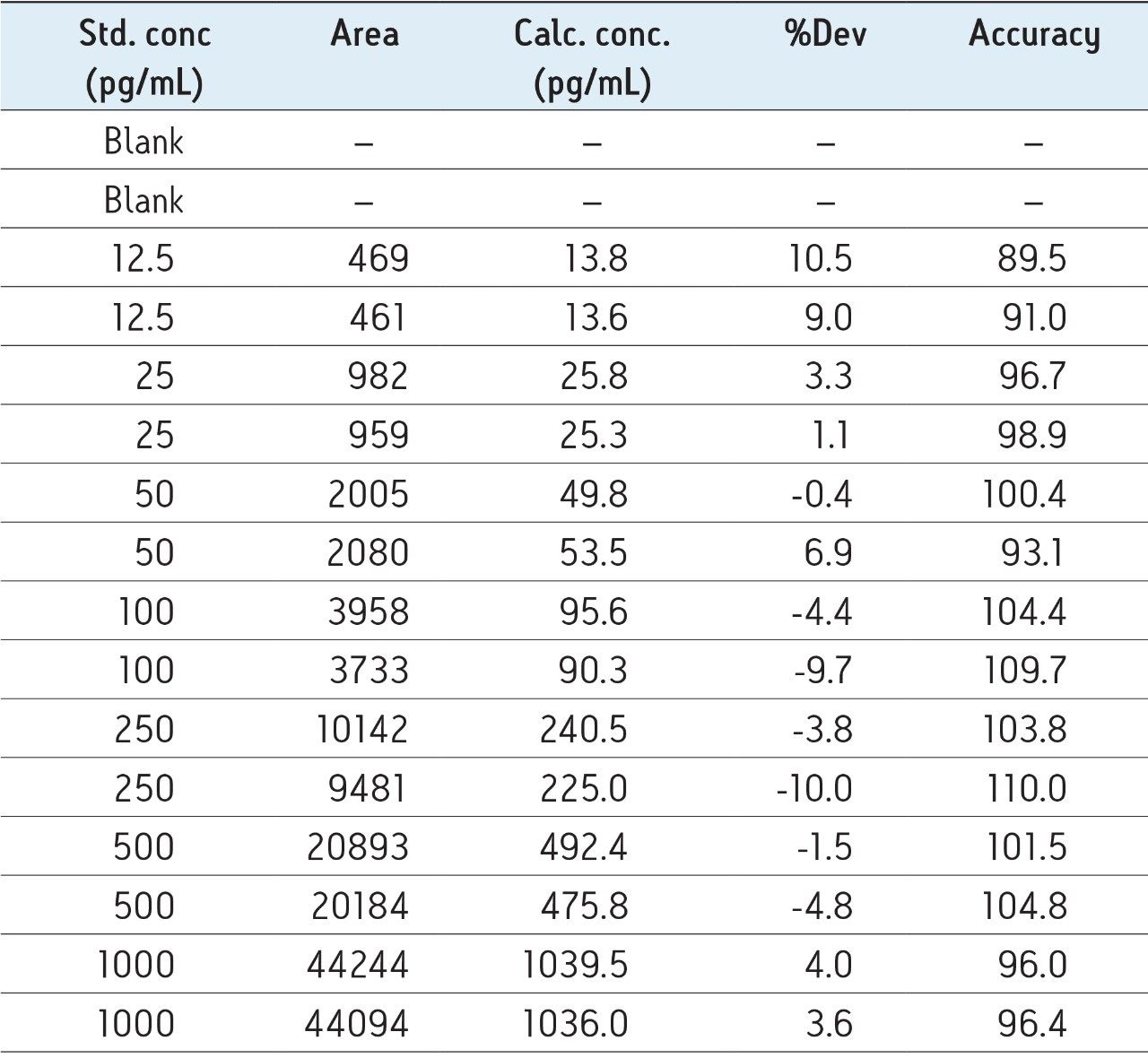
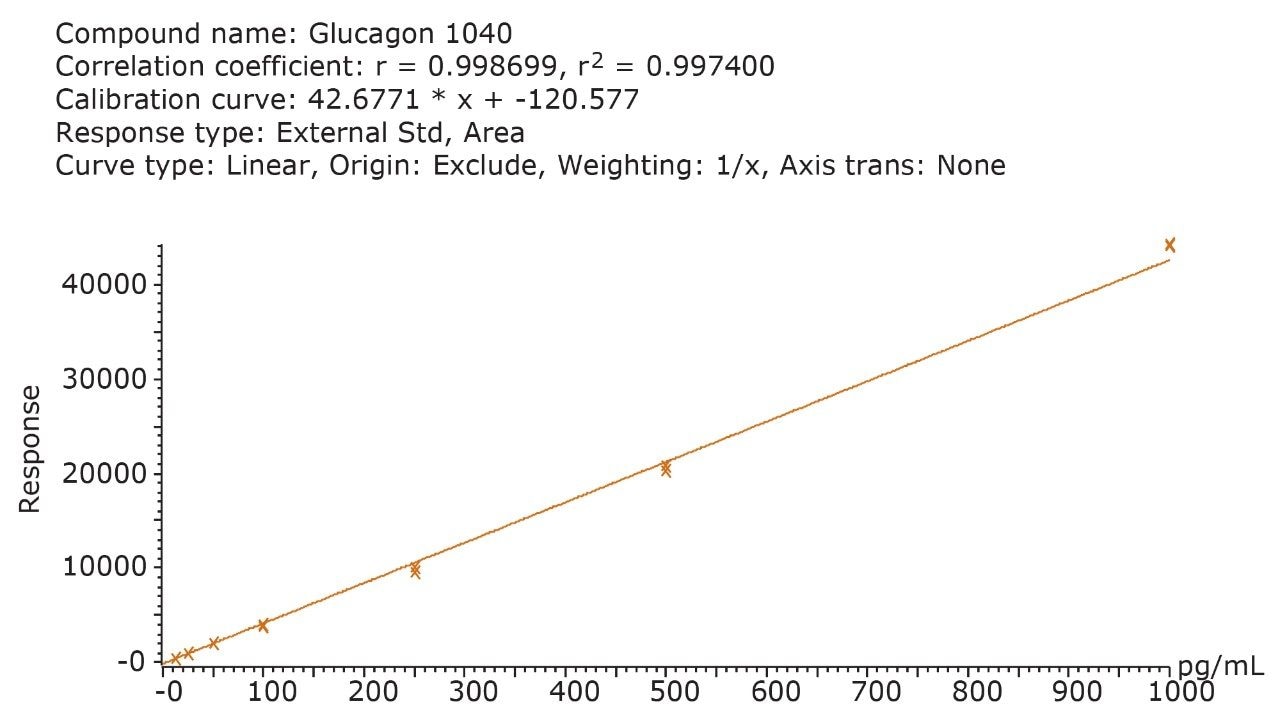
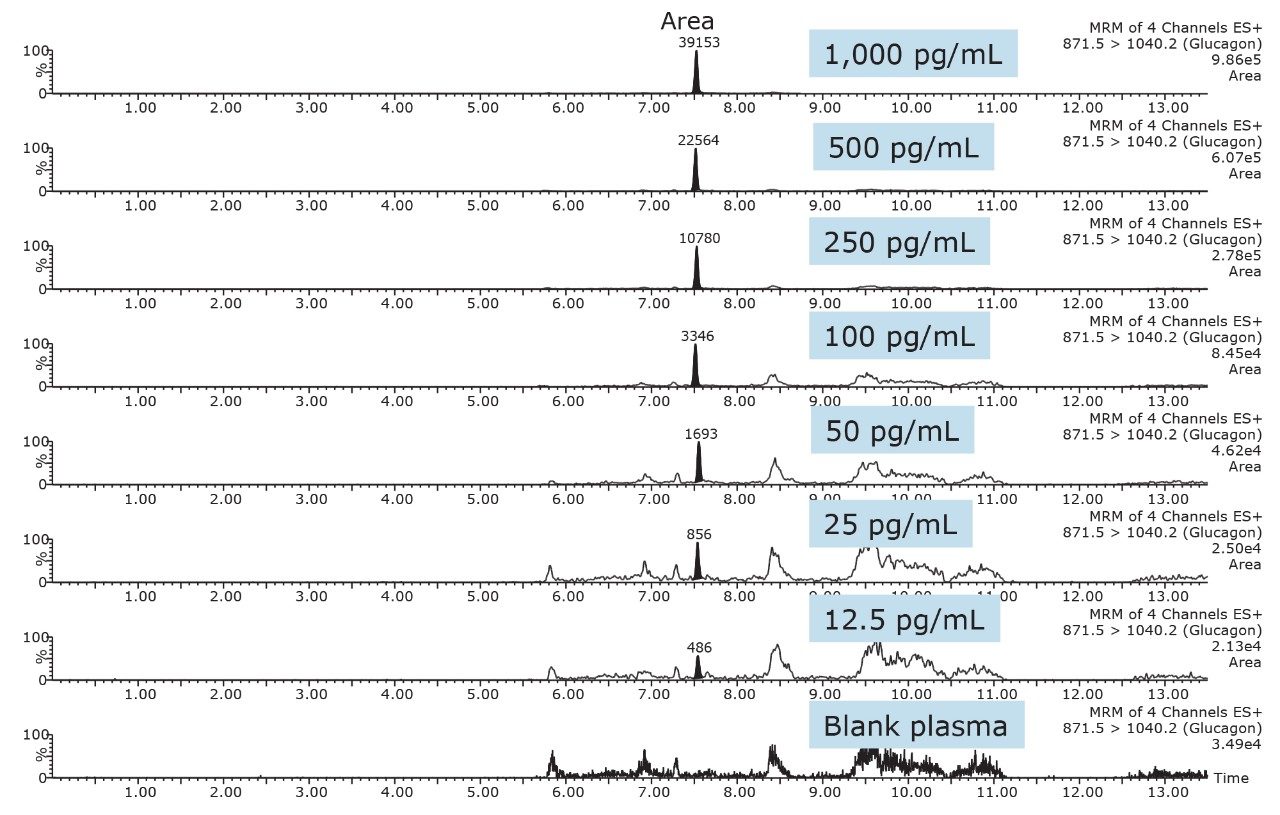
Although the intensity using the 697.1/ 693.5 MRM transition is 10X greater than the b-specific ion transitions (697.1>940.2 and 871.5 >1040.2), its sensitivity is mitigated by the accompanying higher background signal, as is demonstrated by the equivalent S:N ratios of all 3 MRM transitions. Additionally, measured matrix effects for the b-specific ion transitions were less than 10% in plasma, while the ammonia loss transition resulted in matrix effects between 10–15%. A summary of standard curve performance is shown in Table 3, and is illustrated in Figure 9. Using a 1/X regression, glucagon was linear from 12.5–1,000.0 pg/mL with R2 values of >0.99 for all 3 MRM transitions monitored. Representative chromatograms for extracted glucagon plasma standard samples are shown in Figure 10.
The combination of the ionKey/MS System, mixed-mode µElution SPE, and higher m/z b or y ion MS fragments provided the level of selectivity and sensitivity necessary to accurately quantify low pg/mL concentrations of glucagon in extracted plasma. Use of µElution format SPE eliminated the need for evaporation, reducing glucagon losses due to adsorption and non-specific binding. The 150 µm iKey Separation Device enabled the development of a highly sensitive, low flow quantitative MRM method for glucagon with an LOD of 12.5 pg/mL and a dynamic range from 12.5–1,000.0 pg/mL. The current analysis uses 200 µL of plasma and provides a significant improvement in sensitivity and S:N over the analytical scale (2.1 mm I.D.) analysis using 1/5th the sample injection volume. Furthermore, an injection of the same volume (5 µL) of sample corresponded to a 10X increase in on-column sensitivity allowing for greater confidence in results, as compared to the traditional analytical method for this peptide. In addition, the ionKey/MS System reduces solvent and sample consumption, thereby reducing cost and allowing for multiple injections of samples for improved accuracy or to meet the guidelines for incurred sample reanalysis (ISR).
720005073, January 2016