This application note will highlight the considerations that must be made when transferring an SEC method between UPLC and HPLC columns and systems.
Over the last several years Waters has produced the highest resolving and highest sample throughput size-exclusion chromatography (SEC) columns available for protein and peptide analysis.1,2 These size-exclusion UPLC (SE-UPLC) columns consist of 1.7 μm diameter ethylene bridged hybrid (BEH) particles with 200Å or 125Å pore sizes, or a 2.5 μm diameter BEH particle with a 450Å pore size. Based on observations, SE-UPLC technology has been adopted in many biopharmaceutical analytical laboratories as an important tool to monitor protein aggregation during product development due to the high sample throughput that it provides. However, often times SE-UPLC methods cannot be transferred to other laboratories due to the lack of available UPLC instrumentation. Therefore, in an effort to provide the capability to transfer SEC methods between UPLC and HPLC instruments, Waters has introduced 3.5 μm particle diameter BEH-based size-exclusion HPLC (SE-HPLC) columns specifically for use on traditional HPLC instrumentation. These columns provide the analyst with the unique capability to use consistent particle chemistries that can be directly scaled between HPLC and UPLC instrumentation. This application note will highlight the considerations that must be made when transferring an SEC method between UPLC and HPLC columns and systems.
All samples were diluted in mobile phase unless otherwise noted. Proteins were purchased as individual standards or as mixtures (Waters and Sigma-Aldrich). Sample concentrations were 1.0 mg/mL (nominal) unless noted otherwise.
|
LC system: |
Waters Alliance HPLC or ACQUITY UPLC H-Class Bio System with 30 cm Column Heater |
|
Columns: |
XBridge Protein BEH SEC, 200Å, 3.5 μm, 7.8 x 300 mm (p/n: 176003596); XBridge Protein BEH SEC, 450Å, 3.5 μm, 7.8 x 300 mm (p/n: 176003599); ACQUITY UPLC Protein BEH SEC, 200Å, 1.7 μm, 4.6 x 300 mm (p/n: 186005226); ACQUITY UPLC Protein BEH SEC, 450Å, 2.5 μm, 4.6 x 300 mm (p/n: 176002997) |
|
Column temp.: |
Ambient |
|
Sample temp.: |
10 °C |
|
Injection volume: |
10 μL |
|
Flow rate: |
0.84 mL/min |
|
Mobile phases: |
5 mM sodium phosphate, 150 mm sodium chloride, pH 7.2 (prepared using Auto•Blend Plus Technology or 5.5% A: 100 mM NaH2PO4, 14.5% B: 100 mM Na2HPO4, 15.0% C: NaCl, 65% D:H2O) |
|
Gradient: |
Isocratic |
|
Standard: |
BEH200 SEC Protein Standard Mix (p/n: 186006518); BEH450 SEC Protein Standard Mix (p/n: 186006842); Intact mAb Mass Check Standard (p/n: 186006552) |
|
Sample Vials: |
Deactivated Clear Glass 12 x 32 mm Screw Neck Total Recovery Vial, with Cap and pre-slit PTFE/Silicone Septa, 1 mL (p/n: 186000385DV) |
|
Detection: |
Alliance HPLC TUV Detector; ACQUITY UPLC TUV Detector with 5 mm Titanium flow cell |
|
Wavelength: |
280 or 214 nm |
|
Chromatography Software: |
Waters Empower Pro (v2 and v3) |
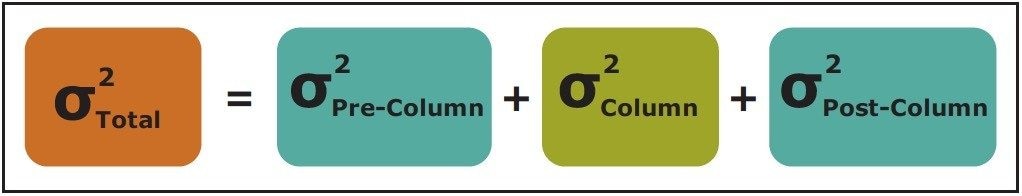
The performance of the LC system in an SEC separation can have a significant effect on the quality of the chromatographic resolution that can be achieved. Fundamentally, this can be represented by the equation highlighted in Figure 1, which has been adapted from Gritti and Guiochon.3 From this equation, the total peak variances s2 Total can be derived from the sum of the peak variances that occur prior to the analyte reaching the column (s2 Pre-Column), on the column (s2 Column), and after the analyte elutes off of the column (s2 Post-Column). Using this relationship, it can be readily derived that as the peak variance contributions from extra-column dispersion (s2 Pre-Column and s2 Post-Column) increase and become significant relative to the variance that occurs on the column (s2 Column), that the SEC separation efficiency will be measurably reduced. Another important concept illustrated by Figure 1 is the additive characteristic of both the pre-column and post-column variances in SEC. This is due to the absence of significant binding interactions between the analyte and the particle surface in SEC. Conversely, in other bind-and-elute based protein separations such as reversed-phase or ion-exchange chromatography, the contributions of s2 Pre-Column become far less significant due to the strong bindingdriven, analyte refocusing that occurs at the head of the column.
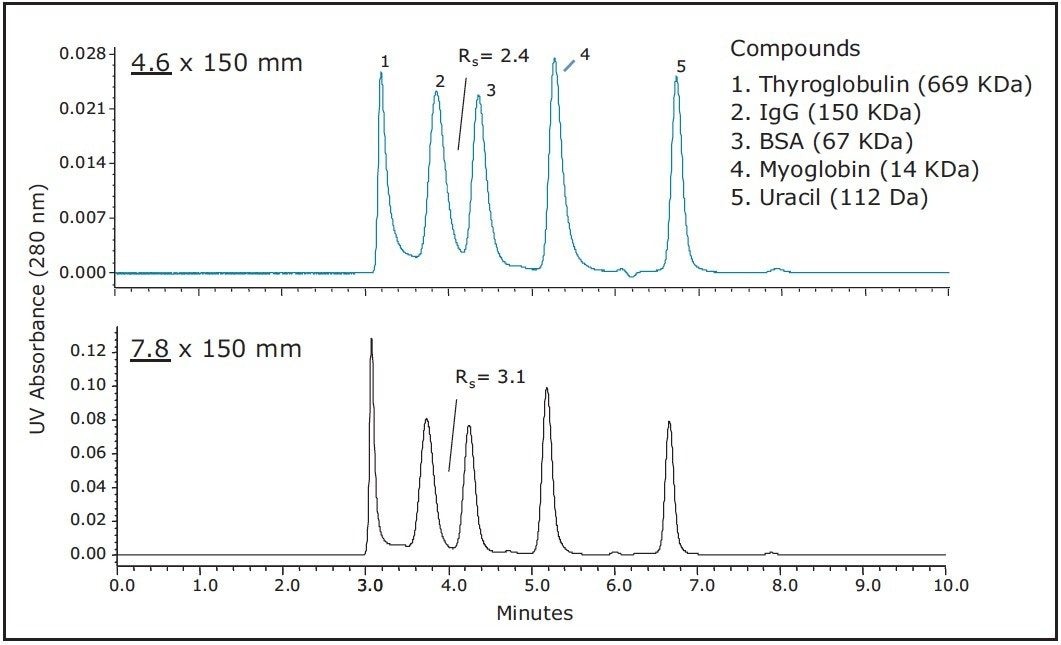
The practical effect of these considerations is demonstrated in Figure 2, which shows a comparison of the separation of protein standards on two columns packed with the same batch of 3.5 μm diameter, 200Å pore-size BEH particles on an Alliance HPLC System. One of the columns was an experimental column with an internal diameter (I.D.) of 4.6 mm, and the other the commercially available 7.8 mm I.D. column. The flow rates were adjusted to provide equivalent linear velocities for each column. It is readily observed that the resolution for the 4.6 mm I.D. column is significantly lower than that observed for the 7.8 mm I.D. column on the same instrument. As an example, the resolution observed between IgG and BSA is 30% higher for the 7.8 mm I.D. column in comparison to the 4.6 mm I.D. column. To understand why the performance of the 7.8 mm I.D. is markedly better, we can reconsider the relationships of the equation presented in Figure 1. In this equation, the peak dispersion that occurs within the column (s2 Column) increases with column I.D., however, this is offset by a proportional increase in pore volume ,which increases separation efficiency, with the end result being that the resolution of an SEC column is fundamentally independent of column I.D.. Therefore, as the I.D. of an SEC column increases, the separation efficiency remains constant , assuming packing efficiencies are maintained, while s2 Column increases. This results in the contribution that s2 Pre- Column and s2 Post-Column have to s2 Total becoming less significant, which practically results in an improved chromatographic separation.
There are two primary considerations to be made when transferring an SEC method from one column to another. Most importantly the surface chemistry of the particles must be comparable. In addition to matching pore size, size-exclusion particles can potentially have both ionic and hydrophobic interactions with biomolecules, and the nature of these interactions must be comparable if the separations are going to be performed using the same mobile phase and temperature. Secondly, the separation must be appropriately scaled with respect to particle size. The first step in scaling relative to particle size is to match as best as possible the ratio between length and particle diameter for the two columns using Equation 1:
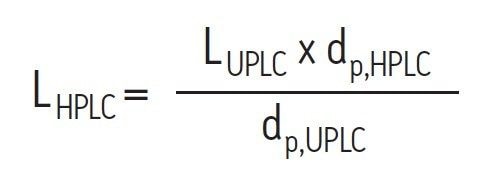
Where: LHPLC and LUPLC are the lengths of the HPLC and UPLC columns (mm), and dp,HPLC and dp,UPLC are the particle diameters of the HPLC and UPLC columns (μm).
Following this, a flow rate can be calculated by running both columns at the same reduced linear velocity, which is proportional to the product of the linear velocity and the particle diameter for a given analyte. Since the flow rate is proportional to the product of the linear velocity and square of the column I.D., the correct scaled flow rate can be readily calculated using Equation 2:
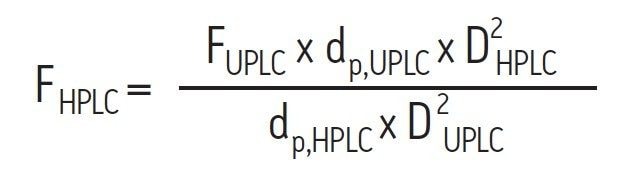
Where: FHPLC and FUPLC are the flow rates (mL/minute) of the HPLC and UPLC columns, and DHPLC and DUPLC are the internal diameters of the HPLC and UPLC columns (mm).
Finally, the injection volume can be scaled for column volume, which is proportional to the product of the square of the column and the column length. This can be represented by Equation 3:
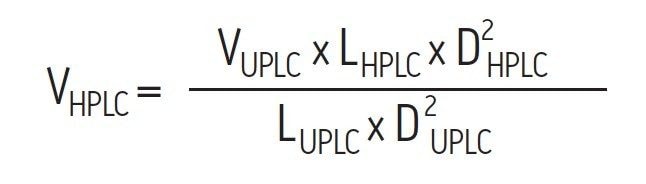
Where: VHPLC and VUPLC are the injection volumes of the HPLC and UPLC columns (μL).
We will first demonstrate the successful scaling between the ACQUITY UPLC Protein SEC 200Å, 1.7 μm, 4.6 X 300 mm column and the XBridge Protein SEC, 200Å, 3.5 μm, 7.8 mm I.D. HPLC column format. Given that there is an approximately 2-fold increase in particle size (based on Equation 1) the HPLC column length will need to be twice that of the UPLC Column (60 cm) to yield comparable resolution. This can be accomplished by connecting two 30 cm length HPLC columns in series. It should be noted that if a 15 cm, 1.7 μm particle size UPLC Column had been used, then the separation should be properly transferred to a single 30 cm length, 3.5 μm particle size HPLC column. Given the column I.D. and particle diameter values for each column, and using Equations 2 and 3, the flow rate should be 1.4 times greater and the injection volume should be 5.75 times greater for the HPLC analysis than for the UPLC analysis.
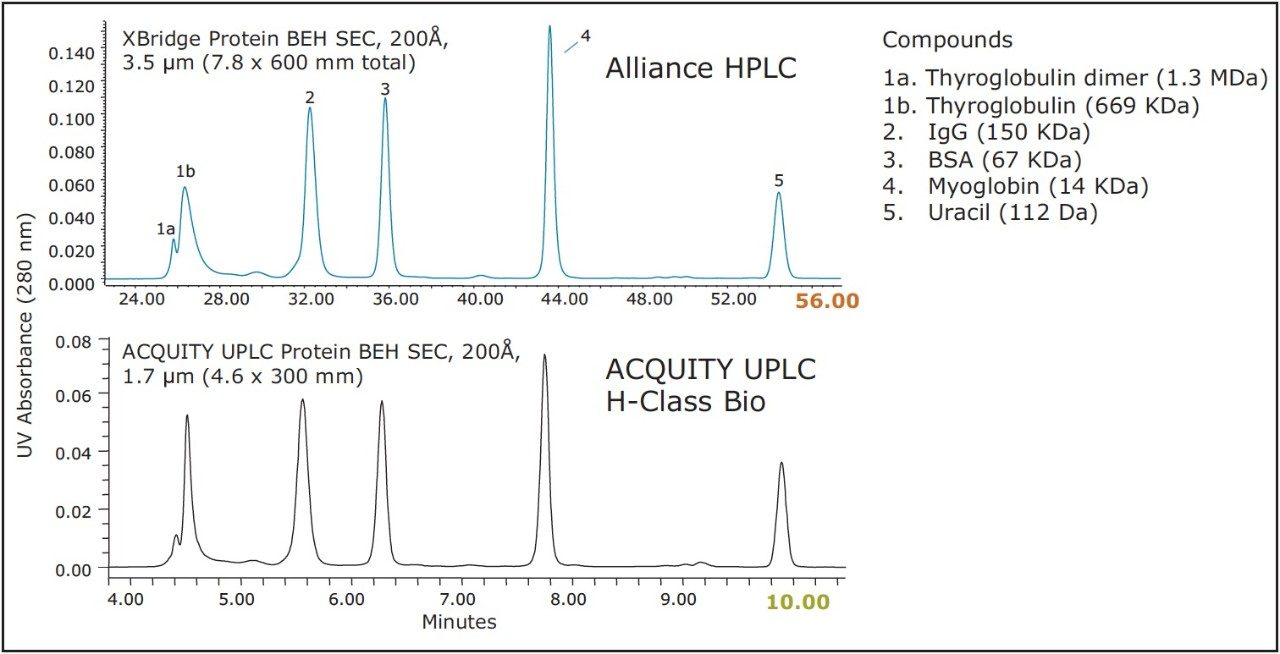
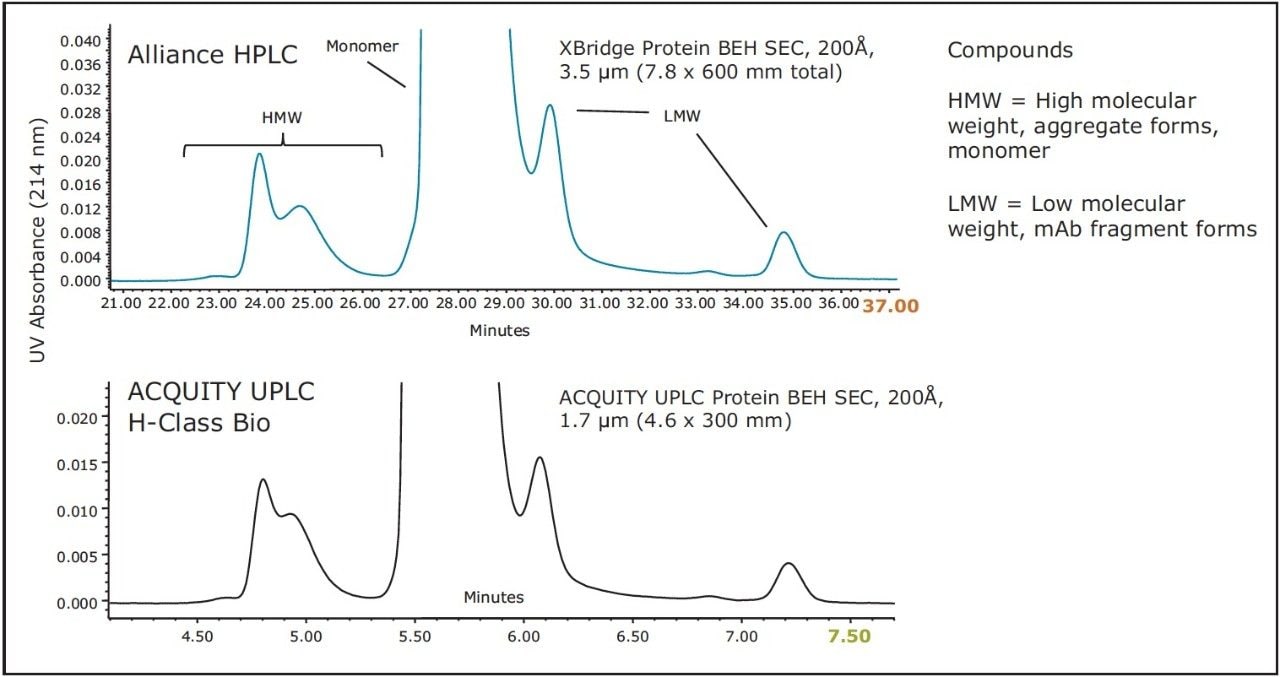
The results of this method scaling are shown in Figure 3 and Figure 4. Figure 3 is a comparison of the separation of the BEH200 SEC Protein Standard Mix, and Figure 4 is a comparison of the separation of the biotherapeutic monoclonal antibody infliximab. The UPLC separations were performed on an ACQUITY UPLC H-Class Bio System and the HPLC separations were performed on an Alliance HPLC System. The mobile phase used for both separations was a phosphate buffered saline (PBS) solution with low ionic strength and a slightly basic pH (25 mM sodium phosphate, 150 mm sodium chloride, pH 7.2). This buffer was selected due to its common use for SEC separations. Additionally, the pH and low ionic strength of this buffer will mask subtle variations in particle surface chemistry that can lead to undesired protein-particle interactions to a much lower extent as compared to higher ionic strength buffers.4 In both comparisons, the time axis has been normalized. Both pairs of chromatograms show comparable profiles with the primary difference being the analysis time, which is four to five times lower for the UPLC separation. While the HPLC analysis time is significantly longer, it has the same selectivity (profile) as the UPLC separation, thus allowing methods to be developed more rapidly and used on UPLC and then subsequently transferred to HPLC when the receiving lab does not have UPLC instrumentation.
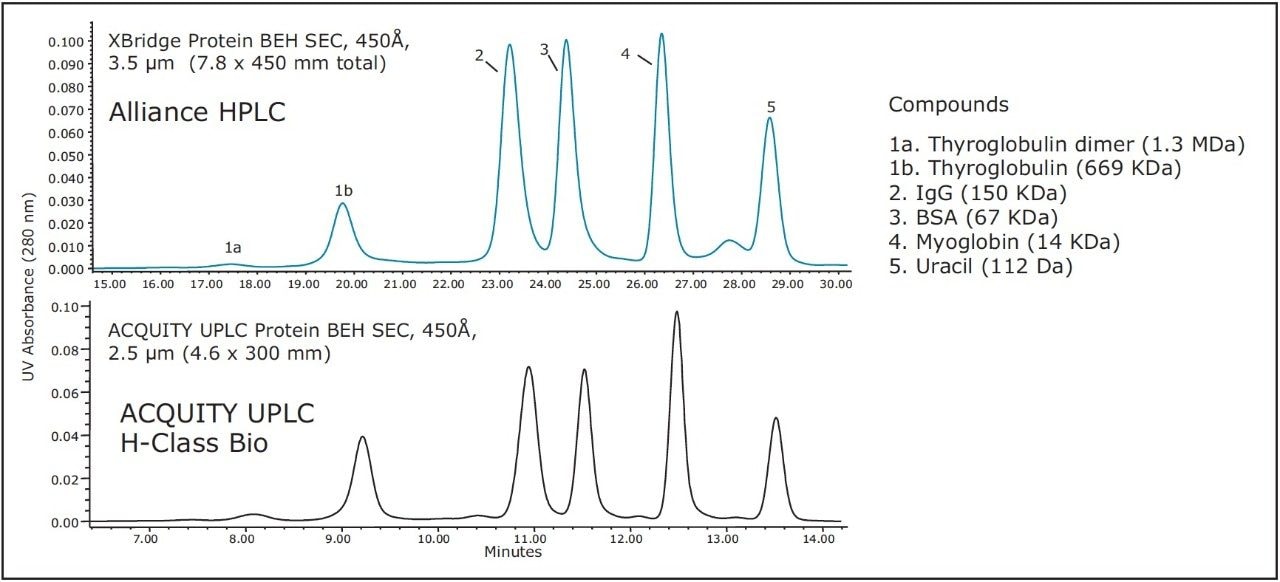
Next, we present the transfer between an ACQUITY UPLC Protein BEH SEC, 450Å, 2.5 μm, 4.6 x 300mm, column and the XBridge Protein BEH SEC, 450Å, 3.5 μm particle size, 7.8 mm I.D. HPLC column format. Using Equation 1, the length of the HPLC column should be 42 cm, which can be approximated by connecting a 30 cm and a 15 cm XBridge SEC, 3.5 μm Column in series. In the event that the method transfer is from a 15 cm length, 2.5 μm UPLC Column, the appropriate length HPLC column would be 21 cm, which is not an available configuration. In this case, a 30 cm HPLC column could be used. However, if matching reduced linear velocities were used, the resolution for the HPLC column should be approximately 20% greater, as the resolution difference is approximately proportional to the square root of compared column lengths.
Figure 5 and Figure 6 demonstrate the effectiveness of scaling from a 30 cm, 2.5 μm, 450Å, 4.6 mm I.D., SE-UPLC column to a total 45 cm length 3.5 μm particle size, 450Å, 7.8 mm I.D., HPLC column. Figure 5 is a comparison of the separation of the BEH450 SEC Protein Standard Mix, and Figure 6 shows the separations observed for IgM in its pentameric and dipentameric forms with molecular weights of approximately 900 KDa and 1.8 MDa, respectively. As in the previous example, the UPLC separations were performed on an ACQUITY UPLC H-Class Bio System and the HPLC separations were performed on an Alliance HPLC System, and in both comparisons, the time axis has been normalized. Both pairs of chromatograms show comparable profiles over the molecular weight range thereby demonstrating transferability between the UPLC and HPLC formats. In this example, UPLC is approximately 2-fold faster than HPLC.

Successful implementation of SE-UPLC separation technology requires high efficiency columns and low dispersion LC systems capable of operating at high back pressures. The advantage in doing so is the capability to dramatically improve sample throughput while decreasing sample requirements and mobile phase use. However, there are several situations where it is beneficial to be able to transfer between SE-UPLC and SE-HPLC separations, including instrumentation limitations in a method transfer, or in the event that a UPLC separation needs to be scaled up in order to facilitate the structural or functional characterization of low abundance species. However, in some cases, a direct method transfer using the same mobile phase and temperature conditions is not possible between SE-UPLC Columns packed with BEH-based particles and SE-HPLC columns packed with traditional silica-based particles due to the differences in the surface characteristics of the two particle types. These differences can necessitate the re-optimization of the method in order to get comparable results. Waters’ recent development of HPLC compatible 200Å and 450Å, 3.5 μm particles based on the same diol-coated BEH-particle chemistry as that used in ACQUITY UPLC Protein BEH SEC Columns provides the capability to readily transfer between SE-UPLC and SE-HPLC for the first time. This application note details and provides examples of the methodology that can successfully employed for this method transfer.
720005214, February 2015