This application note illustrates a simple massdirected approach to peptide isolation and discuss the options for improving the efficiency of the purification protocol. The AutoPurification System configured with an ACQUITY QDa Detector provides an alternative choice to UV-only systems, where mass detection improves isolation efficiency, and also helps in the identification of synthetic and cleavage by-products.
Peptides play a unique role in the development of new drug candidates and fill a specialized niche between traditional small molecule therapies and larger protein remedies. Because of their high specificity, stability, and safety, they are becoming more popular as a means for targeting many medical conditions.1,2 Whether peptides are made step-wise using solid-phase peptide synthesis,3 solution phase synthesis, or some combination of the two, almost all crude peptide mixtures are complex. While traditional peptide isolation is usually performed using UV-directed chromatography, mass-directed isolation makes the purification process easier with unambiguous discrimination between the target peptide and the contaminants formed during synthesis and cleavage. The AutoPurification System configured with an ACQUITY QDa Detector provides an alternative choice to UV-only systems, where mass detection not only improves isolation efficiency, but also helps in the identification of synthetic and cleavage by-products. This useful information suggests how the process can be improved and lead to greater target peptide yield. Here, we illustrate a simple massdirected approach to peptide isolation and discuss the options for improving the efficiency of the purification protocol.
|
Analytical columns: |
XBridge C18, 4.6 x 50 mm, 5 μm; SunFire C18, 4.6 x 50 mm, 5 μm; XSelect CSH Phenyl-Hexyl, 4.6 x 100 mm, 5 μm |
|
Analytical flow rate: |
1.46 mL/min |
|
Prep column: |
XSelect CSH Phenyl-Hexyl OBD Prep, 19 x 150 mm, 5 μm |
|
Prep flow rate: |
25 mL/min |
|
Mobile phase A: |
0.1% trifluoroacetic acid in water |
|
Mobile phase B: |
0.1% trifluoroacetic acid in acetonitrile |
|
Makeup solvents: |
50:50 or 90:10 water:acetonitrile with 0.1% propionic acid |
|
Cone voltage: |
10 V |
|
Probe temp.: |
500 °C, 600 °C |
|
Ionization mode: |
ES+, continuum |
|
Sampling frequency: |
2 Hz |
|
Scan range: |
100–1200 amu |
|
Wavelength: |
220, 280 nm |
|
Gradients and injection volumes: |
as noted in figures |
|
Sample: |
Crude synthetic peptide comprised of the following 16 amino acid residues: 7 polar, 6 nonpolar, 1 acidic, and 2 basic; purity 56% by HPLC |

Waters AutoPurification System: 2545 Binary Gradient Module, 2767 Sample Manager, System Fluidics Organizer, 8–30 mL Flow Splitter, two 515 HPLC pumps, 2998 Photodiode Array Detector, ACQUITY QDa Detector

Almost all crude peptides are challenging to isolate and purify due to the inherent complexity involved in synthesizing, deprotecting, and cleaving them from the resins used in solid phase peptide synthesis. Incomplete couplings, amino acid deletion sequences, incomplete cleavage impurities and adduct formations are among the myriad of by-products that complicate the isolation of the target peptide from the crude sample, yet the majority of them must be removed before the peptide candidate can be used in subsequent research. While UV-directed peptide purification is quite common, mass-directed purification removes the ambiguity associated with identifying the product of interest. Furthermore, developing a separation that encompasses both mass and UV detection ensures that more components will be revealed. Components that do not ionize will be detected by UV and, conversely, those that do not have a chromophore will be detected by mass. In addition, selecting an appropriate column and focusing the gradient4 improve separations quickly and easily, and coupled with mass detection, make tracking the product straightforward.
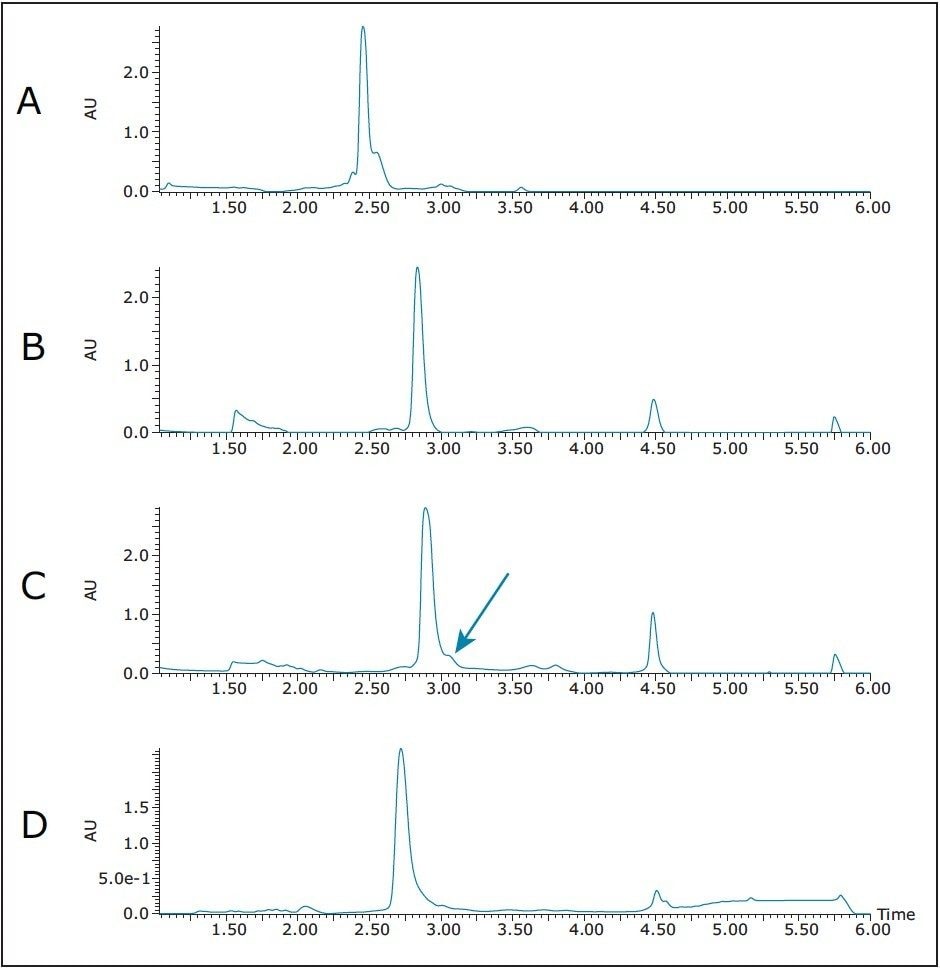
For these studies, the crude synthetic peptide was dissolved in dimethylsulfoxide (DMSO) and filtered with a 13 mm Acrodisc GHP syringe filter. As shown in Figure 2A, the peptide product eluted at about 2.5 minutes on a short SunFire Column, but the fast screening gradient also revealed a coeluting impurity. Focusing the gradient (Figure 2B) appeared to move the coeluting impurity away from the product, but with a larger injection (Figure 2C) a small shoulder reappeared on the backside of the target product peak. Changing to a short XBridge C18 Column (Figure 2D) did not resolve the contaminant, but seemed to make it merge back under the tailing product peak.
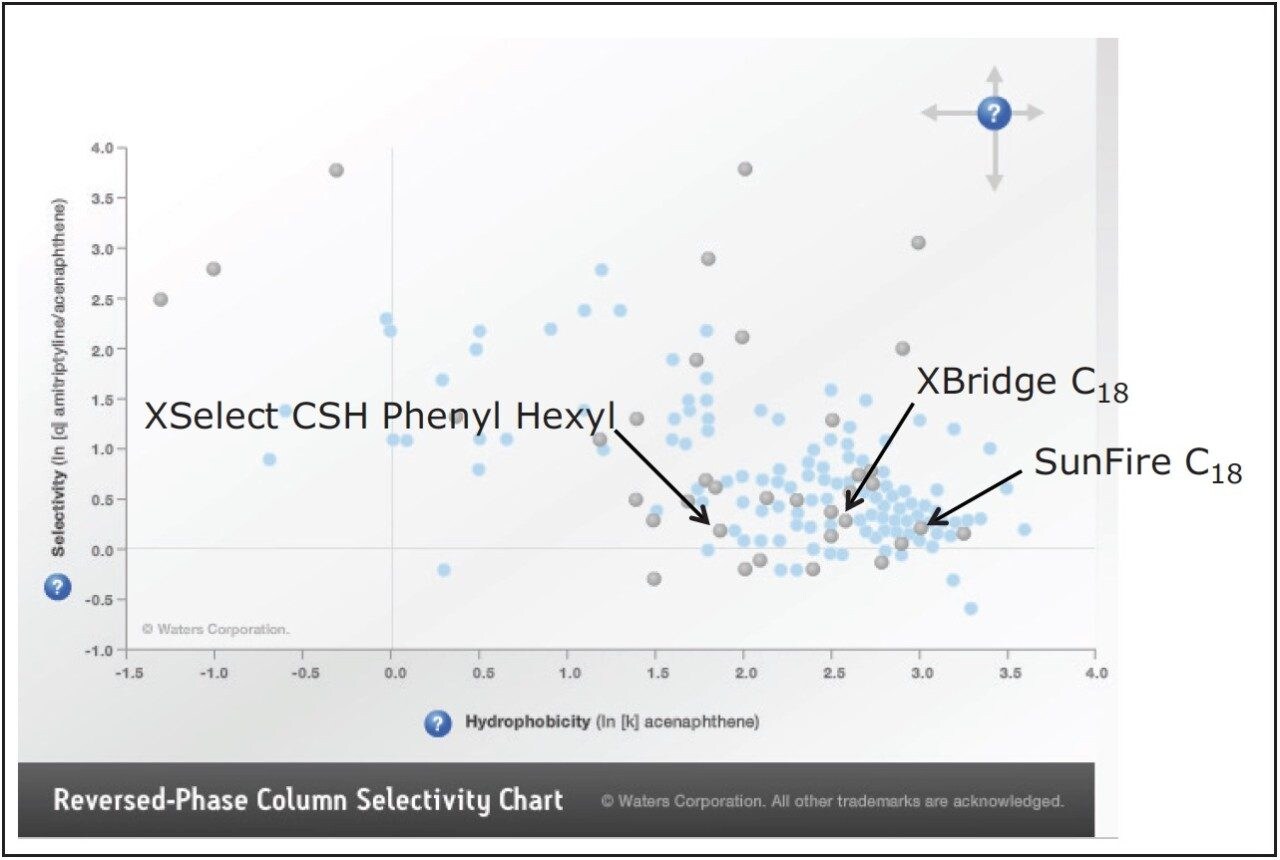
Although the selectivities of the SunFire and XBridge C18 columns were different, it was evident that the separation could not be improved without using a column with a more dramatically different selectivity than the ones already tested. For this reason, and using the reversed-phase column selectivity chart as a guide, the XSelect CSH Phenyl Hexyl column was selected for the subsequent evaluation (Figure 3).
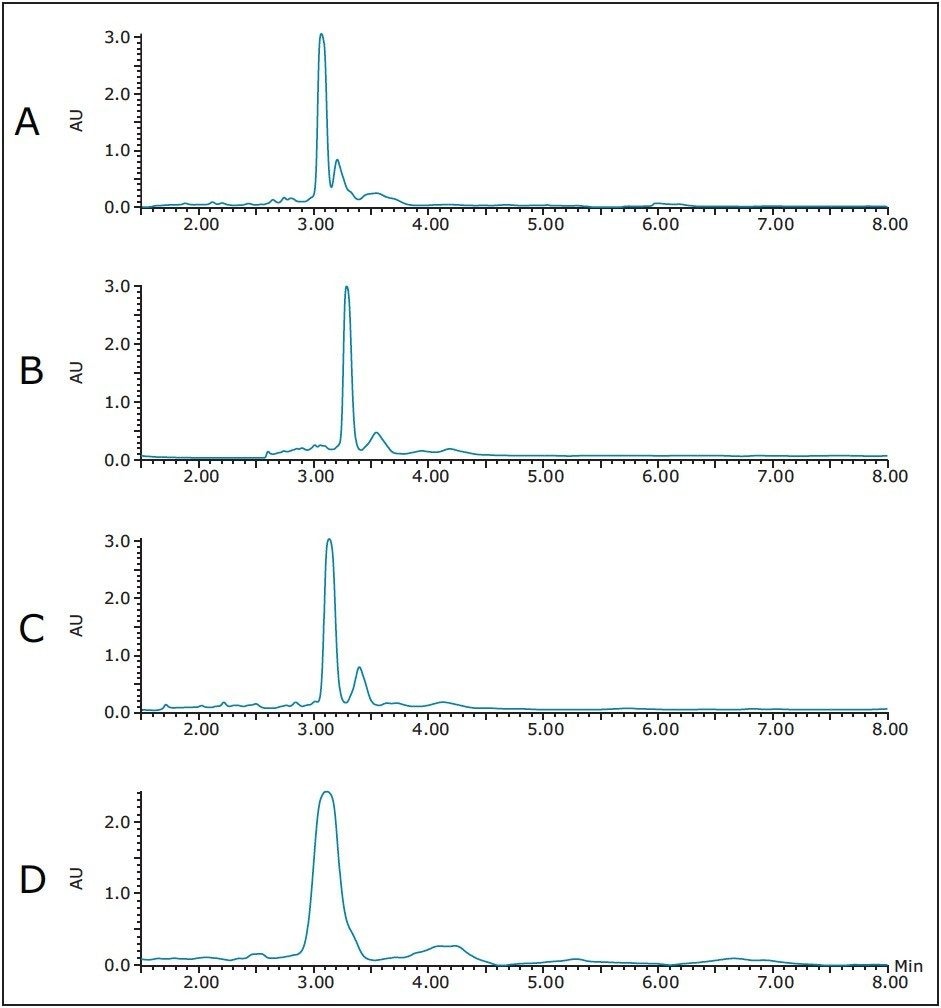
Although the slope of the gradient on the XSelect analytical column was slightly steeper, because the column was 50 mm longer and the packing had a different selectivity, the first scaled 20 μL injection revealed the emergence of 2 contaminant peaks (Figure 4A). As shown in Figure 4B, refocusing and reducing the rate of gradient change from 1.42%/column volume (cv) to 0.71%/cv almost fully resolved the contaminant peak. An even shallower gradient slope at 0.28%/cv fully resolved the contaminant (Figure 4C). In the interest of further improving the resolution between the peptide product and the contaminating by-product, the gradient was modified to run from 16–24%B, yet the slope was maintained at 0.28%/cv (Figure 4D). As expected, the impurity eluted later and the resolution between it and the peptide product was improved. Since the gradient slope was so shallow, it was unlikely that any further method development on this column would yield additional improvement in resolution.
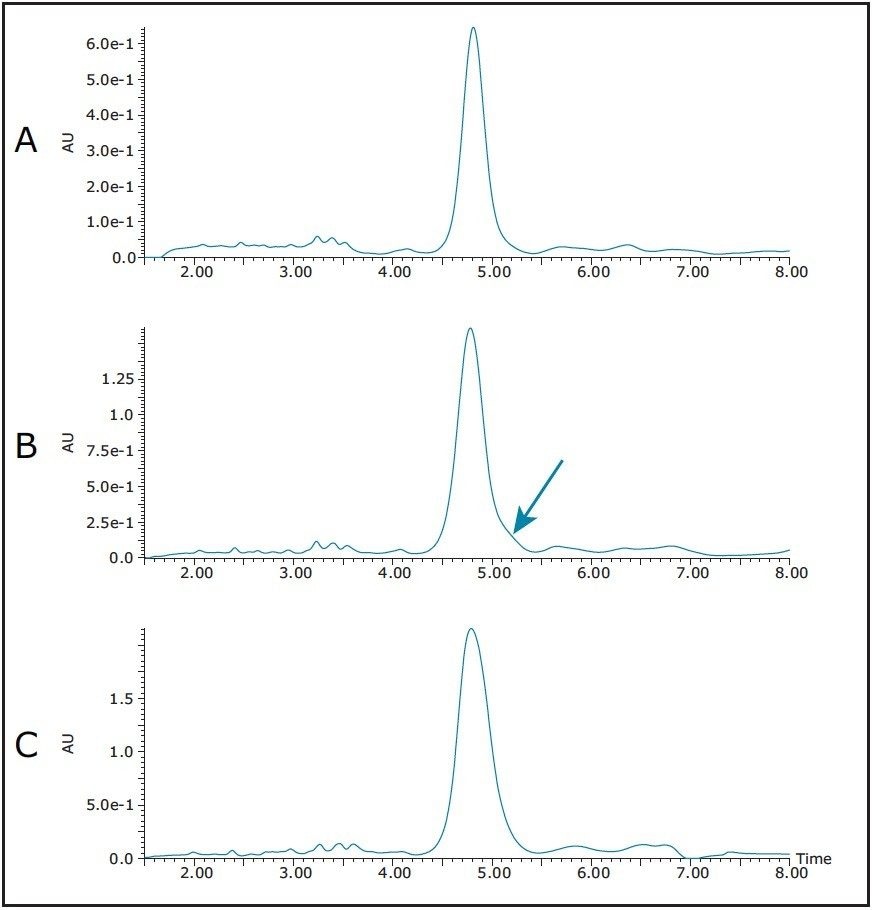
More peptide sample was dissolved in DMSO and a loading study was performed on the 4.6 x 100 mm XSelect CSH Phenyl Hexyl column. A very conservative loading volume of 5 μL (Figure 5A) showed excellent separation between all of the sample components. A 10 μL injection revealed yet another slight shoulder eluting on the backside of the peptide product peak (Figure 5B), and the shoulder completely hiding under the product peak with a 15 μL loading (Figure 5C). The 10 μL loading was chosen for scale up because this would give a higher load on the prep column, yet still give enough resolution to effectively isolate pure product.
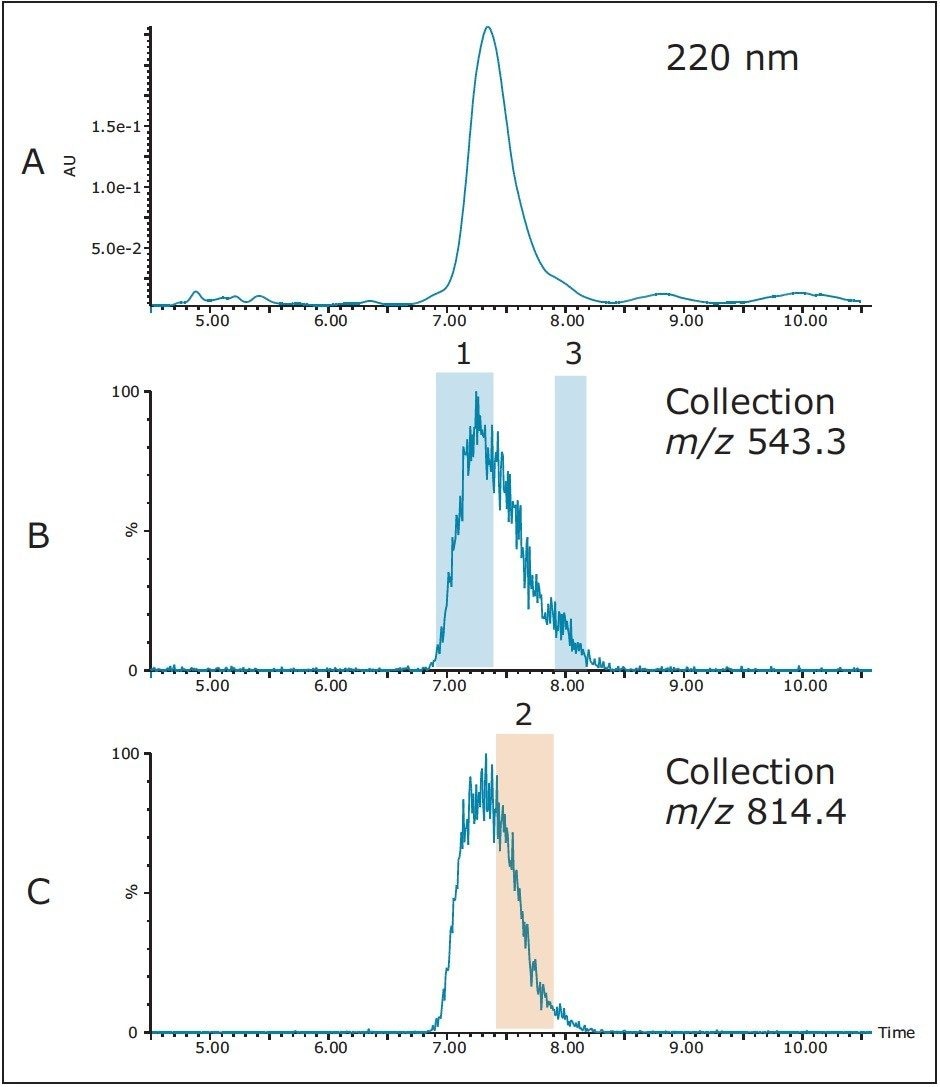
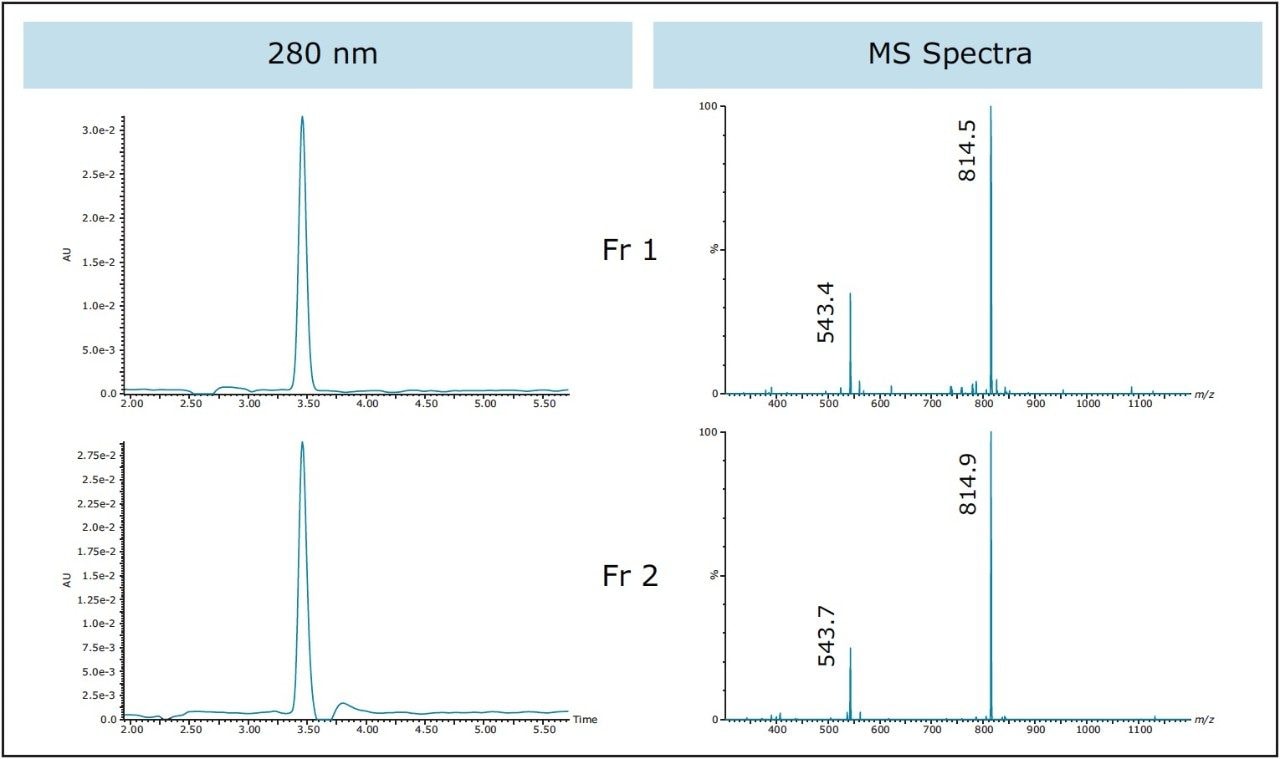
Geometric scaling from 10 μL on the 4.6 x 100 mm analytical column to 256 μL on the 19 x 150 mm preparative column gave identical chromatography at 220 nm, as expected (Figure 6A), shown here for reference. Mass-directed collection was used for fractionation with the mass triggers corresponding to the most abundant triply-charged and doubly-charged ions in the sample. Fractions 1 and 3 were triggered on the triply-charged ion [M+3H]3+ with an m/z of 543.3 (Figure 6B), while fraction 2 was triggered on the doublycharged ion [M+2H]2+ with an m/z of 814.4 (Figure 6C). With the mass range of the ACQUITY QDa Detector spanning from 30 to 1250 Da, only peptides with multiple charges which fall within this range can be analyzed and isolated. In the interest of conserving time and solvent, the purification run was terminated shortly after the target peptide was collected. UV and mass analysis of the fractions showed the product to be very pure (Figure 7).
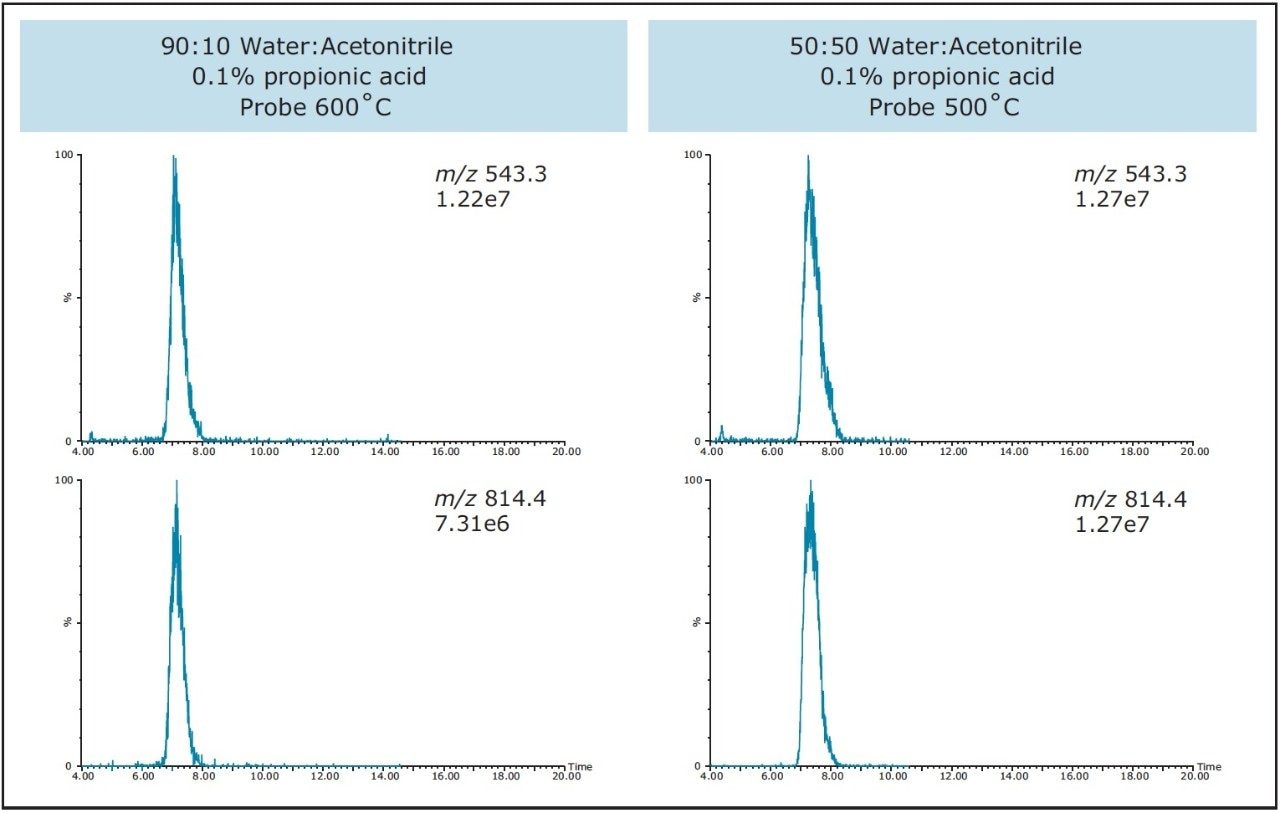
Mass detectors configured in purification systems almost always use a makeup solvent to dilute the tiny sample split from the main prep flow for analysis during the preparative run. The makeup solvent also simultaneously carries the sample to the detectors. Once the target ion is detected and it meets the collection parameters specified by the user, the detector signals that the peak is found and collection begins after the appropriate offset time to the fraction collector is reached. Trifluoroacetic acid (TFA) is commonly used as an ion-pairing reagent in peptide separations because it helps to improve chromatographic peak shape. Unfortunately, TFA also suppresses ionization in mass detection. For this reason, the makeup solvent for this isolation was 50:50 water:acetonitrilewith 0.1% propionic acid. The propionic acid displaces the TFA and improves the ionization in the mass detector. Because of the very hydrophilic nature of the peptide used in these experiments, a second makeup solvent was evaluated – 90:10 water:acetonitrile with 0.1% propionic acid. As shown in Figure 8, both makeup solvents are acceptable for use in the isolation of this peptide. For peptides that are hydrophobic, the 50:50 Water:Acetonitrile makeup solvent is recommended to prevent sample precipitation. For the 90:10 water:acetonitrile composition, the probe temperature was 600 °C. Peak widths measured at half height were slightly narrower for the 90:10 water:acetonitrile composition.
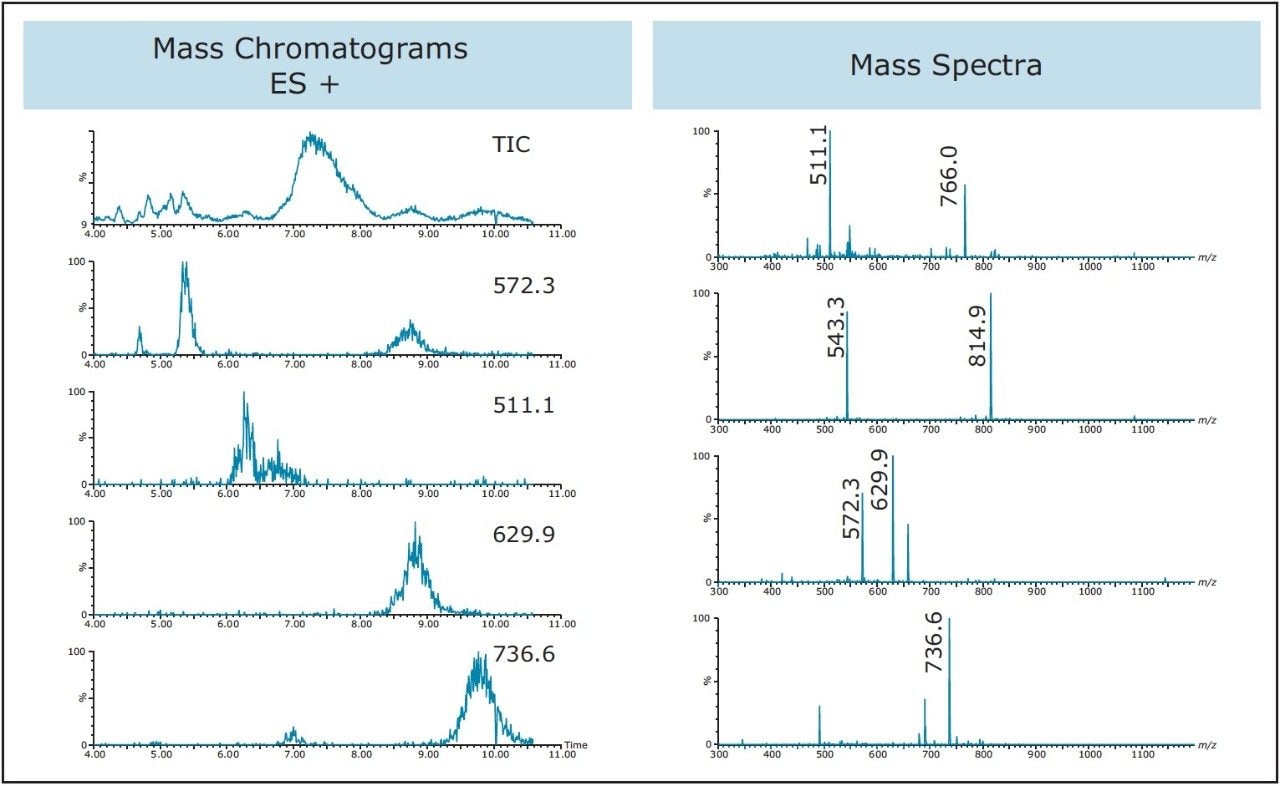
Not only is an ACQUITY QDa effective in identifying and triggering the masses of the target product in peptide isolations and purifications, it is also useful for obtaining mass information about the other compounds that are present in the sample. As discussed earlier, the peptide synthesis and cleavage procedure has the potential to create many different types of impurities. With the chemist’s knowledge of the synthetic protocol and with information obtained from the mass detector, assumptions can be made relating to how the synthesis and/or cleavage procedure can be improved. As shown in Figure 9, masses of impurities in the sample can be determined by looking at the mass spectra and mass chromatograms. Armed with this information, the chemist can then make prudent decisions for process enhancements.
720005450, July 2015