In this application note, we report a method for analysis of vitamin A and D3 in vitamin premixes and concentrates in one analytical run without purification or dilution. The metrological properties of the UPC2 and the advantages of the method compared to the HPLC are also discussed.
Simultaneous analysis of fat-soluble vitamins in foods is challenging due to their different properties and concentrations. A typical method involves extraction and saponification of fat, followed by high performance liquid chromatography (HPLC) with UV/Vis detection. After saponification the extracts can be analyzed for vitamin A directly, but they have to be diluted due to high abundance of the vitamin A in foods and its high molar extinction. Unfortunately, the dilution makes it impossible to detect vitamin D3 in the same solution. To measure vitamin D3, the extracts have to be cleaned on a semi-preparative chromatograph and concentrated. For these reasons vitamins A and D3 had to be analyzed separately. The HPLC of these compounds suffers from a long runtime, slow equilibration, and poor reproducibility.
As the extraction and saponification of separate vitamin A and D3 methods are identical, we investigated whether it would be possible to apply Waters UltraPerformance Convergence Chromatography (UPC2) to analyze extract for vitamin A and D3 in a single chromatographic run.
UPC2 is a separation technique that uses compressed carbon dioxide as the primary mobile phase. It takes advantage of sub-2 μm particle chromatography columns, the low viscosity of CO2, and an advanced chromatography system. This differs from traditional HPLC and improves the sensitivity of this assay. UPC2 also generates much less solvent waste compared to conventional liquid chromatography.1 In this application note, we report a method for analysis of vitamin A and D3 in vitamin premixes and concentrates in one analytical run without purification or dilution. The metrological properties of the UPC2 and the advantages of the method compared to the HPLC are also discussed.
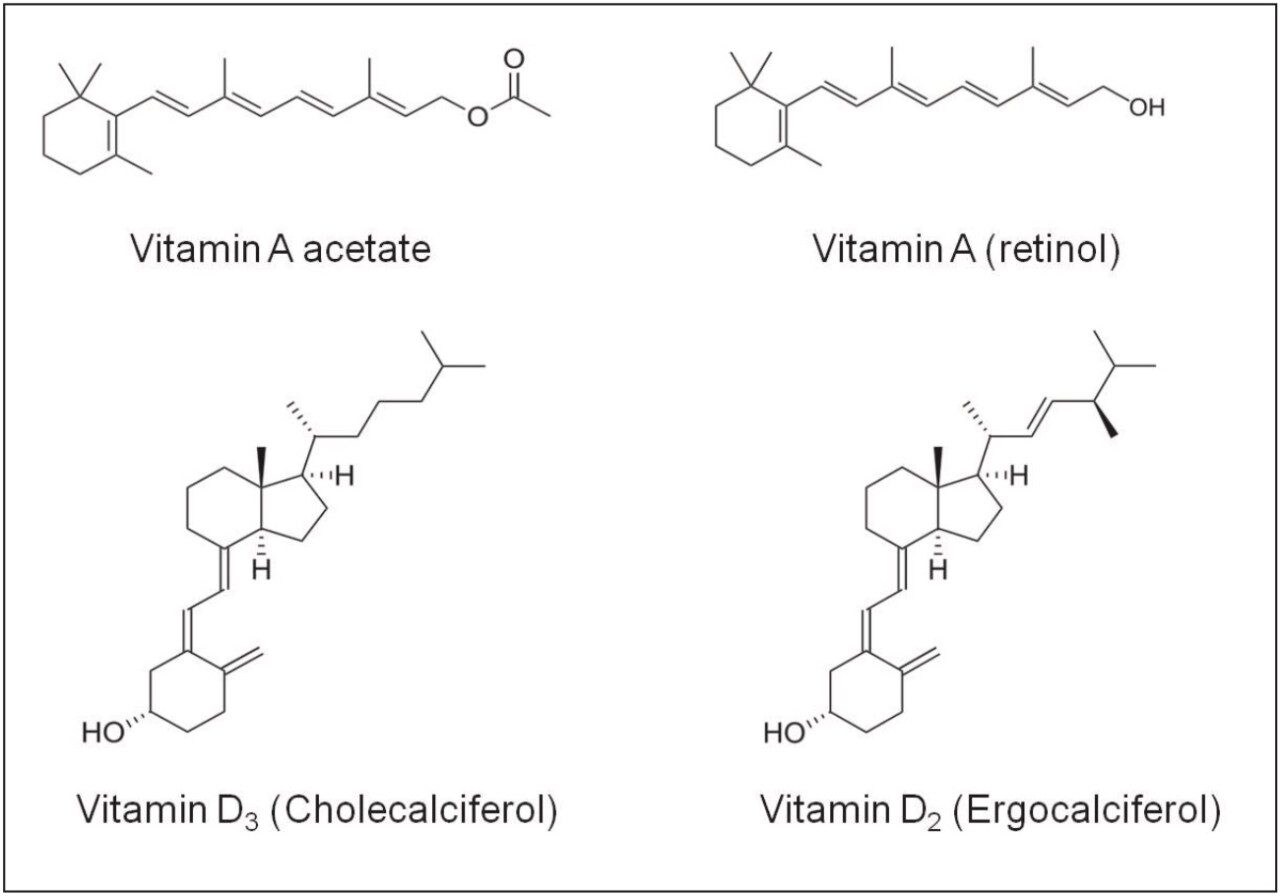
Retinyl acetate (vitamin A acetate) was purchased from Sigma-Aldrich, cholecalciferol (vitamin D3) and ergocalciferol (vitamin D2) were purchased from the US pharmacopeia. The vitamin premix was from DSM Nutritional Products, Switzerland. Figure 1 shows the structures of relevant compounds used in this study.
1.5 g vitamin raw sample was weighed and spiked with 1 mL of vitamin D2 solution (internal standard) into a 250-mL Erlenmeyer flask. 50 mL (±10%) ethanol was added, along with 10 mL of 50% KOH, and 2 mL of 33% sodium ascorbate. The samples were saponified for about 1 hour at 80 °C to 85 °C in a water bath. After saponification, the samples were cooled down to room temperature and extracted with a n-hexane diethyl ether mixture. The extract was washed with de-ionized water until neutral, and the extracts were made up to 50-mL with n-hexane. The solution was filtered through a 0.2 μm syringe filter into 2-mL autosampler vials and analyzed by UPC2/PDA.
|
UPC2 conditions |
|
|---|---|
|
System: |
ACQUITY UPC2 |
|
Detector: |
PDA |
|
Software: |
Empower 3 |
|
Column: |
ACQUITY UPC2 BEH 3.0 mm x 100 mm, 1.7 μm |
|
Mobile phase A: |
Compressed CO2 |
|
Mobile phase B: |
isopropanol |
|
Wash solvent: |
Methanol |
|
Flow rate: |
1.7 mL/min |
|
APBR: |
2,000 psi |
|
Column temp.: |
55 °C |
|
Sample temp.: |
10 °C |
|
Injection volume: |
7.0 μL |
|
Detection: |
UV 260 nm |
|
Gradient: |
0.5% to 20% B in 9.9 min Hold at 20% for 2 min, re-equilibrate for 3 min |
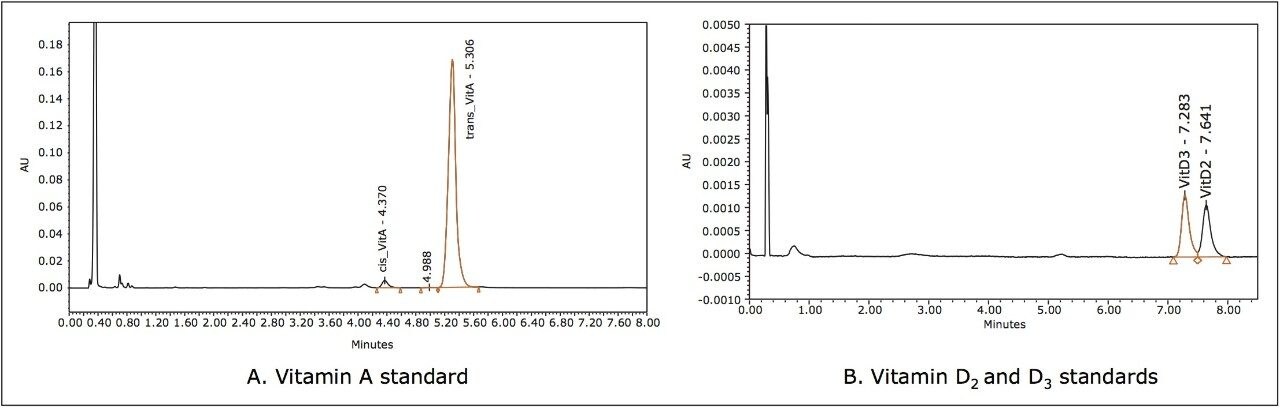
Due to the fact that the molar absorbance of vitamin D3 is low and its concentration in samples is ten times lower than vitamin A, both vitamins A and D3 were quantified at the wavelength of the maximum absorbance of D3. The typical chromatograms obtained at 260 nm UV wavelengths for vitamin standards in solvent are shown in Figure 2. Chromatograms of the sample extract are shown in Figure 3.
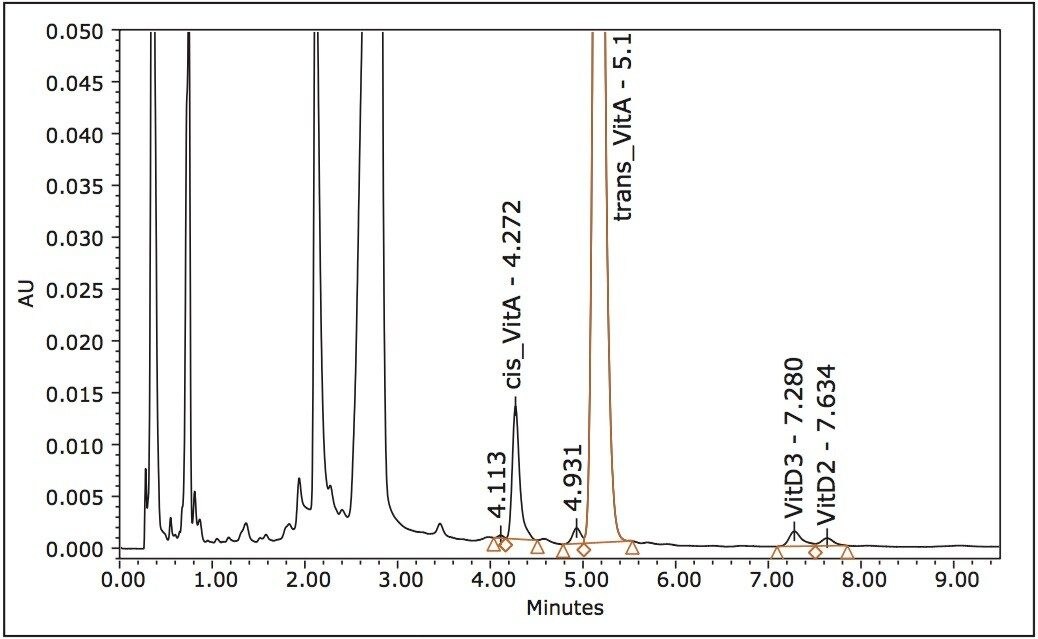
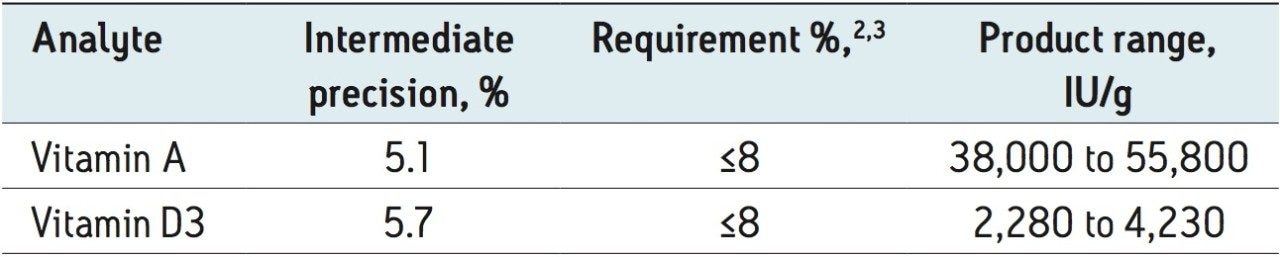
The linearity of the assay was investigated using standard solutions. The average peak areas of triplicate injections of seven concentrations were used. Vitamin D2 was used as the internal standard for the quantification of vitamin D3; there was no internal standard used for vitamin A. The coefficient of the determinations R2 for vitamins A and D3 were 0.9998 and 1.0000, respectively. The intermediate precision and reproducibility of the assay over a three-month period are shown in Table 1. The intermediate precision of the method for vitamin A was 5.1% and 5.7% for vitamin D3, both falling well within the requirement of ≤8%.
To evaluate ruggedness, changes were made to the flow rate, system pressure, and column temperature to observe their effects on the results. There was no statistically significant difference to the ±2% changes on the results, and the chromatographic resolution of the critical D2/D3 pair was maintained above 1.6 (requirement for R ≥1.2).
The utilization of supercritical fluid chromatography using CO2 combined with separation on sub 2-μm particle size columns provided fast and sufficient separation of the vitamins A and D within the same chromatographic run in a broad range of concentrations. The excellent selectivity of UPC2 provided baseline separation of analytes from interfering impurity peaks and allowed quantification of the cis- and trans- retinols and the vitamin Ds at 260 nm UV in the same analytical run. Vitamins D3 and D2 (internal standard) were separated from each other, and eluted before the matrix peak, in contrast to our traditional method. The extracts in n-hexane can be injected directly into the system, which eliminates the solvent exchange step previously required in our laboratory.
A simple and cost-effective assay to analyze vitamin A and D3 in different vitamin raw materials, premixes, and concentrates using UPC2 has been developed and validated. The UPC2/PDA method demonstrates excellent linearity, resolution, and repeatability. The intermediate precision of the method calculated over a three-month period was less than 6%. The introduction of the ACQUITY UPC2 System significantly reduced the consumption of HPLC solvents. Beside CO2, the only other solvent required was isopropanol. The consumption of isopropanol was 0.12 mL per test. The laboratory has therefore significantly reduced the consumption of HPLC solvents as well as the disposal of waste solvents. By simplifying the procedures and eliminating the repartitioning steps, the reduction of solvent usage has streamlined our workflow and decreased the potential exposure of both laboratory staff and the environment to harmful solvents.
720005220, November 2014