
This application note describes the isolation of the flavonoids from Ginkgo biloba leaf powder using a Waters Prep 150 LC System
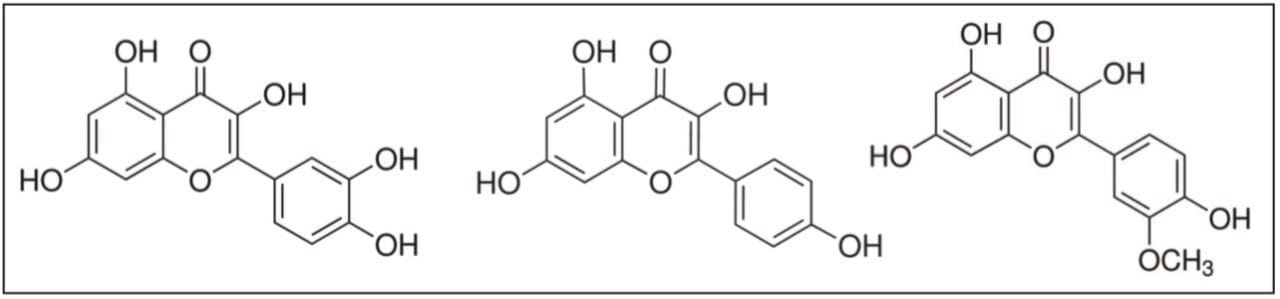
Ginkgo biloba leaf has a long history of medicinal use for the treatment of numerous conditions, going back thousands of years.1 Extracts of Ginkgo biloba leaves contain a wide variety of active compounds and are a particularly rich source of flavonoids, primarily quercetin, kaempferol, and isorhamnetin (Figure 1).
Often, the goal in natural product purification is to isolate individual component compounds that may have biological activity. Sufficient amounts of these compounds are isolated for a wide variety of purposes, such as preparation of standards, or for use in other studies (e.g. clinical trials, bioassays). Any isolated compound needs to be of as high purity as possible and needs to be obtained quickly and efficiently. Many techniques for extraction, isolation, and purification of natural products have been previously described.2 Preparative reversed-phase high performance liquid chromatography (RP-HPLC) is a separation technique that is widely used in this kind of endeavor. It is considered to be a rapid, reliable, and robust technique that has wide applicability over many classes of compounds. This application note describes the isolation of the flavonoids from Ginkgo biloba leaf powder using a Waters Prep 150 LC System (Figure 2).

Preparative chromatographic separations were carried out using the Prep 150 LC System (Figure 2) consisting of the following Waters components:
|
Pump: |
2545 Binary Gradient Module |
|
Detector: |
2489 UV/Visible Detector with Semi-Prep TaperSlit Flow Cell |
|
Injector: |
Preparative Injector configured with a 5 mL loop |
|
Collector: |
Fraction Collector III |
|
Software: |
ChromScope Version 1.4 |
Analytical chromatographic separations (for method development and final purity checks) were carried out using an ACQUITY UPLC H-Class System equipped with an ACQUITY PDA Detector controlled with Empower 3 Software. Two initial analytical scale separations were developed (one gradient for purity checks and one isocratic for purification) with the conditions described below.
|
System: |
ACQUITY UPLC H-Class |
|
Column temp.: |
50 °C |
|
Flow rate: |
0.50 mL/min |
|
Mobile phase A: |
0.2% Formic acid in water |
|
Mobile phase B: |
Acetonitrile |
|
Gradient: |
85:15 to 30:70 over 13 minutes, then to 5:95 13 to 15 minutes |
|
Detection: |
UV @ 371 nm |
|
Column: |
ACQUITY UPLC BEH C18 Column, 130Å, 1.7 μm, 2.1 mm x 100 mm |
|
System: |
ACQUITY UPLC H-Class |
|
Column temp.: |
Ambient |
|
Flow rate: |
1.4 mL/min |
|
Mobile phase A: |
0.2% Formic acid in water |
|
Mobile phase B: |
Acetonitrile |
|
Composition: |
73% A and 27% B |
|
Detection: |
UV @ 371 nm |
|
Column: |
XBridge BEH C18 Column, 130Å, 5 μm, 4.6 mm x 100 mm |
The preparative separation was geometrically scaled from the analytical method to the 19 x 100 mm XBridge Column and is described below.
|
System: |
Prep 150 LC |
|
Column temp.: |
Ambient |
|
Flow rate: |
24.8 mL/min |
|
Mobile phase A: |
0.2% Formic acid in water |
|
Mobile phase B: |
Acetonitrile |
|
Composition: |
73% A and 27% B |
|
Injection vol.: |
0.25 mL |
|
Detection: |
UV @ 371 nm |
|
Column: |
XBridge BEH C18 OBD Prep Column, 130Å, 5 μm, 19 mm x 100 mm |
Ginkgo biloba leaf powder (20 g) was added to 100 mL of methanol and sonicated for 60 minutes. Following the addition of 40 mL of 3N hydrochloric acid, the mixture was brought to a boil and allowed to reflux for 90 minutes. Using this reflux technique provided hydrolysis of the flavone glycosides to the respective flavonoids. After cooling to room temperature, this extract passed through a Whatman #1 filter paper and was used without further treatment.
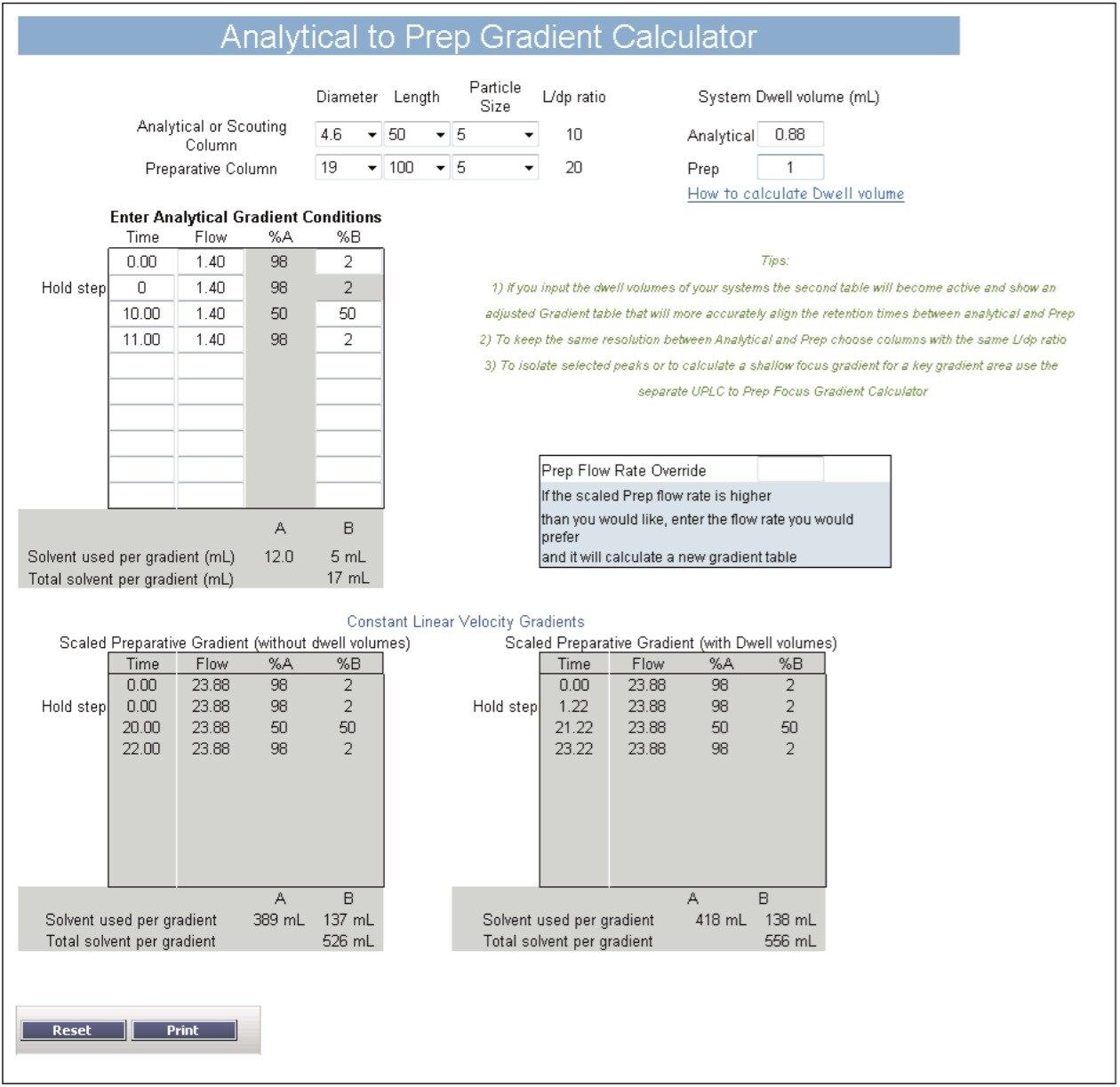
Gradient UPLC analysis of the prepared extract indicated quercetin and kaempferol concentrations of 0.088 and 0.104 mg/mL, respectively (a quantitative standard of isorhamnetin was not available), and an approximate overall purity of 11%, 14%, and 5% for the 3 compounds by UV (371 nm) area percent (Figure 3). Using a previously developed analytical isocratic HPLC method (Figure 4), the flow rate for the preparative separation was scaled to a 19 mm I.D. column using the Analytical to Prep Gradient Calculator built into the ChromScope Software (Figure 5). The Analytical to Prep Gradient Calculator is an easy-to-use tool that aids in analytical-to-preparative scaling calculations providing both flow rate and gradient time calculation information. Proper scaling of analytical methods to prep allows for method development at an analytical scale, preserving sample and lowering solvent usage during method development. Scaled preparative chromatography will be very similar to the analytical chromatography, increasing confidence in the ability of the preparative method to collect peaks of interest. A previously published application note3 describes in greater detail these scaling calculations.
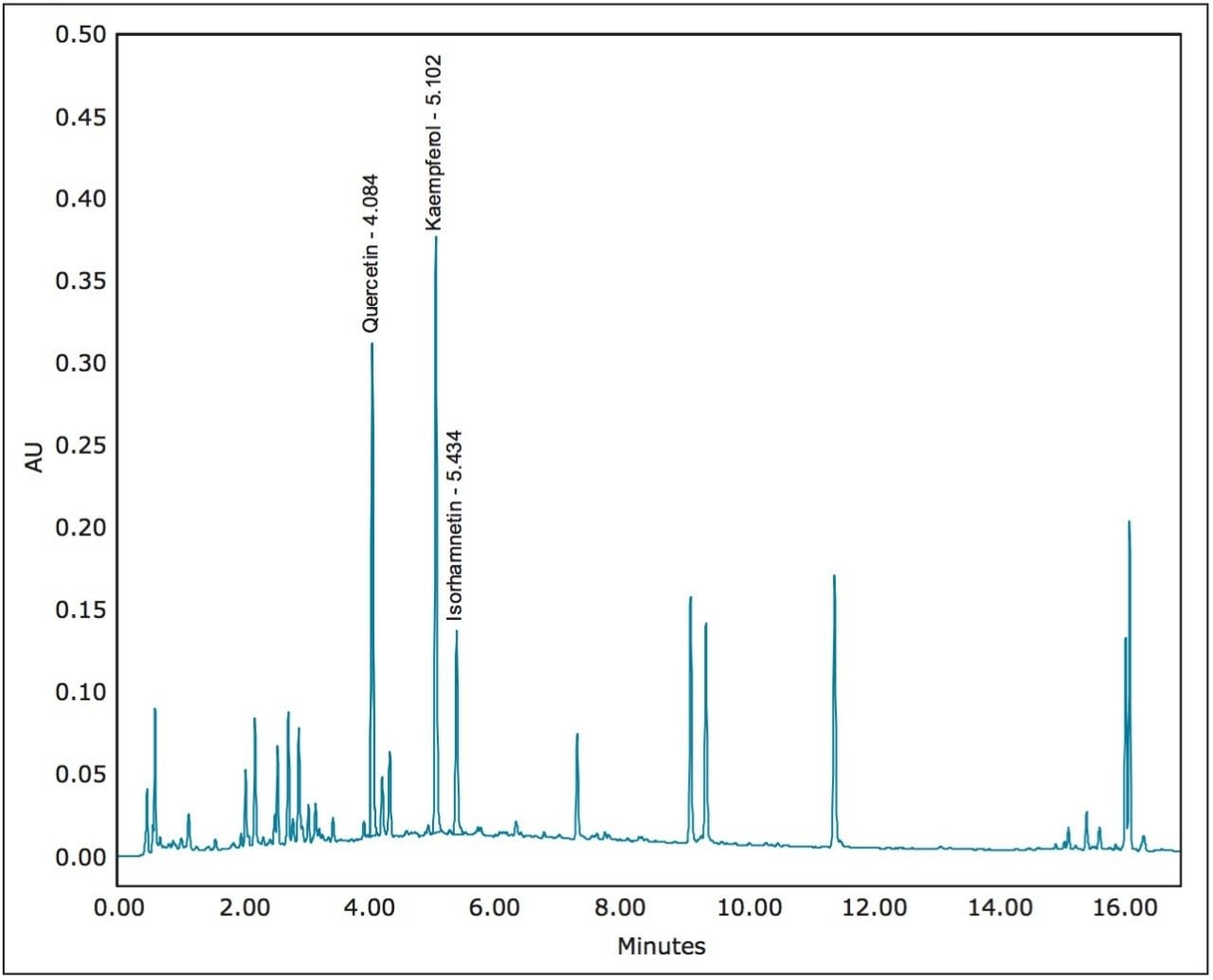
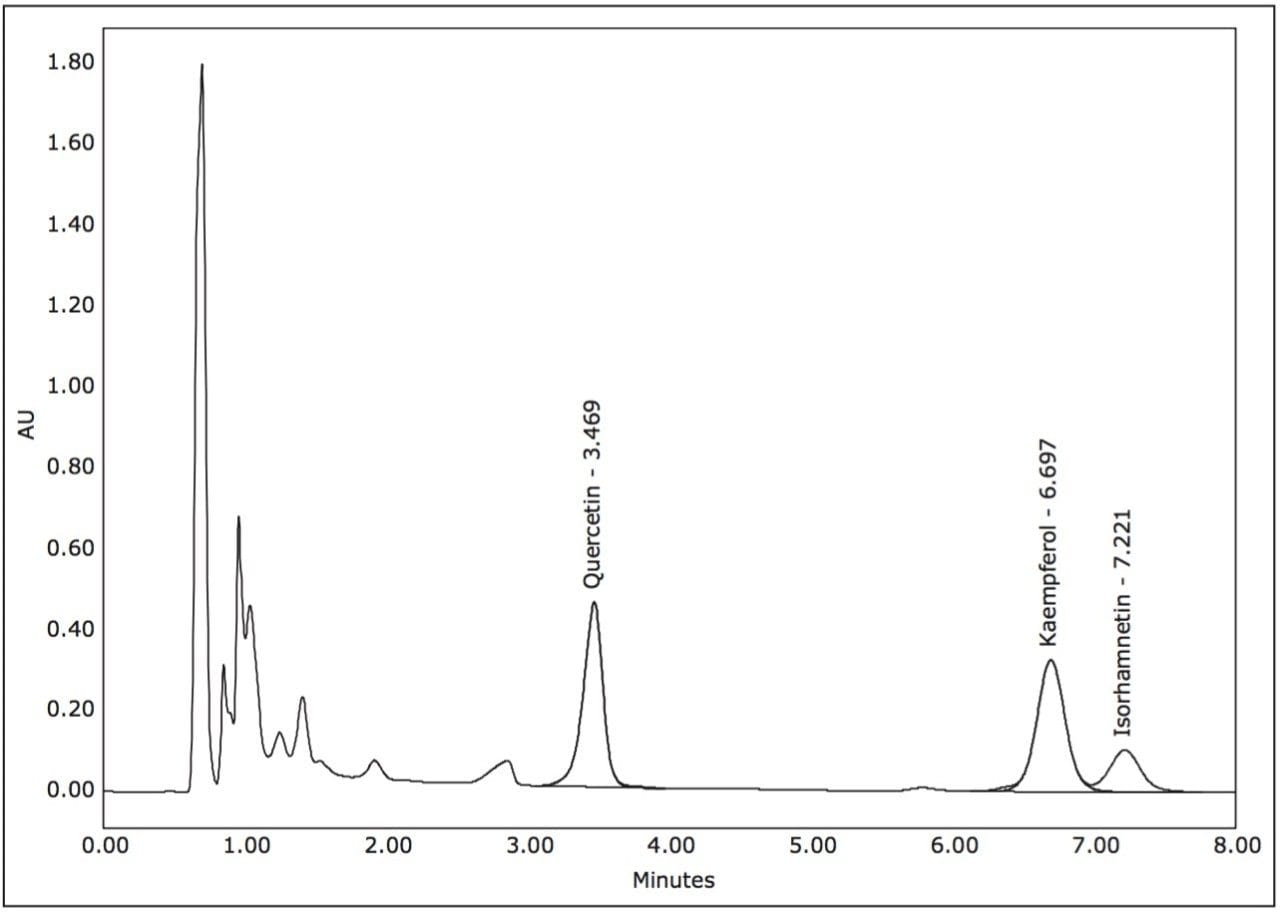
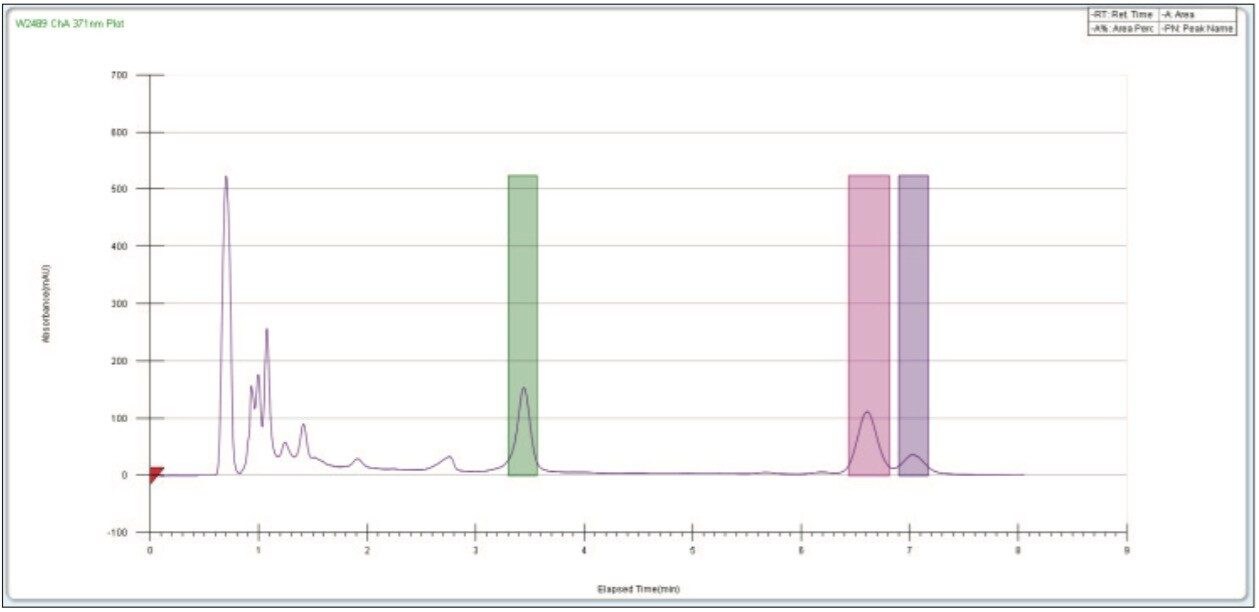
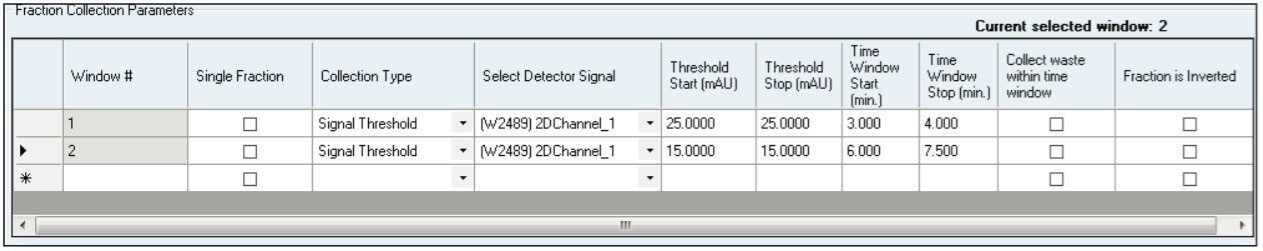
Using a 0.25 mL injection, acceptable preparative separations were achieved (Figure 6) with the isocratic purification separation method and threshold based fraction collection method. Fraction collection was easily set up through the Chromscope Software for threshold collection within windows (Figure 7). In this example, a pair of collection windows is defined (3.0 to 4.0 and 6.0 to 7.5 minutes), and any peaks that eluted outside of that window are not collected. To be collected, peaks eluting within the window must meet peak threshold criteria (in this case, 25 mAU for the first window, 15 mAU for the second window). Purity analysis of the collected fractions showed UV area% purity of 87, 96, and 85% for the three compounds, respectively, based on UPLC analysis (data not shown).
720004839, November 2013