This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates improved resolution of XP 2.5 μm Columns over traditional HPLC particle size columns for challenging separations.
Improving the resolution of a related-compounds separation using eXtended Performance (XP) columns.
It is widely accepted that transferring methods to smaller particle sizes can result in faster analysis time. By transferring a method directly to a smaller particle size, there may also be improvements in resolution. As the particle size gets smaller, however, the back pressure across the column will increase. While the use of sub-2-μm columns may necessitate the use of a UPLC System, HPLC users can still realize significant benefits in resolution by transferring their HPLC methods to a eXtended Performance (XP) 2.5 μm Column. This may be particularly beneficial for the separation of complex mixtures, where the added resolution from a smaller particle size column may help to identify impurities or target compounds without resorting to increasing column length and run times.
An example of improved resolution using an XP 2.5 μm Column is demonstrated using a related compounds mixture of abacavir. Abacavir is a nucleoside reverse-transcriptase inhibitor that is used in anti-HIV therapy. The mixture of related compounds contains five compounds, including the main component, abacavir, shown here in glutarate form (Figure 1). The separation of abacavir from its trans-isomer is particularly challenging. Here, the overall improved separation of abacavir from its related compounds is demonstrated, comparing the use of a 3.5-μm column to a high efficiency XP 2.5 μm Column.
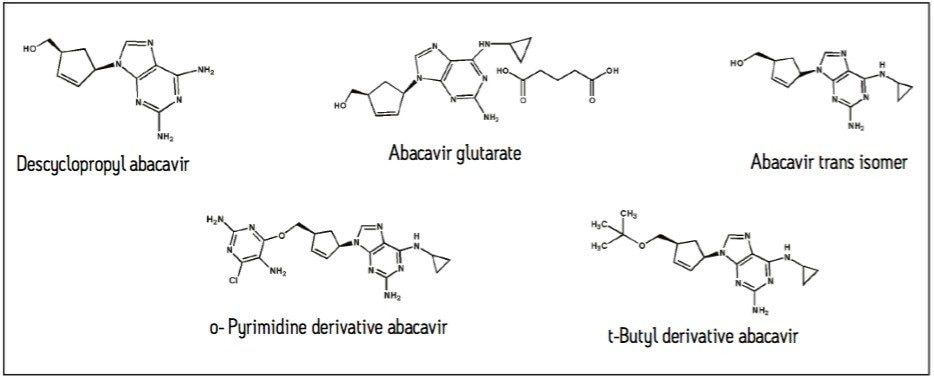
To properly separate related compounds while minimizing extensive method development in HPLC, a highly efficient column with higher resolving power should be used. XP Columns contain 2.5-μm particles packed at high pressures in UltraPerformance hardware. The back pressure allowances of the XP 2.5 μm particle column still allow for use on an HPLC system.
To demonstrate the improvement in performance using XP Columns, the related compounds mixture for abacavir was tested on a 100-mm 3.5-μm, XSelect CSH C18 Column. The same method was run using the same column chemistry and dimensions with XP 2.5 μm Columns. The comparative separations are shown in Figure 2.
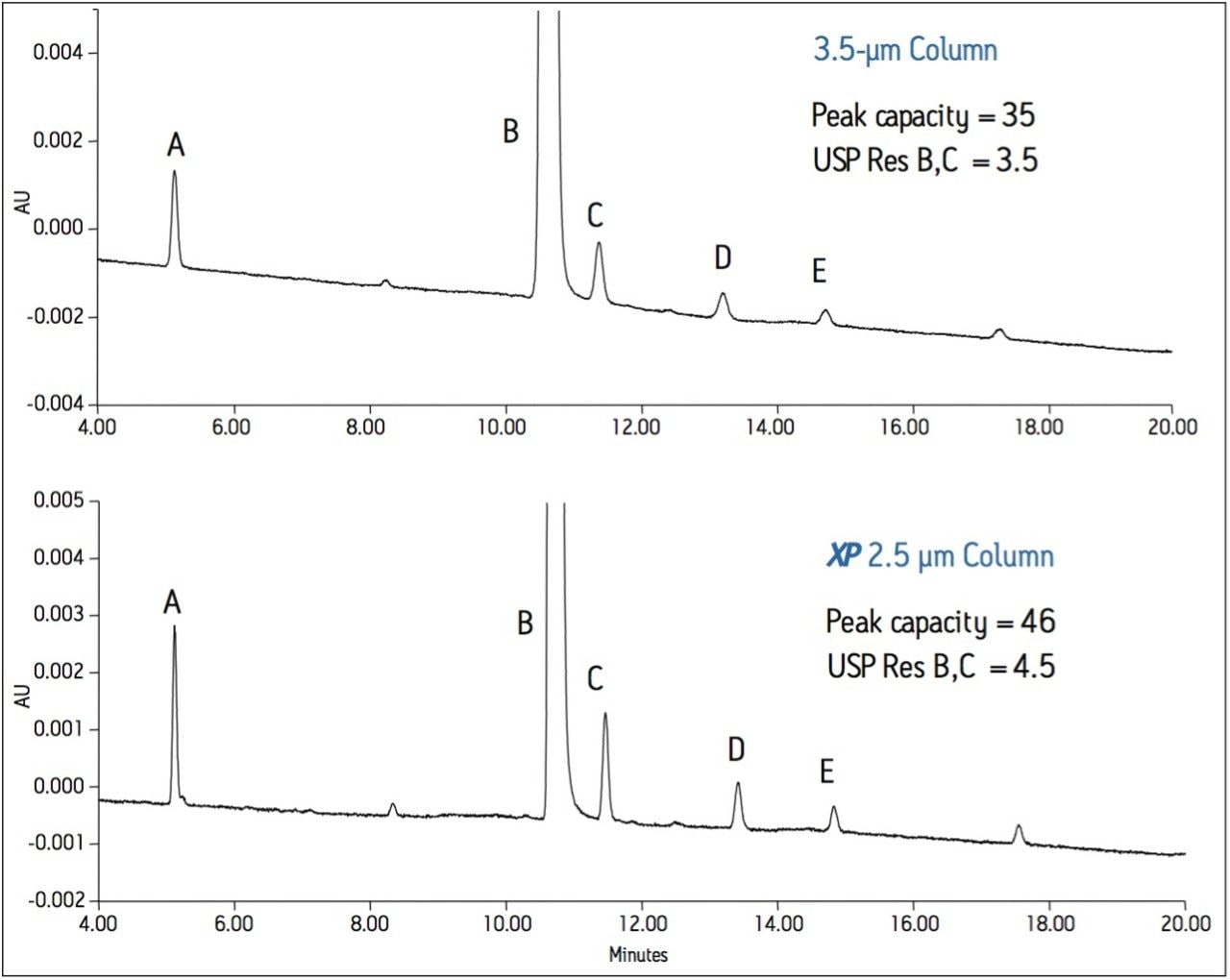
In this case, by simply changing the 3.5-μm column to an XP 2.5 μm Column, significant performance improvements are seen as the overall peak capacity for the separation increases 31%, and the peak heights increase up to 42%. A 28% increase in resolution demonstrates the improved separation between closely eluting compounds abacavir and trans-abacavir. This example illustrates the capability to increase sensitivity and resolution by using an XP Column, which can result in more accurate identification and quantification of target compounds such as impurities.
By transferring HPLC methods to an XP 2.5 μm Column, improvements in resolution and sensitivity can be achieved. This was demonstrated with a related compounds method for abacavir, in which an overall improvement of 31% in peak capacity, up to 42% in peak height, and 29% in resolution were observed by changing the 3.5-μm column to an XP 2.5 μm Column. The use of XP Columns allows HPLC users to maximize the separation performance on their HPLC systems, lessening the need for further method development, and promoting cost-effective asset utilization.
720004706, May 2013