
This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates the benefits of utilizing MassLynx 4.1 Software with OpenLynx Open Access Application Manager in conjunction with an ACQUITY UPLC System and the ACQUITY QDa Detector in a medicinal chemistry laboratory.
By combining UV and mass detection with open access software in a synergistic UPLC system, medicinal chemists can rely on rapid chromatographic separation and confirmation of the identity of reaction products in a chemical synthesis.
There has been a growing demand in the pharmaceutical industry to improve and accelerate the drug development process. This has resulted in new working practices being introduced into all stages of the drug discovery and development process, with goals to ensure quality, drive efficiency, and increase productivity.
Since about 1995, medicinal chemists have been able to analyze more than 100,000 samples a year by liquid chromatography/mass spectrometry (LC-MS) to monitor synthetic reactions and investigate the structure or purity of new chemical entities. One technology that has enabled this high workload is MassLynx Software’s OpenLynx Open Access. This tool provides chemists with fully automated workflows that lead them through sample submission, method selection and reporting options.
The ACQUITY QDa Detector is a mass detector that has been designed to work synergistically with a separation system. Its mass detection capabilities are built around the needs of analytical scientists who routinely perform chromatographic assays. The mass spectral information combines seamlessly into the same workflow as an LC analysis, providing more complete separation characterization by confirming the identity of components. By combining an ACQUITY UPLC System with ACQUITY UPLC PDA and ACQUITY QDa detectors in an open access environment, medicinal chemists can to confirm the identity and purity of their synthetic reactions right next to their synthesis.
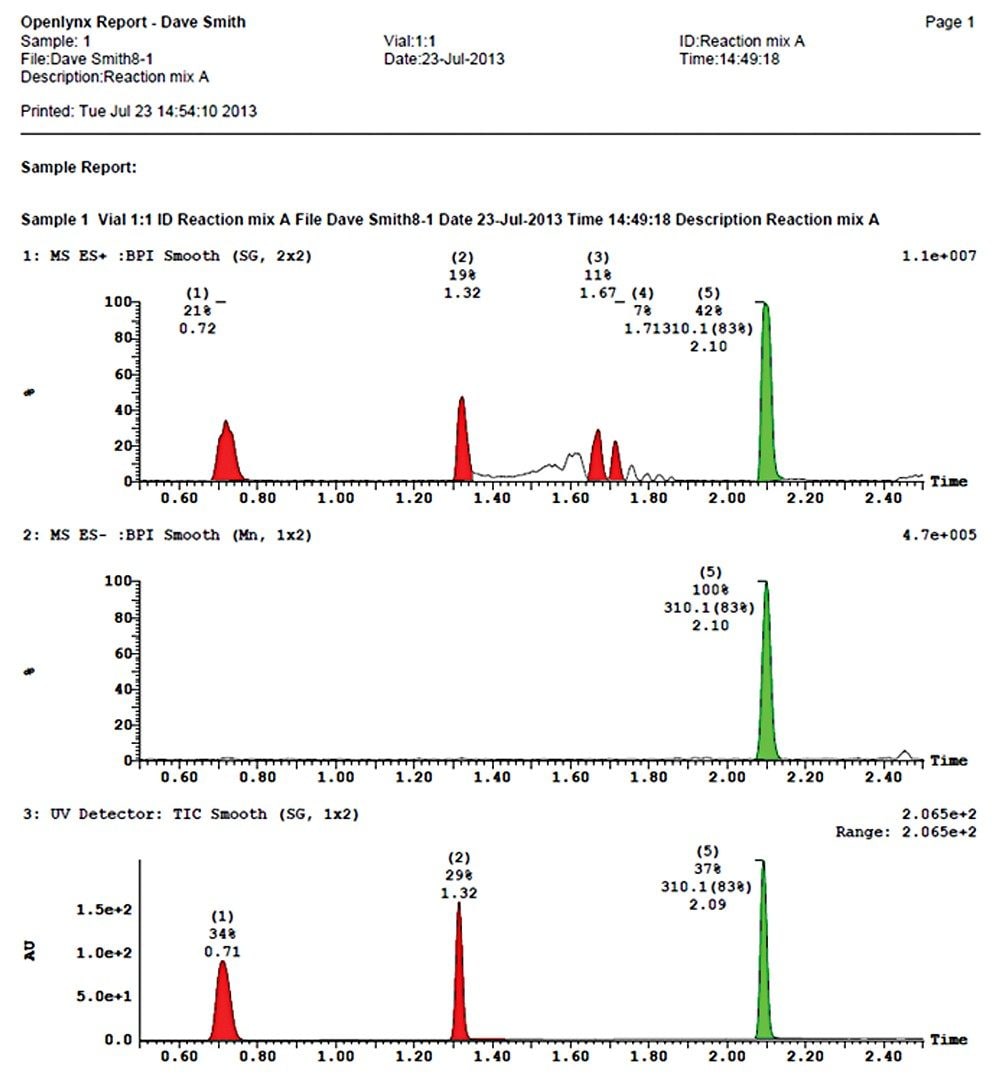
In this example, we demonstrate the UPLC analysis of a typical synthetic reaction mixture using our open access tools along with UV and mass detection.
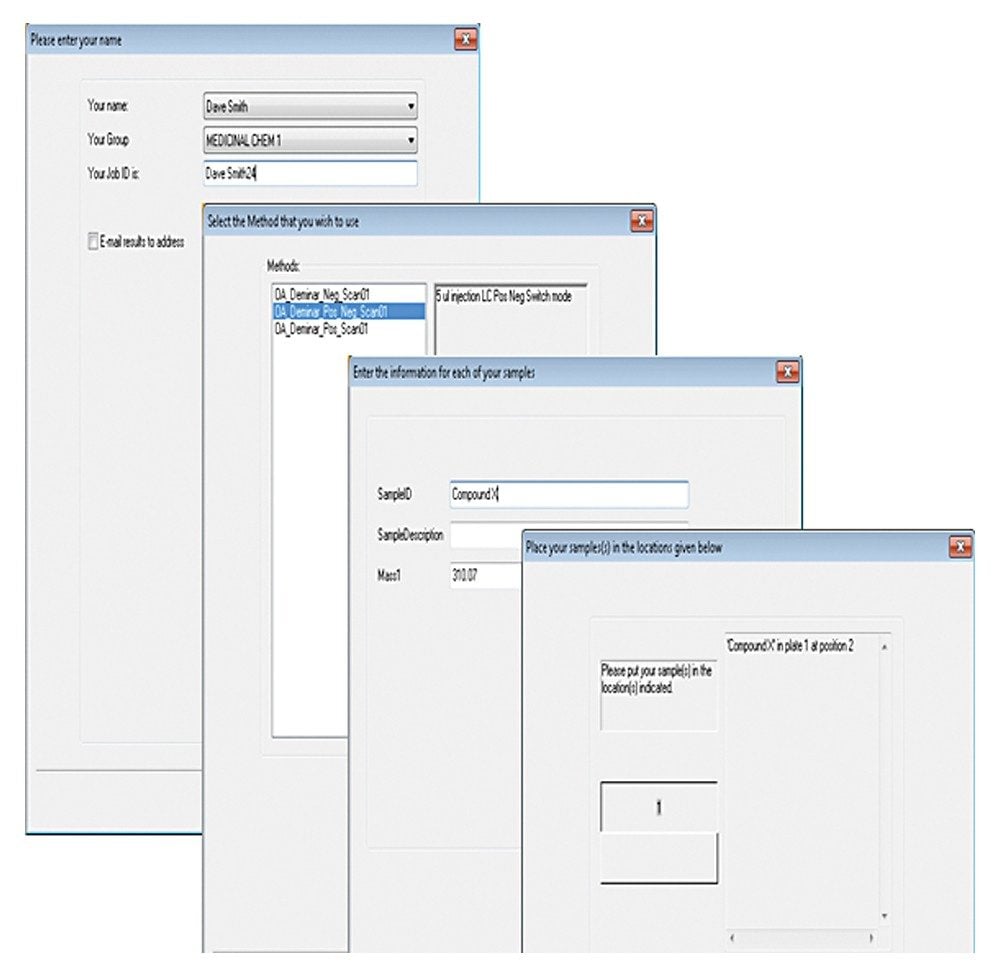
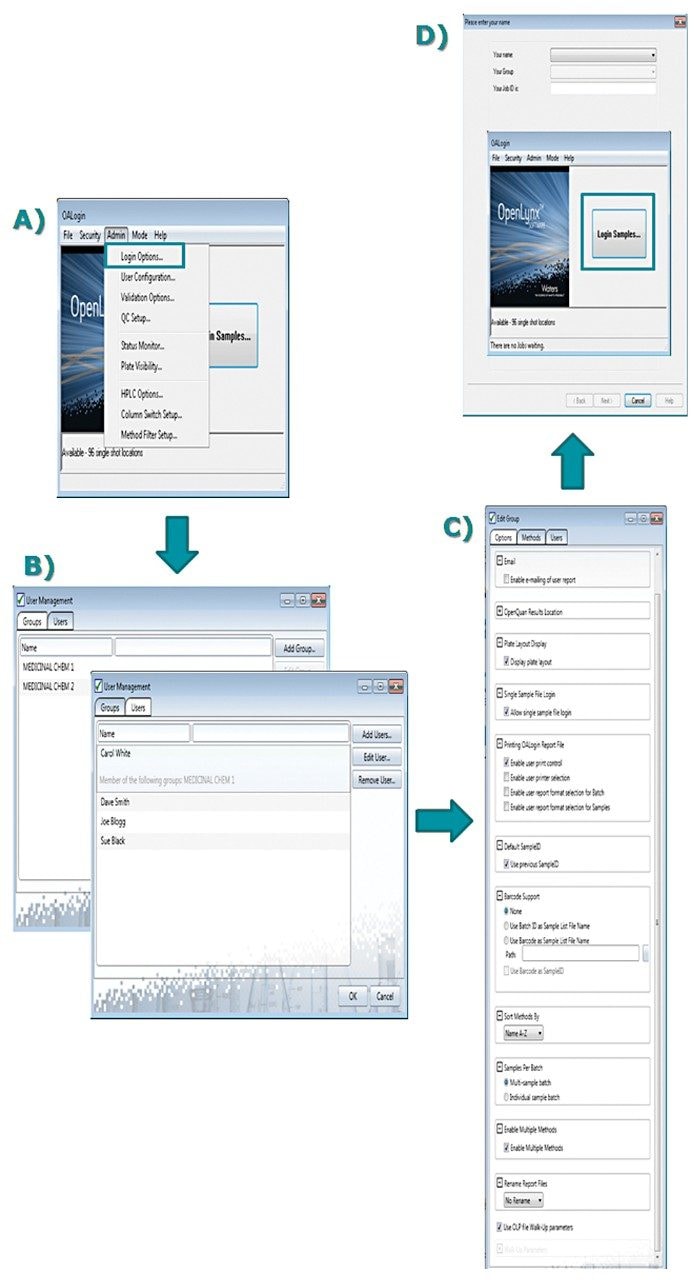
Managing use of this open access environment is straightforward:
By combining an ACQUITY UPLC System with the ACQUITY UPLC PDA and ACQUITY QDa detectors on MassLynx OpenLynx Open Access Software, Waters has streamlined the workflow of medicinal chemists with a complete package for the rapid chromatographic separation and confirmation of identity of reaction products from a chemical synthesis.
720004802, October 2013