
In this study, a compendial assay method for mometasone furoate 0.1% ointment written in the United States Pharmacopeia (USP) was transferred from HPLC to UPLC using sub-2 μm particle technology with reduced time and narrower peak shape for better chromatography.
Mometasone furoate ointment is a prescription skin medication that belongs to the group of synthetic corticosteroids and is used to relief inflammation and itching caused by various skin problems, such as dermatitis, psoriasis, and eczema. It is applied directly on the affected areas of the skin and it works by reducing inflammation and suppressing an overactive immune system.1 Mometasone furoate 0.1% ointment is available in brand and generic forms.
In this study, a compendial assay method for mometasone furoate 0.1% ointment written in the United States Pharmacopeia (USP)2 was transferred from HPLC to UPLC using sub-2 μm particle technology with reduced time and narrower peak shape for better chromatography. The routine use of the method was conducted over 3000 injections using standard and sample preparations. The goal of transferring methodology to UPLC is to provide cost reductions for manufacturing and improvements in method robustness for routine analysis.
|
LC System: |
Alliance HPLC with 2489 UV/Visible detector |
|
Column: |
Waters XBridge Shield RP18, 4.6 x 250 mm, 5 μm, part number 186003010 |
|
Column Temp.: |
25 °C |
|
Sample Temp.: |
15 °C |
|
Injection Volume: |
20 μL |
|
Flow Rate: |
2.0 mL/min |
|
Mobile Phase A: |
100% water |
|
Mobile Phase B: |
100% acetonitrile |
|
Separation Mode: |
Gradient |
|
Wash Solvents: |
70:30 water:acetonitrile |
|
Detection: |
UV at 254 nm |
|
LC System: |
ACQUITY UPLC with PDA detector |
|
Column: |
ACQUITY UPLC BEH Shield RP18, 2.1 x 75 mm, 1.7 μm, part number 186005605 |
|
Column Temp.: |
25 °C |
|
Sample Temp.: |
15 °C |
|
Injection Volume: |
1.3 μL |
|
Flow Rate: |
0.55 mL/min |
|
Mobile Phase A: |
100% water |
|
Mobile Phase B: |
100% acetonitrile |
|
Separation Mode: |
Gradient |
|
Weak wash (600 μL): |
70:30 water:acetonitrile |
|
Strong wash (200 μL): |
5:95 water:acetonitrile |
|
Detection: |
UV at 254 nm |
|
Data Management: |
Empower 2 CDS |
For 5 replicate injections
Internal standard, stock standard, working standard, and sample solutions were prepared as per the assay method defined in the USP Monograph for Mometasone Furoate Ointment.
Diethyl phthalate internal standard was prepared in acetonitrile at approximately 1.4 mg/mL concentration.
Mometasone furoate stock standard was prepared by dissolving an accurate amount of mometasone furoate reference material in Diluent A (100:1 tetrahydrofuran:acetic acid) to make a solution at approximately 0.2 mg/mL concentration. Internal standard and mometasone furoate stock were diluted with Diluent B (50:50:1 acetonitrile:water:acetic acid) to obtain a working standard of approximately 0.05 mg/mL of mometasone furoate and 0.35 mg/mL of diethyl phthalate, respectively.
Mometasone Furoate Ointment USP, 0.1% manufactured by Perrigo was used in this study and prepared according to the USP methodology. Approximately 1 gram of ointment dissolved in Diluent A (100:1 tetrahydrofuran:acetic acid) was mixed with internal standard, diluted with Diluent B (50:50:1 acetonitrile:water:acetic acid), centrifuged, and syringe filtered through 0.2 μm polyprolylene filter prior to chromatographic analysis. The final concentrations were approximately 0.05 mg/mL of mometasone furoate and 0.35 mg/mL of diethyl phthalate, respectively.
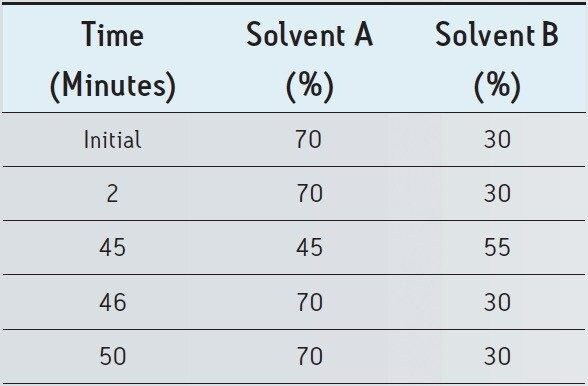
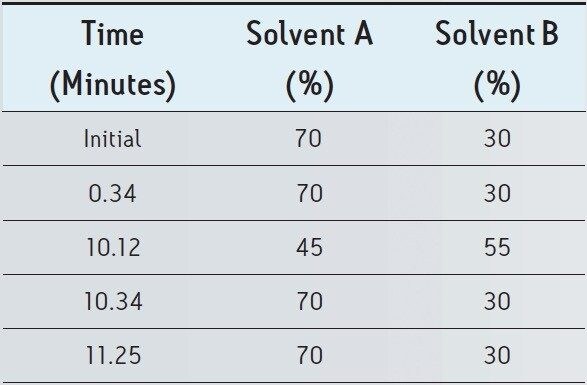
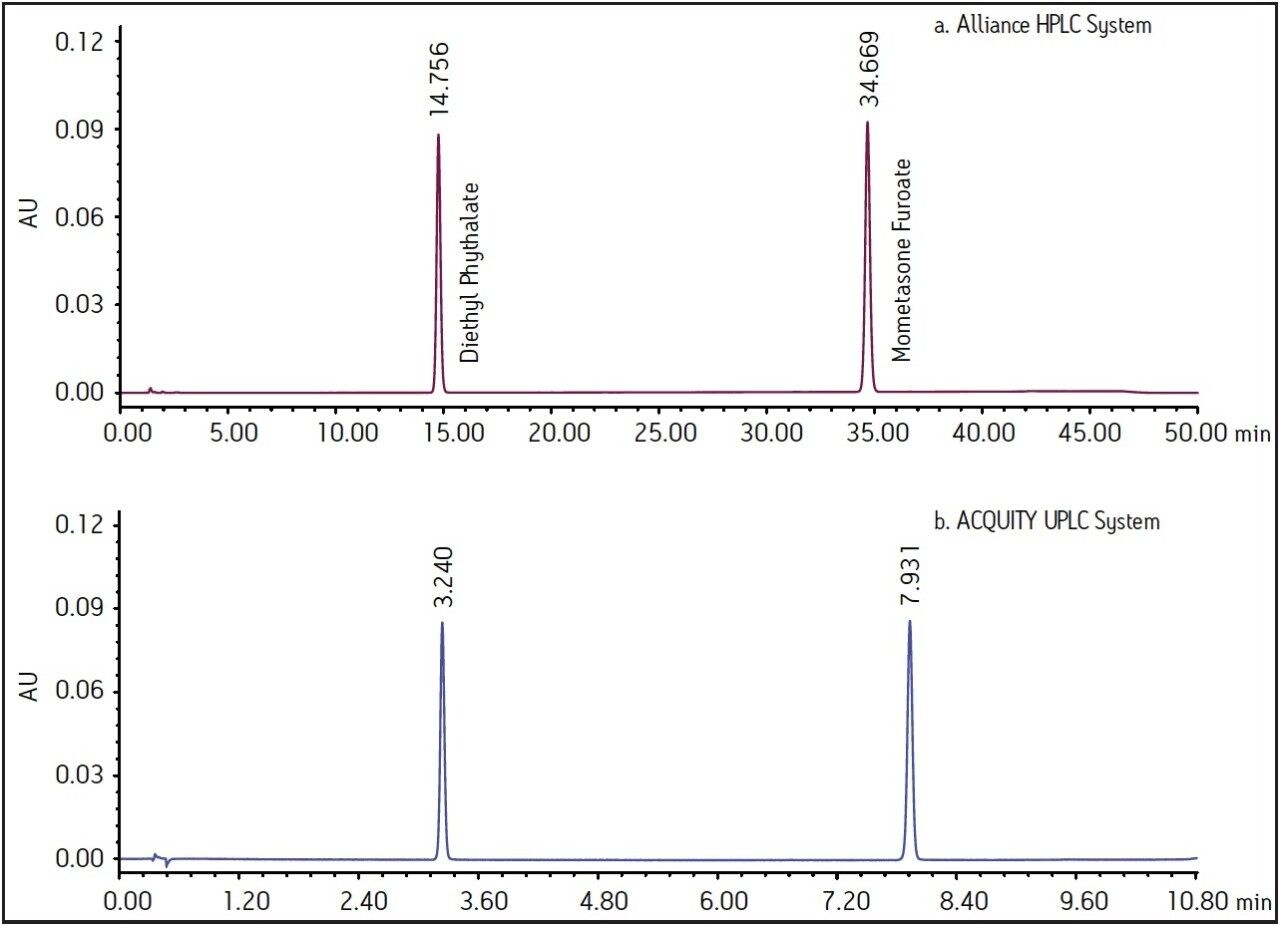
The USP monograph for mometasone furoate ointment specifies a gradient separation at 2.0 mL/min. Standard and sample solutions are injected with an amount of 20 μL on a column with dimensions of 4.6 mm x 250 mm, 5 μm particle size and L60 packing. Column temperature is maintained at 25 °C and the detection wavelength is 254 nm for analysis of standard and sample solutions. The USP methodology for mometasone furoate ointment was tested, as written, using Alliance HPLC and appropriately selected Waters column guided by the Waters Reversed-Phase Column Selectivity Chart. The HPLC analysis was performed with a Waters XBridge Shield RP18 column (4.6 x 250 mm, 5 μm), meeting all the system suitability requirements specified in the USP compedial method. See Table 1 for system suitability results acquired using Alliance HPLC system. The HPLC methodology was transferred to UPLC using the Waters ACQUITY UPLC Columns Calculator as described in the Application Note 720003721EN.3 The resulting method was run on ACQUITY UPLC instrumentation, resulting in narrower peaks and reduced analysis time. The data obtained for the mometasone furoate standard solution acquired using Alliance HPLC and ACQUITY UPLC systems are displayed in Figure 1.
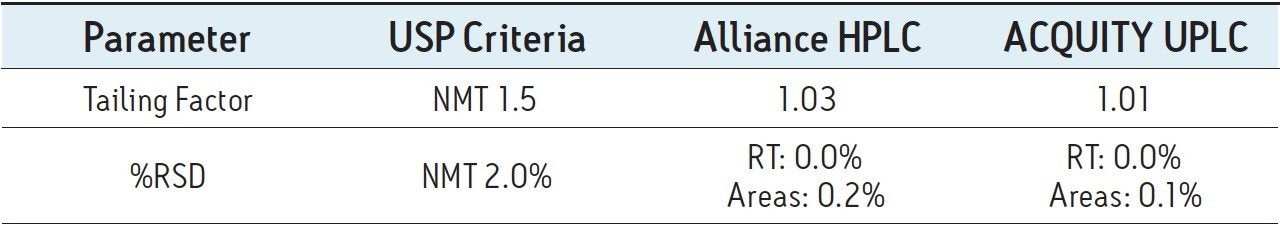
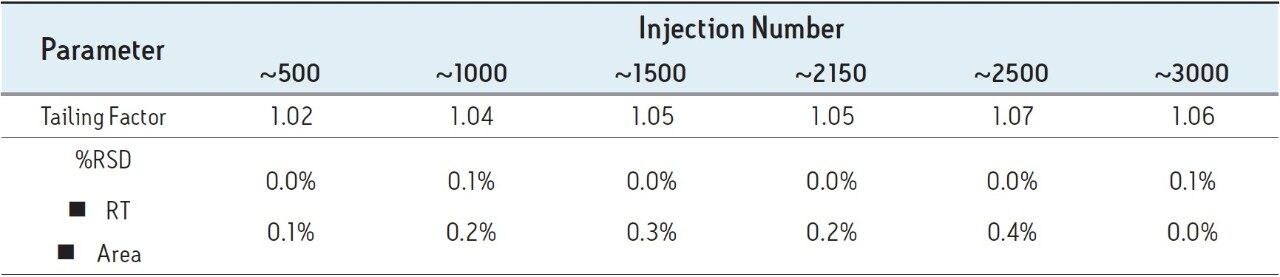
In summary, system suitability requirements for the mometasone furoate ointment on the Alliance HPLC system and the ACQUITY UPLC system passed the USP criteria for the assay method and are summarized in the Table 1.
The routine use study of the UPLC assay method for mometasone furoate ointment was conducted after completion of method transfer to demonstrate that the method is suitable for long-term use within a quality control laboratory. The goal of the study was to evaluate the system performance, column behavior and robustness of the transferred method over 3000 injections of standard and ointment preparation solutions.
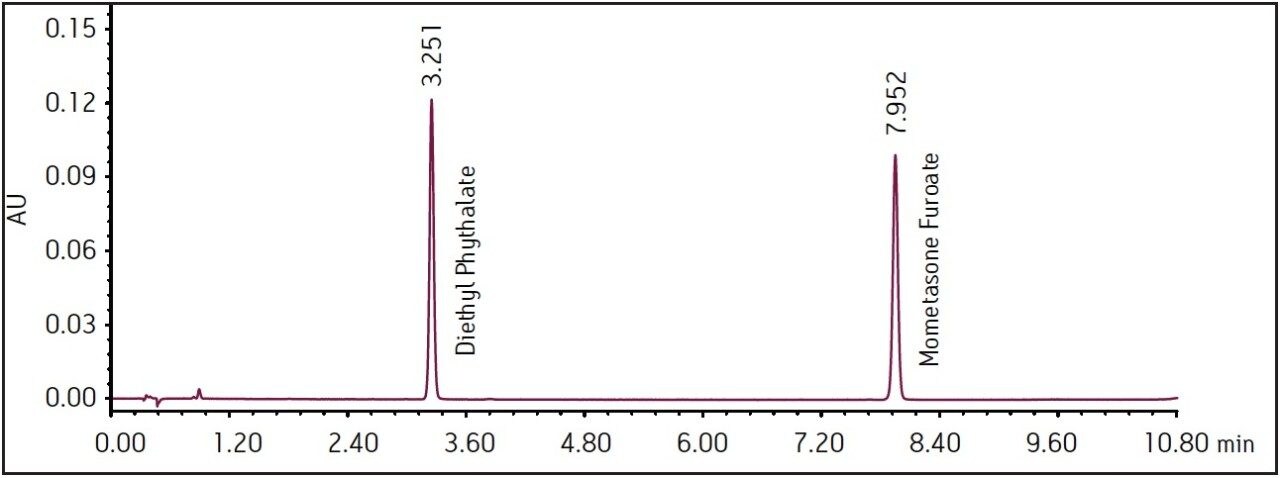
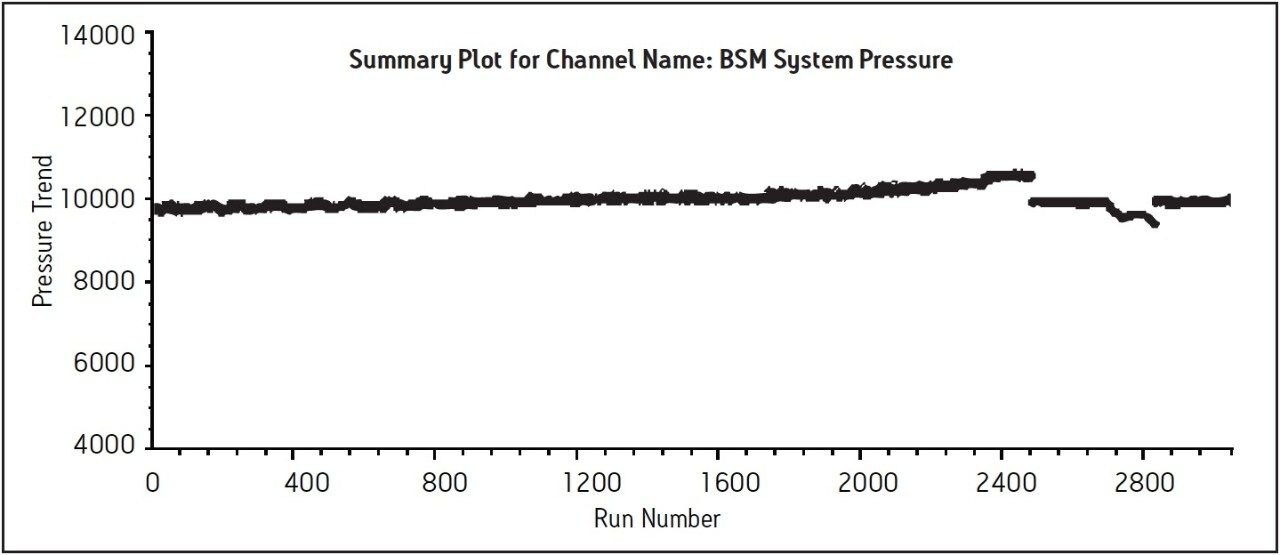
Three separate sample preparations proved to have good reproducibility of the sample preparation procedure. The sample set consisted of 2 injections of blank and 6 injections of sample solution, bracketed with two replicate injections of a mometasone furoate standard. The data obtained for the mometasone furoate ointment sample solution is displayed in Figure 2. The system suitability parameters for the 5 replicate injections of standard were evaluated throughout the course of the study and are summarized in Table 2. Overall, method performance was excellent and system pressure remained stable over the 3000 injections as displayed in Figure 3. At approximately 2500 injections, some increase in system pressure was observed, however replacing column in-line filter returned pressure to the original value. After an additional 200 injections, column temperature increased due to ambient temperature fluctuations in the laboratory, which then slightly reduced pressure until room temperature was controlled. The ACQUITY UPLC column demonstrated excellent performance for over 3000 injections, which is important for a long-term routine analysis in a quality control laboratory.
The transfer of this compendial method proves applicability of successfully transferring existing USP methods from HPLC to UPLC. The resulting UPLC method provides a 78% reduction in run time while maintaining the integrity of the specifications of the USP method. Furthermore, the amount of mobile phase used per injection equals to 6.2 mL compared to 100 mL for HPLC method, which accounts for 94% reduction in mobile-phase consumption per each injection. The other benefit through this transfer is 94% reduction in sample consumption. Overall, the resulting UPLC method provides cost savings for solvent and waste disposal. Moreover, approximately 3000 injections were performed onto the ACQUITY UPLC column with no decrease in method performance and no change in system pressure. The excellent method performance over the course of the study demonstrated that the UPLC assay method for mometasone furoate ointment is applicable for a long-term routine use in a quality control laboratory.
720004147, April 2013