This application note describes a novel, non-invasive sampling method and a sensitive, repeatable LC-TQ MS method for the detection of progesterone and cortisol in different whale species.
The conservation and management of large whale populations require information about all aspects of their biology, life history, and behavior. However, it is extremely difficult to determine many of the important life history parameters, such as reproductive status, without using lethal or invasive methods. As such, efforts are now focused on obtaining as much information as possible from the samples collected remotely, with a minimum of disturbance to the whales. Various excreted samples, such as sloughed skin and feces are being used to determine sex and maturity, as well as life history stage.1,2
Attention has recently shifted more towards what can be analyzed from samples of whale blow for health assessment3 or steroid hormone analysis.4 Hormone analysis is of particular interest as high levels of progesterone can be used as an indicator of pregnancy status, while other steroids, such as glucocorticoids may be markers of the short-term, acute stress response.
A recent study5 reported that it was possible to detect both male and female reproductive hormones in whale blow samples using liquid chromatography-mass spectrometry (LC-MS). Following these findings, we have investigated improving the detection of progesterone and cortisol (the major glucocorticoid identified in many marine mammal species), with a much more sensitive LC-MS/MS quantitative method.
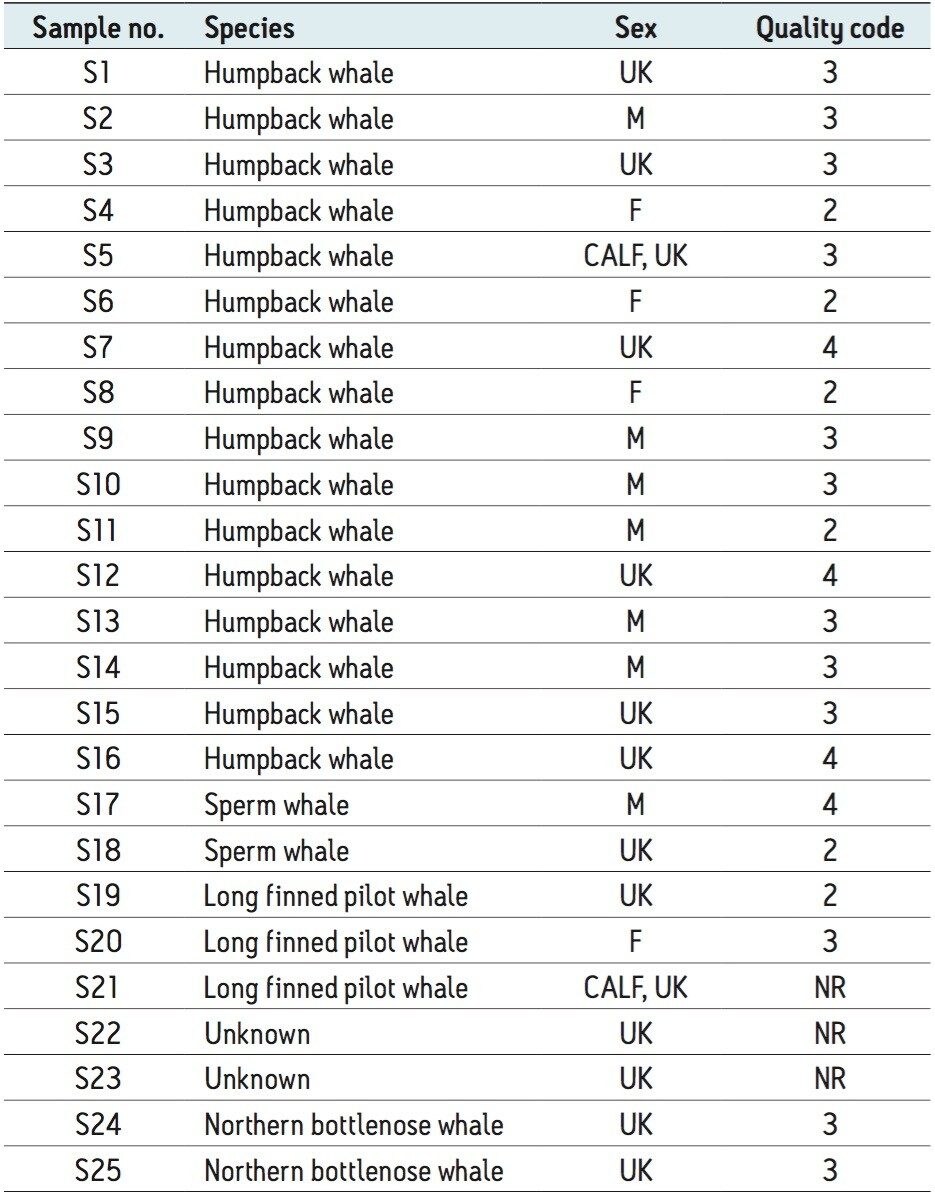
The blow collection, on-board storage, and clean-up method outlined by Hogg et al. (2009)5 was followed in this study. Samples were collected from a variety of mysticete and odontocete whale species from Canada and Norway, including the humpback whale (Megaptera novaeangliae), sperm whale (Physeter macrocephalus), long-finned pilot whale (Globicephala melaena) and northern bottlenose whale (Hyperoodon ampullatus). In some cases the animals were individually identifiable and sex could be determined as animals were recognized by their fluke patterns, known from previous studies (largely humpback whales sampled in the St Lawrence Estuary, Canada). However, in most cases, it was not possible to determine the sex or maturity of the animal.
|
UPLC system: |
ACQUITY UPLC |
|
Column: |
ACQUITY UPLC BEH C8, 2.1 x 50 mm, 1.7 μm |
|
Column temp.: |
40 °C |
|
Flow rate: |
0.6 mL/min |
|
Mobile phase A: |
Water + 0.5% formic acid |
|
Mobile phase B: |
Acetonitrile |
|
Injection volume: |
1 μL, partial loop (pressure assisted) |
|
Time(min) |
%A |
%B |
|---|---|---|
|
0.0 |
90 |
10 |
|
0.5 |
90 |
10 |
|
3.0 |
10 |
90 |
|
4.0 |
10 |
90 |
|
4.1 |
90 |
10 |
|
5.0 |
90 |
10 |
|
MS system: |
Xevo TQ-S |
|
Ionization: |
ESI positive |
|
Capillary voltage: |
3.3 kV |
|
Source offset: |
50 V |
|
Source temperature: |
150 °C |
|
Desolvation gas temp.: |
650 °C |
|
Desolvation gas: |
1000 L/hr |
|
Cone gas flow: |
150 L/hr |
|
Acquisition: |
RADAR [multiple reaction monitoring (MRM) with full scan] |
|
Collision gas: |
Argon at 3.5 x 10-3 mbar |
Whale blow samples were collected using the same method as described in Hogg et al. (2009).5 Briefly, blow samples were collected remotely using inert nylon stockings stretched over a five-inch embroidery ring attached to a pole, as shown in Figure 1.

The collection material was cleaned by sonication for 15 min in 100% acetonitrile, and then for a further 15 min in deionized water, changing the water every 5 min. Whale blow samples were stored on board the sampling vessel and during transportation to the laboratory in a 5-mL inhibitor (100 mM MnCl2/100 μg/mL amoxicillin/potassium clavulanate).
Hormones were extracted using solid phase extraction (SPE) where samples were centrifuged at 3,000 rpm for 15 min to remove the sample and inhibitor from the stocking. Extracts were loaded onto the SPE cartridges that had been preconditioned with 20.0-mL acetonitrile followed by 5.0 mL deionized water. Samples were loaded at 5.0 mL/min and washed with 7.5 mL deionized water to remove salts. Elution of the hormones was then carried out with 5.0 mL 100% acetonitrile, and the eluent was dried under compressed air. Samples were reconstituted in 60 μL 60% acetonitrile in preparation for LC-MS analysis. Since the LC-MS/MS method can be used to investigate the presence of many different proteins simultaneously, both progesterone and cortisol (the latter being a potential stress marker) were analyzed. Standards of progesterone and cortisol from 2 ng/μL to 100 ng/μL in 60% acetonitrile were also included.
Table 1 shows the different species of whales used, along with their sex (if known).
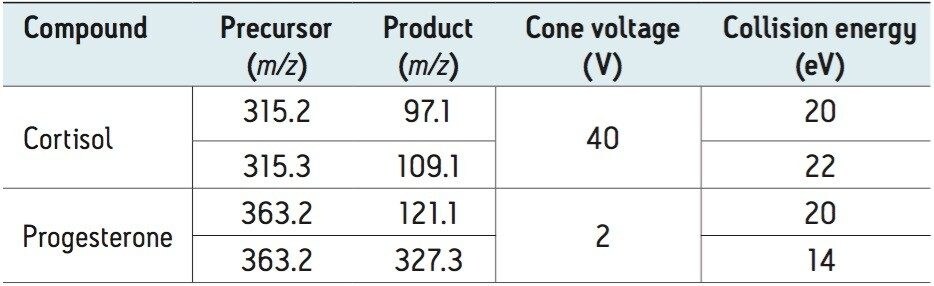
Waters IntelliStart Technology (combining internal calibration fluidics and diagnostics software) integrated into MassLynx 4.1 Software, was used to automatically tune, calibrate, and conduct the systems performance checks before the analytical experiments. IntelliStart was also used to optimize all the MRM transitions for the hormones of interest automatically. For these analyses, twoMRM transitions were used for each compound and the MRMs monitored are summarized in Table 2.
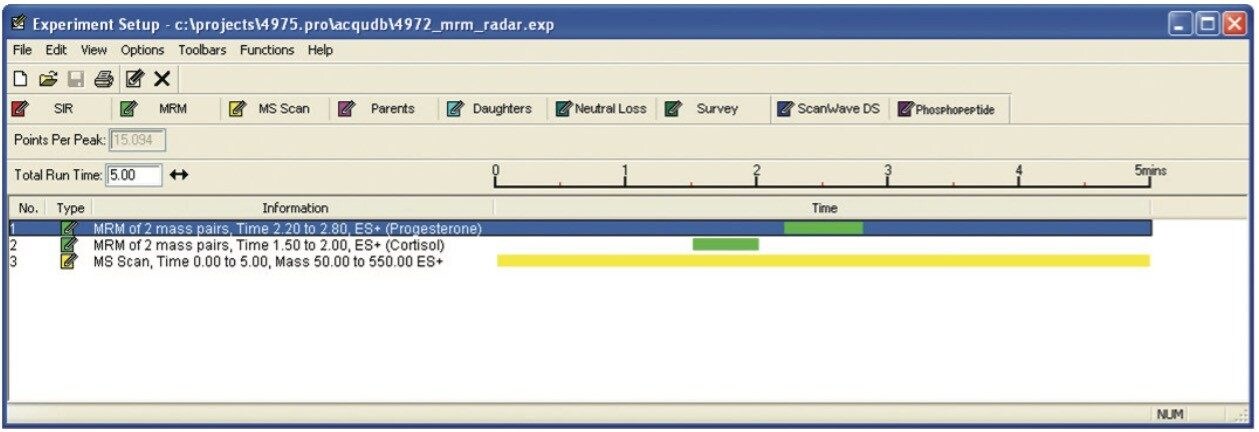
The RADAR Technology of the Xevo TQ-S was also used, which allows both full scan data and MRM data to be acquired in parallel during the same sample analysis. The benefit of this technology is that it enables both targeted quantification and research-based experiments to be performed in the same injection.
The data were processed using the TargetLynx quantification application manager. TargetLynx automates data acquisition, processing, and reporting for quantitative results. It incorporates a range of confirmatory checks to identify samples that fall outside user-specified or regulatory thresholds. For this experiment, the confirmatory checks were not used, as it is the first time these two compounds had been quantified in whale blow samples, hence the thresholds were not yet known.
The sample preparation procedure included the solid phase extraction step, in order to remove any potentially interfering endogenous salts present. (Although as it can be seen later in Figure 7, there was still quite a lot of matrix present in the final sample analyzed).
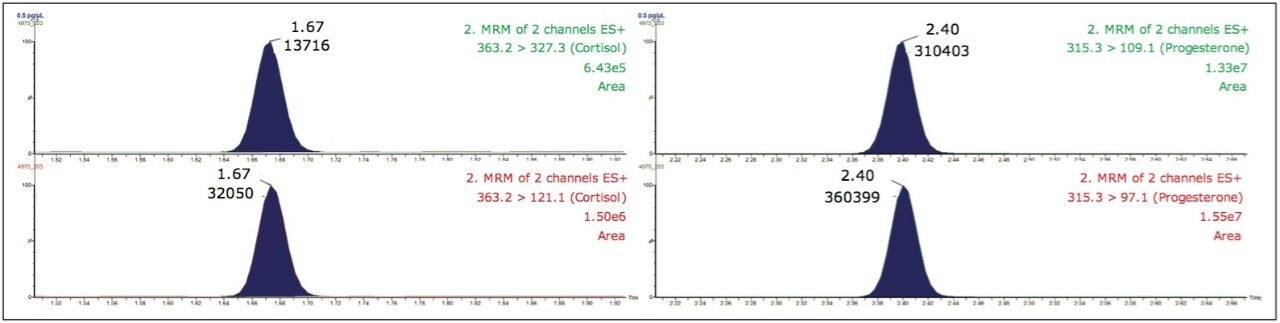
Within the sample analysis a series of calibration standards (between 0.5 and 50.0 pg/μL) were prepared for the hormones, and these were used for quantification. An example chromatogram for the lowest standard injected is shown in Figure 3.
TargetLynx Application Manager was used to produce the calibration curves from these standards and automatically process the sample data. An example of TargetLynx data is shown in Figure 4.
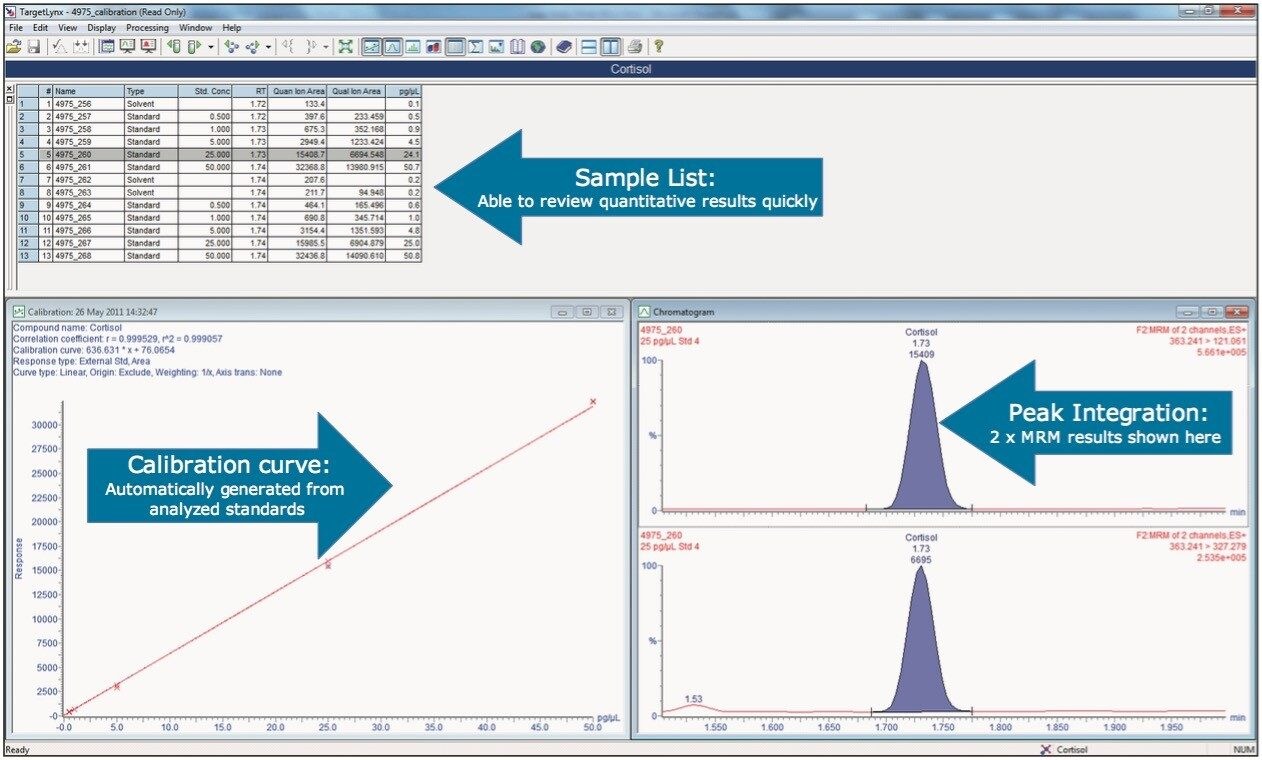
Using the UPLC method conditions, it can be seen that the two compounds of interest were chromatographically well separated from one another. For cortisol and progesterone, the observed retention times were 1.67 and 2.40 min respectively. Both cortisol and progesterone were detected easily in the lowest calibration standard (0.5 pg/μL). Further research around this subject is needed in order to determine the exact levels of detection required, but these preliminary results suggest it is possible to achieve even lower limits of detection with more method development.
For all the standards analyzed, excellent linearity was observed for cortisol and progesterone: the r2 ≥ 0.999 for the two hormones, as shown in Figure 5.

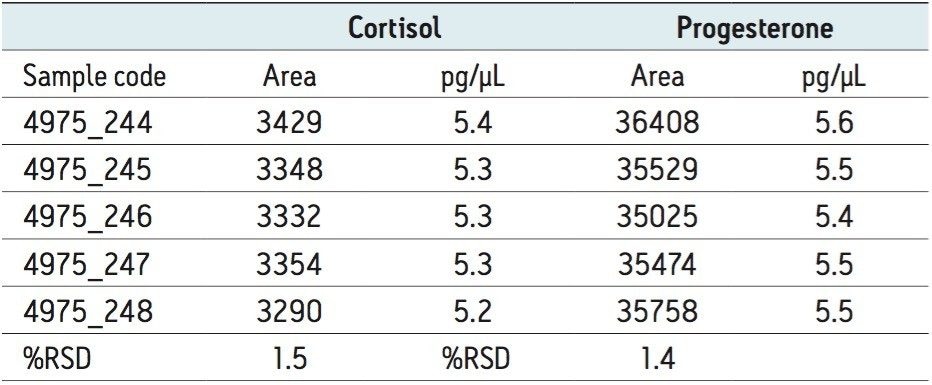
Typical relative standard deviations (%RSDs) for electrospray ionization were observed – this is generally around 5%. For cortisol and progesterone, the data repeatability seen was excellent, with the %RSD being <2%, as shown in Table 3.
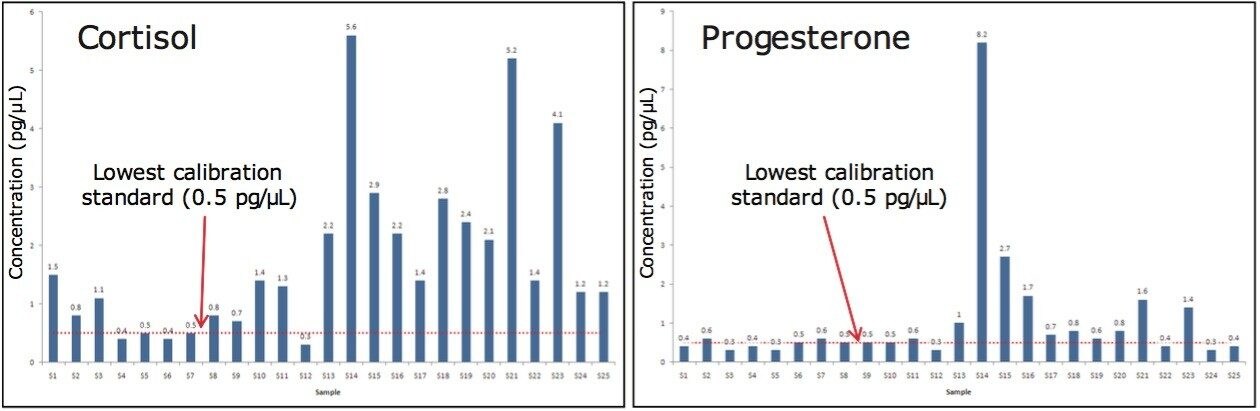
During the experiments, 25 whale blow samples were analyzed and included a variety of whale species and sex. Cortisol was detected in the majority of samples (22 of 25) at a level higher than the lowest calibration point. Progesterone was detected in 13 samples higher than the lowest calibration point. These experiments represent the first time that the levels of the two compounds have been analyzed. The maximum levels observed were 8.2 pg/μL of progesterone in a female humpback species (S14). The highest levels of cortisol (5.6 pg/μL) were also found in the female humpback species (S14).
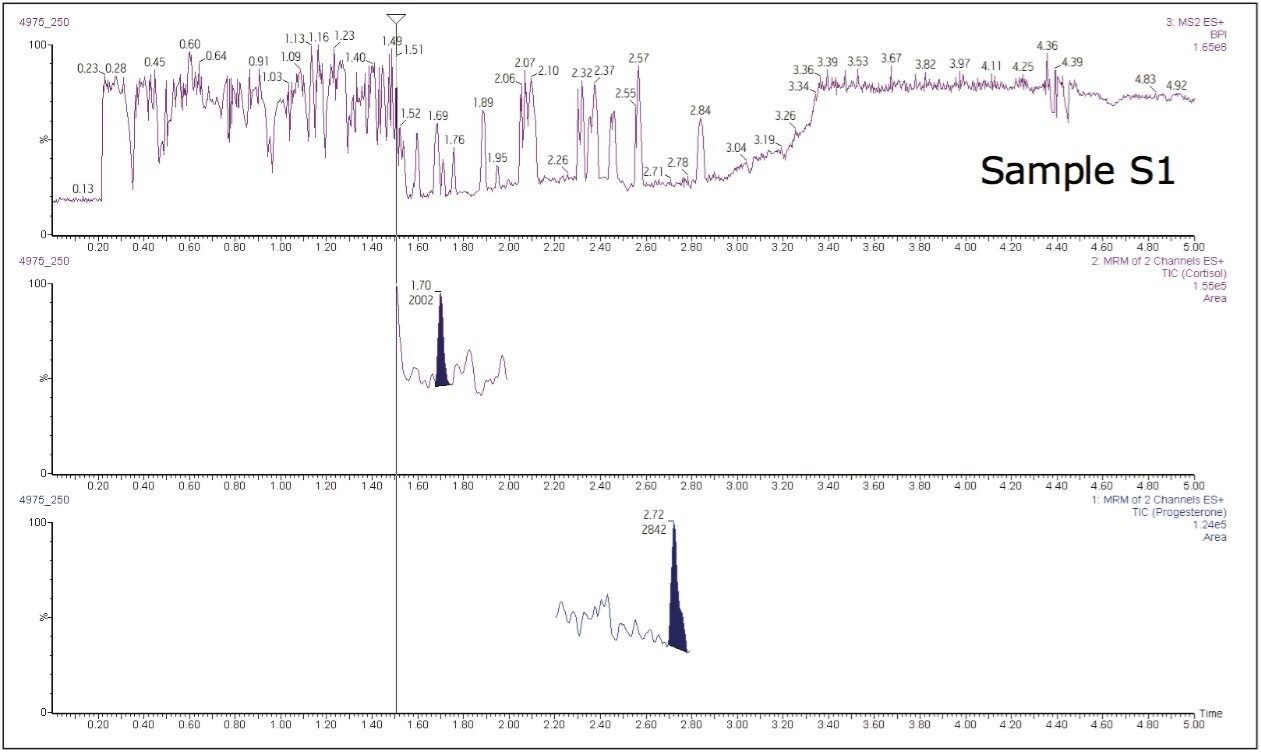
For the samples, the RADAR mode was used during the analysis. RADAR mode is a full scan function used to assess background components during a standard MRM analysis.
The additional functionality of RADAR acquisition was acquired for each of the species types. This allowed us to search for other compounds, while monitoring the matrix background. Figure 7 shows that the matrix background is quite high at the start and at the end of the run, but relatively low at the point where the hormones elute.
720004277, March 2012