
This method allows for the analysis of a pro-drug in human plasma that is highly sensitive and provides carryover less than 20% of the lower limit of quantification.
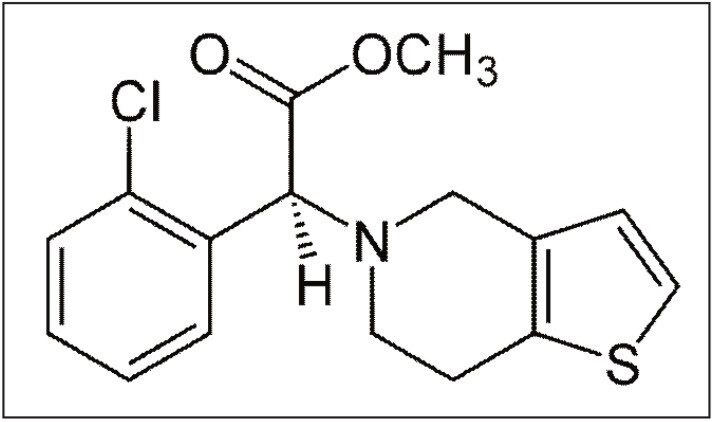
Clopidogrel is a thienopyridine derivative antiplatelet pro-drug used in the prevention of artherosclerotic events. Following oral administration the dosed compound undergoes hepatic metabolism to give rise to the active thiol-metabolite, and the inactive carboxylic acid metabolite. The inactive metabolite accounts for the majority of circulating clopidogrel related material in humans, while the active metabolite and unchanged pro-drug are present at very low levels. The mechanism of action is derived from the binding of the active thiol metabolite to cell receptor P2Y12, irreversibly inhibiting the platelet activation process.1
Due to the reactivity of the active thiol metabolite, coupled with the low levels of the unchanged pro-drug, most quantitative studies are based on the circulating levels of the inactive metabolite. In this application note, we report the development of a highly sensitive solid phase extraction and LC-MS/MS assay for the analysis of the pro-drug clopidogrel in human plasma with an assay sensitivity of 2.5 pg/mL.
The sample was prepared using an Oasis MCX μElution Solid Phase Extraction Plate. 350 µL of plasma sample was mixed with 20 µL of internal standard solution and 350 µL of aqueous buffer. The samples were applied to the solid phase extraction plate, which had previously been primed with methanol and aqueous buffer. The sample was washed with an aqueous – methanol solution and eluted with a 2 x 25 μL of 5% NH4OH in 60:40 IPA:ACN, then further diluted with 25 µL of aqueous buffer prior to injection. The analysis was performed on an ACQUITY UPLC H-Class System. A 10 μL aliquot of the sample was injected onto an ACQUITY UPLC C18 2.1 x 50 mm 1.7 μm Column. The column was operated under gradient conditions over 3 minutes at a flow rate of 600 μL/min. The column effluent was monitored using a Xevo TQD Mass Spectrometer operated in multiple reaction monitoring (MRM) positive ion electrospray mode. The transition 322 ⇒ 212 was employed for the clopidogrel and the transition 326 ⇒ 216 was employed for the d4 internal standard.
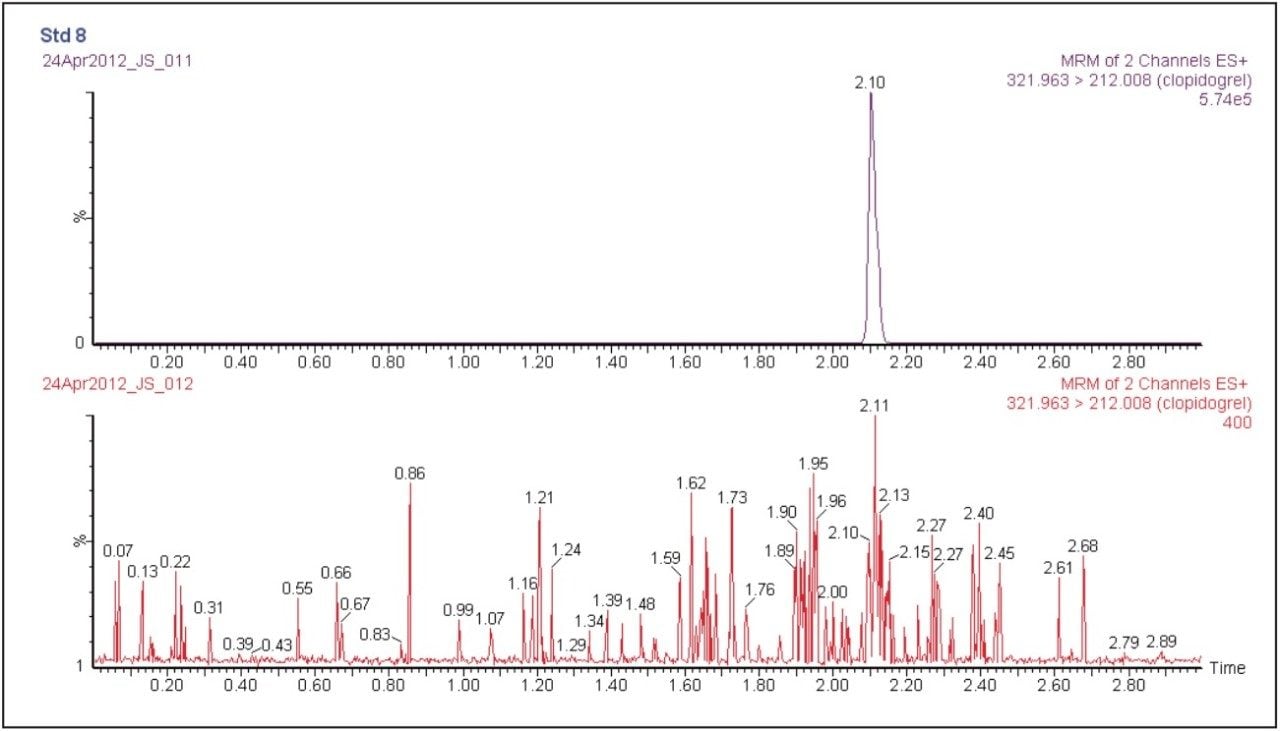
Clopidogrel was eluted with a retention time of 2.10 minutes, as shown in Figure 2. The peak produced by the chromatography system was very symmetrical with a width at the base of 3 seconds. The sharpness of the peak and the symmetrical nature enabled efficient processing and peak integration. The 3 minute analysis time allowed for a total analysis time of five hours for a 96-well SPE plate, allowing two plates to be processed per day. The data displayed in Figure 2 illustrates the injection of an extracted plasma blank injection immediately following the analysis 500 pg/mL standard. We observed from this data that there was no discernable carryover in the blank chromatogram. The extremely low carryover exhibited by the ACQUITY UPLC H-Class System allowed the full sensitivity of the Xevo TQD Mass Spectrometer to be exploited.
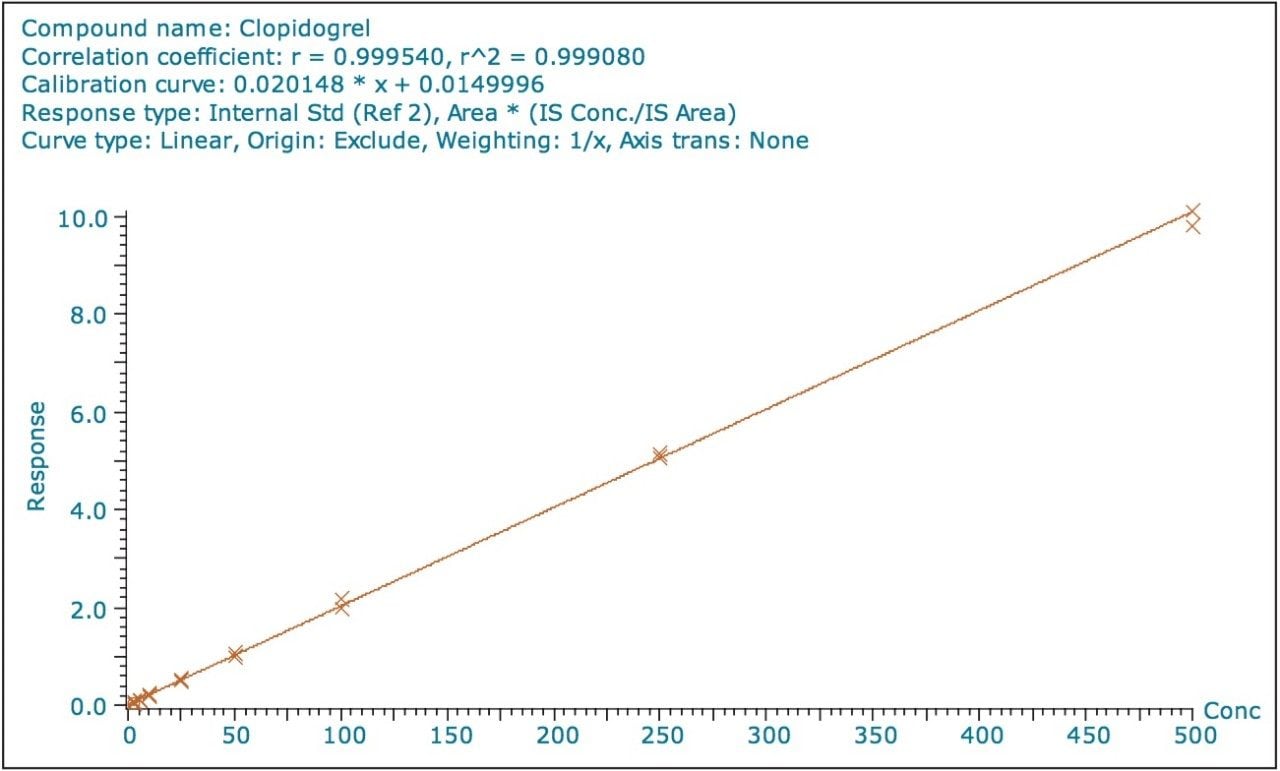
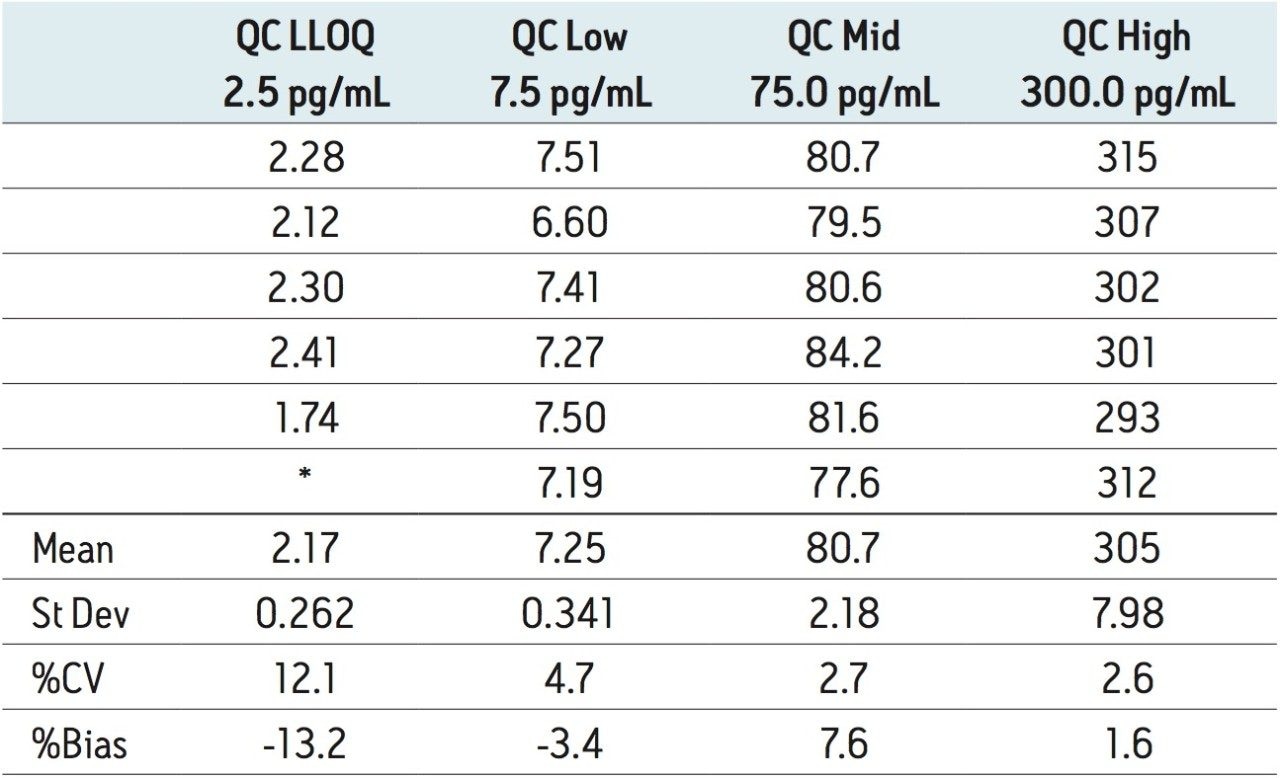
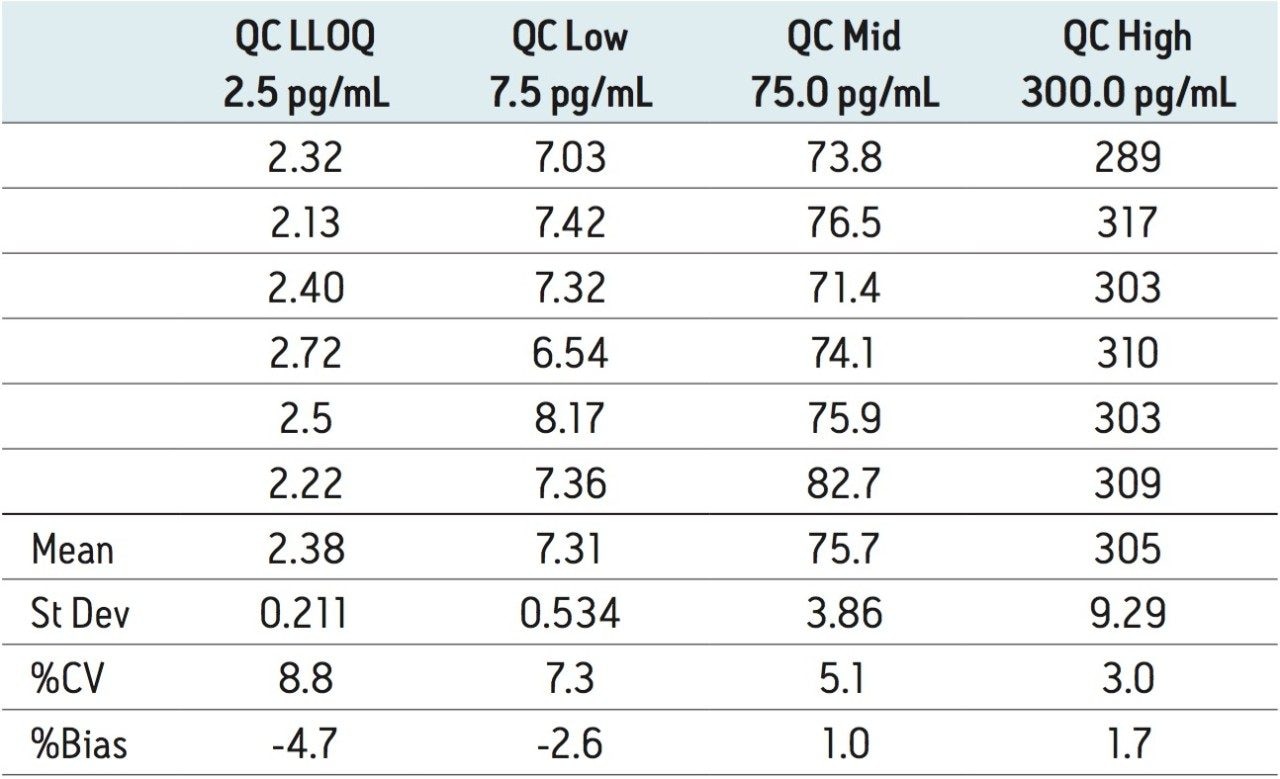
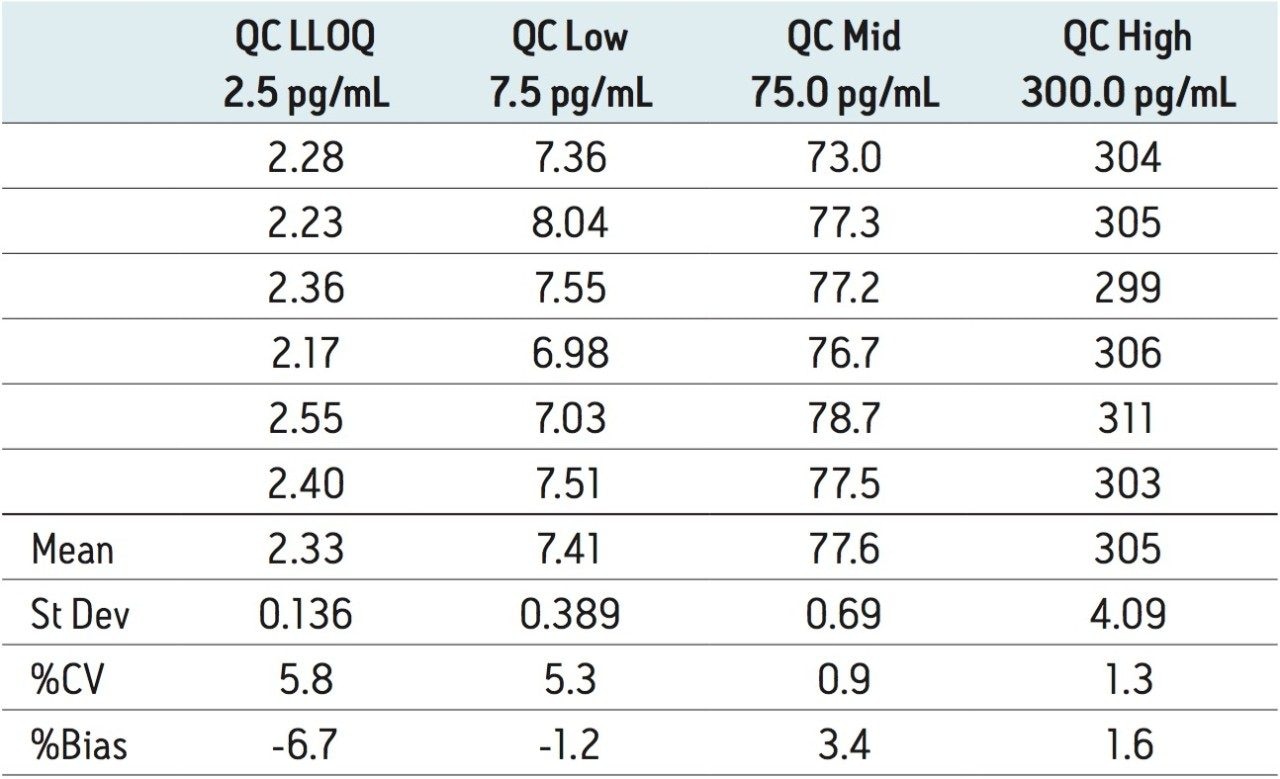
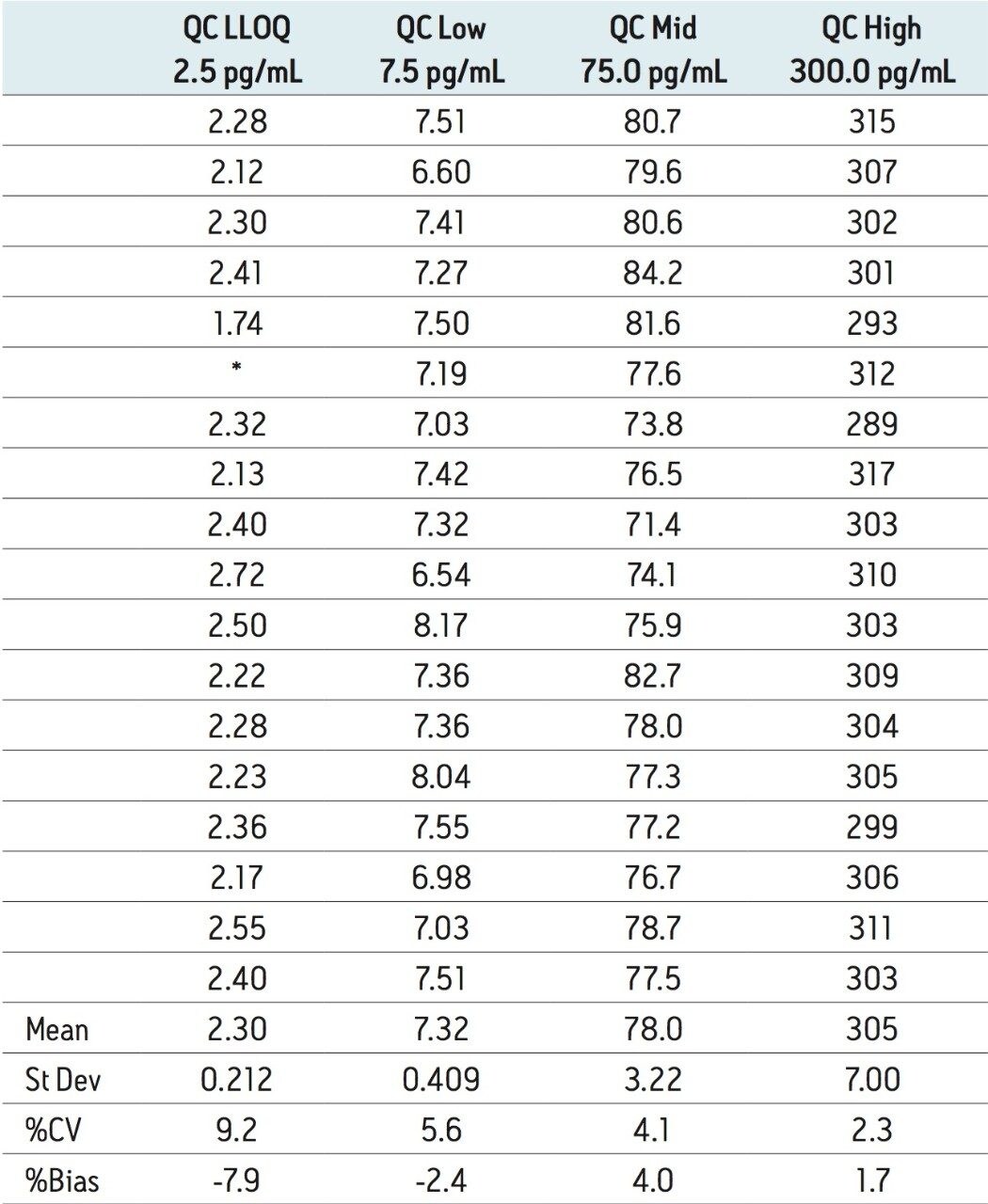
The lower limit of quantification for the assay was determined to be 2.5 pg/mL. The assay was validated with separate 96 well batches on three consecutive days over the range 2.5 to 500.0 pg/mL. A typical calibration obtained for the assay is shown in Figure 3; the correlation coefficient ranged between 0.9990 and 0.9994 using a 1/x weighting linear regression. The intra-day precision and accuracy validation data is shown in Tables 1, 2, and 3. The validation data show that the coefficient of variation ranged from 5.8% to 12.1% for the 2.5 pg/mL LLOQ with a bias between -4.7% and -13.2%. For the high QC (300 pg/mL) the coefficient of variation ranged from 1.3% to 3.0% with a bias between 1.6% and 1.7%. The inter-day precision and accuracy data are displayed in Table 4. The coefficient of variation was determined to be 9.2% for the 2.5 pg/mL LLOQ with a bias of -7.9. For the high QC (300 pg/mL) the coefficient of variation was determined to be 2.3% with a bias of 1.7%.
720004401, June 2012