This application note demonstrates an SFC-MS based quantitation method for R- and S-goitrin with a run time of 6 min and also the capability of SFC-MS in fast qualitative and quantitative analyses of goitrin in a complex TCM formulation.
This application note showcases an optimized SFC system solution for separating enantiomers possessing vastly different bioactivities extracted from Traditional Chinese Medicine (TCM) in six minutes.
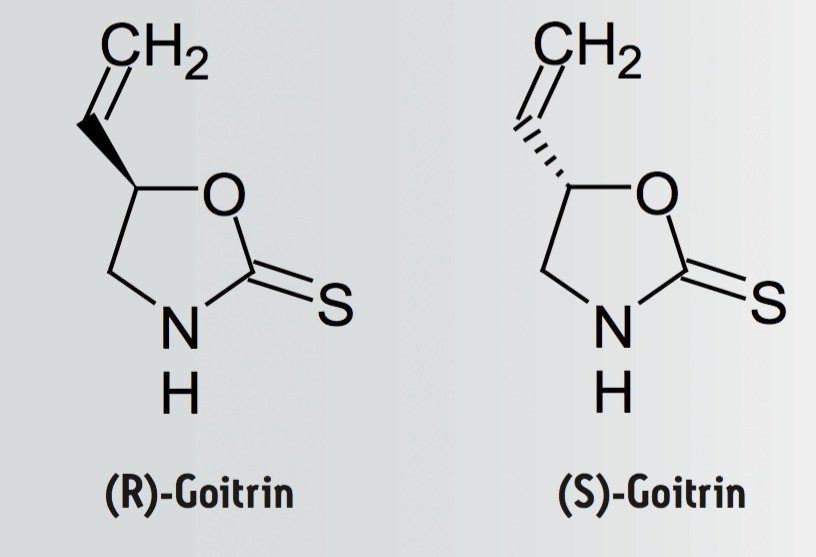
In recent years, much research has been conducted in the chirality of active ingredients in Traditional Chinese Medicine (TCM). For example, the enantiomers (R, S)-goitrin are found in the dried root of Isatis indigotica Fort (known as Bei Ban Lan Gen (板蓝根) in China). Pharmacokinetic studies indicate that only the R-goitrin (epigoitrin) displays the desired anti-viral activity. It is therefore important to separate the R- and S-goitrin to better assess the potency of the Ban Lan Gen on the market, as well as for better quality control in the manufacturing process.
Existing reverse phase liquid chromatography (RPLC)-based methodology does not resolve R- and S-goitrin enantiomers, and therefore cannot accurately quantify the bioactive R-goitrin. Lin et al. have demonstrated a normal-phase liquid chromatography (NPLC)/UV-based chiral separation and quantitation of R- and S-goitrin1. The run time was 50 min.
In this application note we report an SFC-MS based quantitation method for R- and S-goitrin with a run time of 6 min. The optimized method was used to quantify the amounts of R– and S-goitrin in three Ban Lan Gen products manufactured in China, as well as the dried root extract from different geographic region, as shown in Figure 1.

|
Co-solvent: |
methanol |
|
Flow rate: |
3 mL/min |
|
Temp.: |
40 ° C |
|
Backpressure: |
120 bar |
|
Injection volume: |
10 μL |
|
PDA scan range: |
220 to 300 nm. |
The gradient conditions are listed in the respective figure captions.
|
Mode: |
APCI (+) |
|
Corona: |
10 uA |
|
Cone: |
20 V |
|
Source temp.: |
150 °C |
|
Probe temp.: |
400 °C |
|
Desolvation gas: |
400 L/hr |
100 mg of the Ban Lan Gen powder was soaked in 5 mL water, sonicated for 1 hr, and sat for 1 hr. The sample was centrifuged and the supernatant was filtered through a 0.45 μm filter. Next, 5 mL of diethyl ether was added to the filtered extract and the ether portion was collected. This process was repeated three times. The combined diethyl ether extract was dried down and reconstituted in 5-mL methanol.
All experiments were performed on a Waters SFC-MS Resolution System, shown in Figure 2 and controlled by MassLynx Software.

SFC-MS system consists of the following: FDM, CTC autosampler, 10-port column oven, photodiode array (PDA) detector, and a single quadruple mass detector.
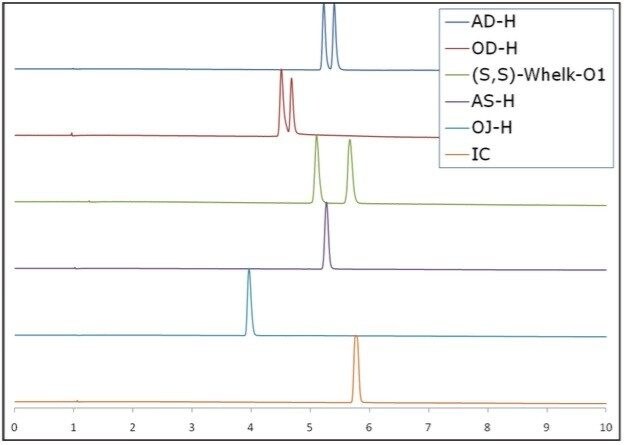
A racemic mixture of (R, S)-goitrin (Purchased from Thermo Fisher Scientific) was used for method development. Six chiral columns (4.6 x 250 mm): AD-H (5 μm), OD-H (5 μm), (S,S)-Whelk-O1 (10 μm), AS-H (5 μm), OJ-H (5 μm), and IC (5 μm) purchased from commercial sources were used for screening.
The initial step was to find optimal chiral separations by screening multiple chiral stationary phases. Figure 3 shows the result of the chiral screening for the (R,S)-goitrin standard on six chiral stationary phases. The AD-H, OD-H, and (S,S)-Whelk-O1 columns were all capable of separating the enantiomers; the (S,S)- Whelk-O1 clearly demonstrated the best resolution.
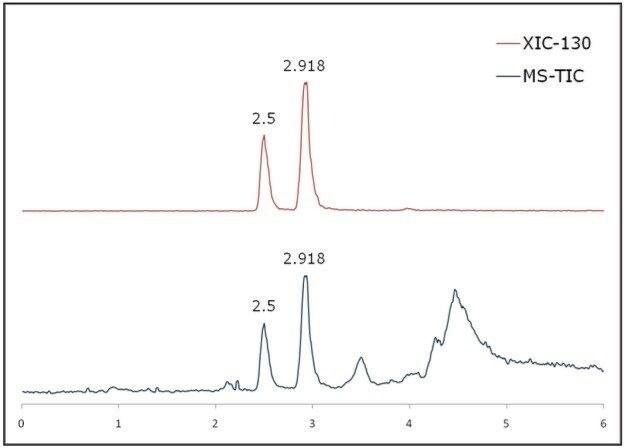
The chromatographic parameters were further optimized to shorten the run time. As shown in Figure 4, the R– and S-goitrin were separated from the extract matrix while preserving the enantiomeric resolution of R– and S-goitrin in 6 min.
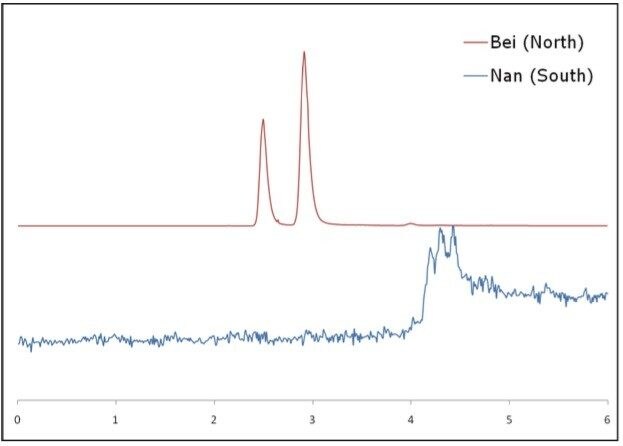
In China, there are two types of herbs, both called Ban Lan Gen. The authentic herb is called Bei Ban Lan Gen, and the second herb, known as Nan Ban Lan Gen, is not considered authentic Ban Lan Gen. This was clearly stated in the 2010 Chinese Pharmacopeia.
Figure 5 shows the SFC-MS chromatograms (SIR, m/z 130) of the Bei Ban lan Gen extract vs. the Nan Ban Lan Gen extract. Here it clearly showed that there is no goitrin detected from the Nan Ban lan Gen extract. This finding is in agreement with previous reports. Clearly, goitrin can be a potential key marker that distinguishes Bei Ban Lan Gen from Nan Ban Lan Gen.
Calibration curves were constructed using the 6-min method, and the results are shown in Figure 6. Excellent linearity was achieved for both R– and S-goitrin. The limit of detection (LOD) and limit of quantification (LOQ) were 2 ng/mL and 10 ng/mL, respectively.
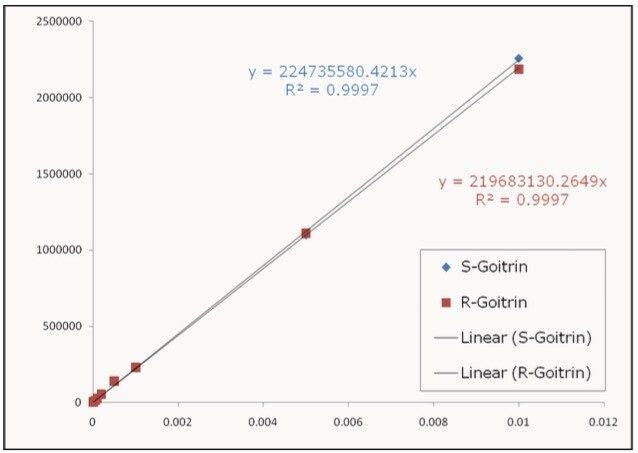
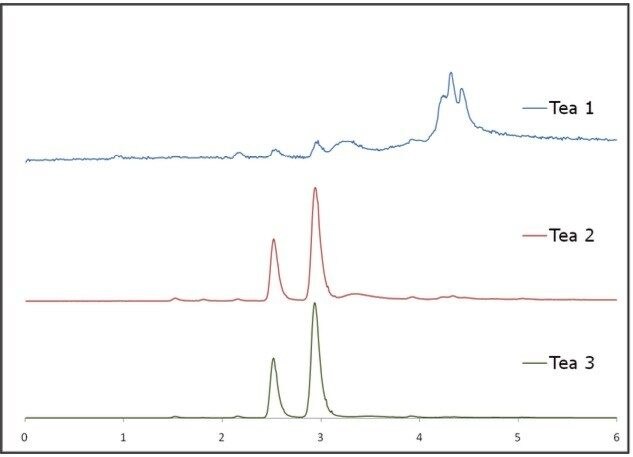
Figure 7 shows the SFC-MS chromatograms of three Ban Lan Gen teas. It is evident that there are substantial differences amongs three Ban Lan Gen products: Tea 1 contains no Goitrin while Tea 3 contains the most Goitrin. The amount of goitrin as well as the ratio of R/S goitrin can be calculated using the calibration curves shown in Figure 6 (results not shown).
The enantiomeric resolution of R- and S-goitrin were achieved in 6 minutes using the SFC-MS Resolution System. The capability of SFC-MS in fast qualitative and quantitative analyses of goitrin in a complex TCM formulation has been demonstrated. SFC is the technique of choice for chiral separation and it holds great promise for replacing NPLC. SFC should be considered a valuable addition to the overall analytical technology platform for TCM research.
720004049, July 2011