This is an Application Brief and does not contain a detailed Experimental section.
This application brief provides a direct analysis tool for mass spectrometric analysis of synthetic polymers and blends with limited method development or sample preparation.
ASAP IMS-MS shows excellent potential for the rapid fingerprinting of complex polymeric samples.
Analysis of specialty polymers and surfactants is often limited to size-based analysis, such as Size Exclusion Chromatography (SEC) with an appropriate detection mode. When employing this technique, a proper test method must be established, including a calibration set that takes into account detector bias and chromatographic stability. Further, as product space expands to include multi-functional materials, this analytical approach has been found to be limited when addressing compositional variations in the material.
ASAP (Atmospheric Solids Analysis Probe) developed by McEwen et. al.1 has been shown to be a useful tool for the rapid direct analysis of volatile and semi-volatile solid and liquid samples, such as synthetic polymers and oligomers2. The ability of Ion Mobility Spectrometry (IMS) to separate ions based on their collision cross sectional area and charge state provides a powerful orthogonal separation technique, when coupled with mass spectrometry for the analysis of complex mixtures.
All analyses were performed using a Waters SYNAPT G2 HDMS System. An ASAP device was used in place of the instrument’s Electrospray probe, as shown in Figure 1. The source was operated in ESCi mode to facilitate the use of the Electrospray desolvation heater in conjunction with a corona discharge. This configuration also allowed the LockSpray interface to be used for exact mass measurements.

Samples were introduced on a sealed glass melting point tube and vaporized in a stream of heated nitrogen. The temperature of the nitrogen was ramped to control the vaporization of components in the complex mixtures. The sample in the gas phase was ionized by proximity to a corona discharge needle. Ions then passed from the atmospheric pressure region into the mass spectrometer.
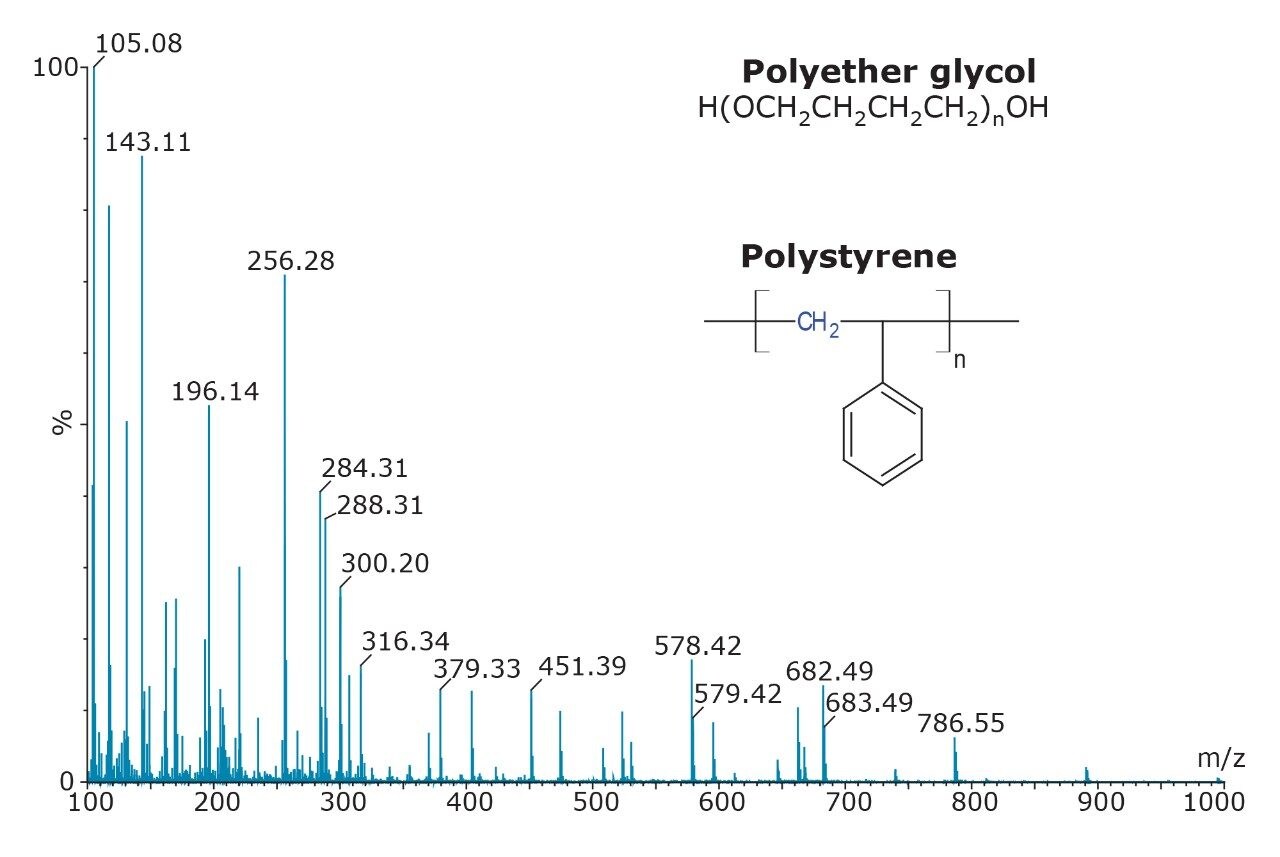
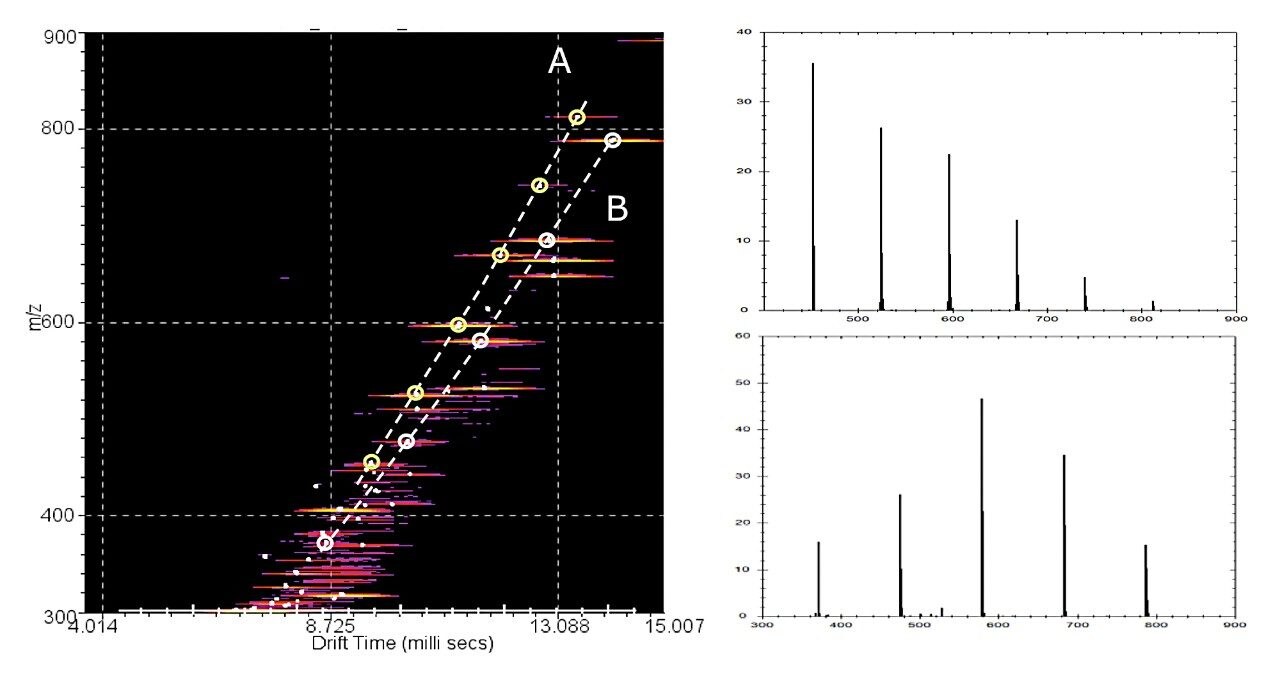
The polymer mixture was analyzed by ASAP on a SYNAPT G2 HDMS System, shown in Figure 2, and the IMS-MS data were post-processed using a 3-dimensional peak detection algorithm ‘APEX 3D’ to determine m/z, drift time (DT), and intensity, as shown in Figure 3. Ion mobility separated spectra of the polyether glycol and polystyrene were readily extracted using this software. This approach has potential for wider application in the rapid characterization of polymeric mixtures.
720004024, September 2011