This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates the capabilities of Waters’ integrated UPLC-SEC/UV/MS system for fast and routine characterization of reduced monoclonal antibodies.
This on-line UPLC-SEC/MS method, accomplished by utilizing a sub-2-μm ACQUITY UPLC BEH SEC Column, the ACQUITY UPLC System, and the SYNAPT G2, offers a powerful solution for antibody characterization.
The pipeline for biotherapeutics is growing rapidly as pharmaceutical organizations shift their focus from small molecule drugs to biotherapeutic drugs. The efficient characterization of antibody drugs is increasingly important to both regulatory agencies and pharmaceutical companies to ensure the safety and efficacy of biotherapeutic products.
Although the selectivity of antibodies varies appreciably, the overall structures of antibodies are highly conserved within an antibody class. The ability to analyze the same class of antibodies using a generic method is highly desirable for improving the efficiency of analyses in the pharmaceutical industry.
Reversed-phase (RP) chromatography coupled with mass spectrometry is frequently used for the analysis of reduced antibodies. RP chromatography separates proteins based on their hydrophobicity and is a popular technique for rapid, intact mass analysis. Nonetheless, proteins with different sizes may show similar hydrophobicity and are therefore difficult to separate by RP. High temperature is often used in RP chromatography to achieve better peak shape and to minimize carryover. This, however, raises questions on whether high temperature introduces changes to the protein structure.
Size exclusion chromatography (SEC) separates proteins predominantly based on their size differences using an isocratic elution rather than hydrophobicity and avoids the use of high column temperature and gradient optimization. SEC is widely used in the biopharmaceutical industry to detect antibody aggregates and fragments.
However, traditional SEC mobile phases are incompatible with electrospray ionization MS, and historically optical detection methods with lower specificity have been used. Lower specificity presents a problem for organizations wishing to characterize biotherapeutics according to the well-characterized biotherapeutic product model as adopted by regulators around the world.
This work describes an efficient on-line UPLC-SEC/ MS method for the direct mass analysis of reduced humanized monoclonal antibodies (mAb), and demonstrates that rapid, high-resolution SEC separations in combination with high-performance SYNAPT G2 MS provides an efficient, generic method for routine antibody characterization.
The successful coupling of SEC to ESI-MS was achieved using an MS-friendly mobile phase. The SEC separation was achieved using an ACQUITY UPLC BEH200 SEC Column (1.7-μm, 4.6 x 300 mm) with an ACQUITY UPLC System using isocratic elution with a flow rate of 0.46 mL/min. The mobile phase contains 30% acetonitrile, 0.1% TFA, and 0.1% FA in Milli-Q water. The flow passed through a TUV detector operated at 280 nm wavelength and then directed to the SYNAPT G2 MS.
The use of sub-2-μm UPLC BEH column packing materials along with the low-dispersion, highpressure ACQUITY UPLC System resulted in significant improvements in size-based separation as displayed in Figure 1. The 10-minute SEC run completely resolved the earlier eluting glycosylated heavy chain (HC) from the later eluting light chain (LC), a separation not achievable in such a limited timeframe with existing techniques.
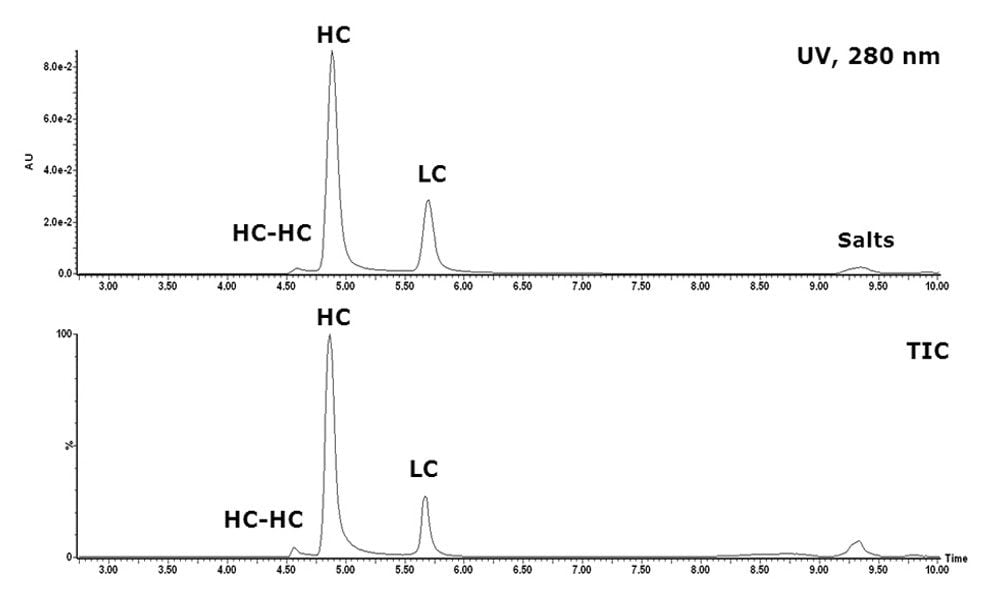
In addition, the glycosylated HC-HC fragment/clip was well separated from the heavy chain. In contrast to RP columns, the Waters UPLC BEH SEC column showed no memory effect. Consequently, no blank runs were needed between sample runs.
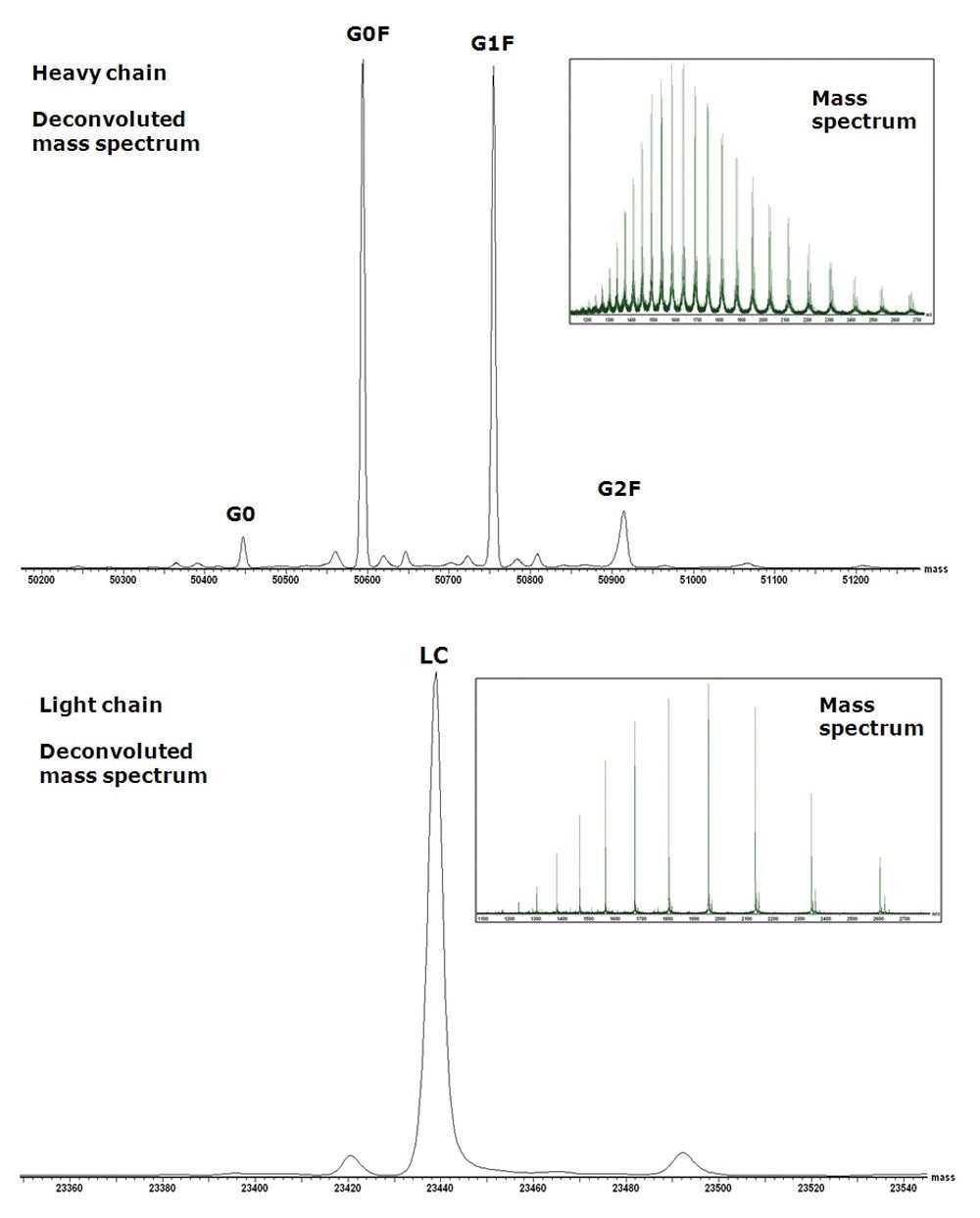
Figure 2 (top) depicts the summed raw mass spectrum (inset) and the resulting MaxEnt1 deconvoluted mass spectrum of the glycosylated heavy chain. The major peaks correspond to the heavy chain containing the core fucosylated glycan (G0F),a minor nonfucosylated form (G0), and core glycan variants extending by one or two terminal galactose residues (G1F, G2F). Below, it also shows the summed raw mass spectrum (inset) and the MaxEnt1 deconvoluted spectrum of the light chain.
The optimized UPLC-SEC/UV/MS method enables the direct pairing of size exclusion chromatography with a mass spectrometer to measure the molecular weights of reduced mAbs. This well-established separation technique is now linked to mass spectrometric detection for those needing to characterize their biotherapeutics and satisfy regulators that they have sufficiently understood their biotherapeutic products.
The performance of the UPLC system and SEC column allows scientists to resolve chain, heavy chain, and heavy chain-heavy chain clip of an antibody without employing high column temperature. This optimized SEC method with separation based on size differences provides a complement to RP chromatography, and, coupled with MS, offers a powerful, routine, and generic solution for antibody characterization.
720004018, July 2011