For research use only. Not for use in diagnostic procedures.
This application note describes a new, sensitive UPLC-MS approach developed to measure bile acid reproducibly and reliably in biological fluids. Over 30 individual bile acids were separated and detected in a 5-minute window using an ACQUITY UPLC HSS T3 2.1 x 100 mm, 1.8 μm Column coupled to a Q-ToF Premier Mass Spectrometer.
Individual bile acids are endogenous markers of liver cell function and studies of both qualitative and quantitative bile acid changes have been conducted as a result of liver and intestinal diseases. The measurement of serum bile acid concentrations can provide information pertaining to liver damage, as well as hepatic and biliary tract diseases.1-4 However, traditional chromatographic methods have not typically provided sufficient separation in order to differentiate between structurally similar bile acids. Utilization of the Waters UltraPerformance LC (UPLC) Technology high resolution chromatographic system has greatly improved the abilility to separate metabolites from endogenous matrice. UPLC provides superior resolution, sensitivity, and throughput compared with conventional LC approaches. Using UPLC, previously co-eluting metabolites can be separated and matrix effects, such as ESI ion suppression, are minimized. By combining UPLC with oa-TOF mass spectrometry, both high-resolution and exact mass measurements can be achieved, aiding the identification of metabolites. This application note describes a new, sensitive UPLC-MS approach developed to measure bile acid reproducibly and reliably in biological fluids. Over 30 individual bile acids were separated and detected in a 5-minute window using an ACQUITY UPLC HSS T3 2.1 x 100 mm, 1.8 μm Column coupled to a Q-ToF Premier Mass Spectrometer. Bile acids were extracted from serum using methanol and a gradient elution of water and acetonitrile was employed, which also enabled the detection of a wide range of endogenous metabolites, such as lipids. MSE data were acquired using a patented acquisition method that collects precursor and product ion information for virtually every detectable species in a mixture. This allowed for characteristic metabolite fragmentation information to be obtained in a single analytical run, easily distinguishing glycine and taurine bile acid conjugates. This assay was applied to the study of the hepatotoxin galactosamine (gaIN) in rat. Serum bile acid changes were observed after gaIN treatment, including elevated taurine-conjugated bile acids, which correlated to liver damage severity. This UPLC-MS approach to bile acid analysis offers a sensitive and reproducible tool that will be of great value in exploring both markers and mechanisms of hepatotoxicity.
|
LC System: |
ACQUITY UPLC |
|
Column: |
ACQUITY UPLC HSS T3, 2.1 x 100 mm, 1.8 μm |
|
Column temp.: |
40 °C |
|
Sample temp.: |
4 °C |
|
Mobile phase A: |
Water |
|
Mobile phase B: |
Acetonitrile |
|
Flow rate: |
0.5 mL/min |
|
Injection volume: |
5 μl |
|
Data processing: |
MarkerLynx XS Application Manager |
|
Time (min) |
%A |
Curve |
|---|---|---|
|
0.0 |
0 |
0 |
|
2.0 |
0 |
6 |
|
12.0 |
5 |
6 |
|
17.0 |
5 |
6 |
|
18.0 |
100 |
6 |
|
22.0 |
100 |
6 |
|
22.5 |
5 |
6 |
|
23.0 |
0 |
6 |
|
26.0 |
0 |
6 |
|
MS system: |
Q-ToF Premier Negative electrospray mode |
|
Scan range: |
50 to 1000 Da |
|
Capillary voltage: |
2.4 Kv |
|
Source temp.: |
120 °C |
|
Desolvation temp.: |
350 °C |
|
Cone voltage: |
35 V |
|
Desolvation gas flow: |
900 L/hr |
|
Collision energy (CE): |
Low CE: 5 eV High CE: ramp of 10 to 70 eV |
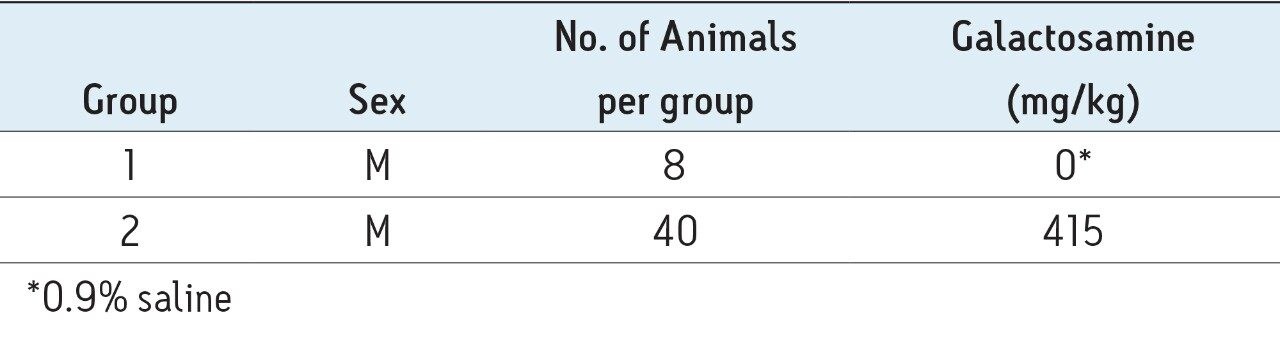
Table 1 details the dosing of 40, six-week old male Sprague Dawley rats. The animals were euthanized 24 hrs after galactosamine (gaIN) or vehicle administration. Serum was isolated from blood samples collected at necropsy from the abdominal vena cava and stored at -40 °C pending analysis.
Clinical Chemistry Analysis. Serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), and total bilirubin levels were analyzed using a Vitros 950 analyzer (Ortho-Clinical Diagnostics, Rochester, NY).
Histological Analysis. Liver samples were fixed in 10% buffered formalin, embedded in paraffin, sectioned, and stained with hematoxylin and eosin. Liver sections were assigned the following histoscores: 0) absence of hepatocellular necrosis, 1) minimal necrosis, 2) mild necrosis, 3) moderate necrosis, and 4) marked necrosis.
Ice-cold methanol (150 μL) was added to 50 μL serum and vortexed for 30 seconds. The samples were then incubated at -20 °C for 20 mins, centrifuged at 13,000 rpm for 10 min, and the supernatant was removed to a clean tube. The supernatant was then dried down in a vacuum evaporator (Savant), reconstituted in 100 μL water, and transferred into 96-well 350 μL plates.
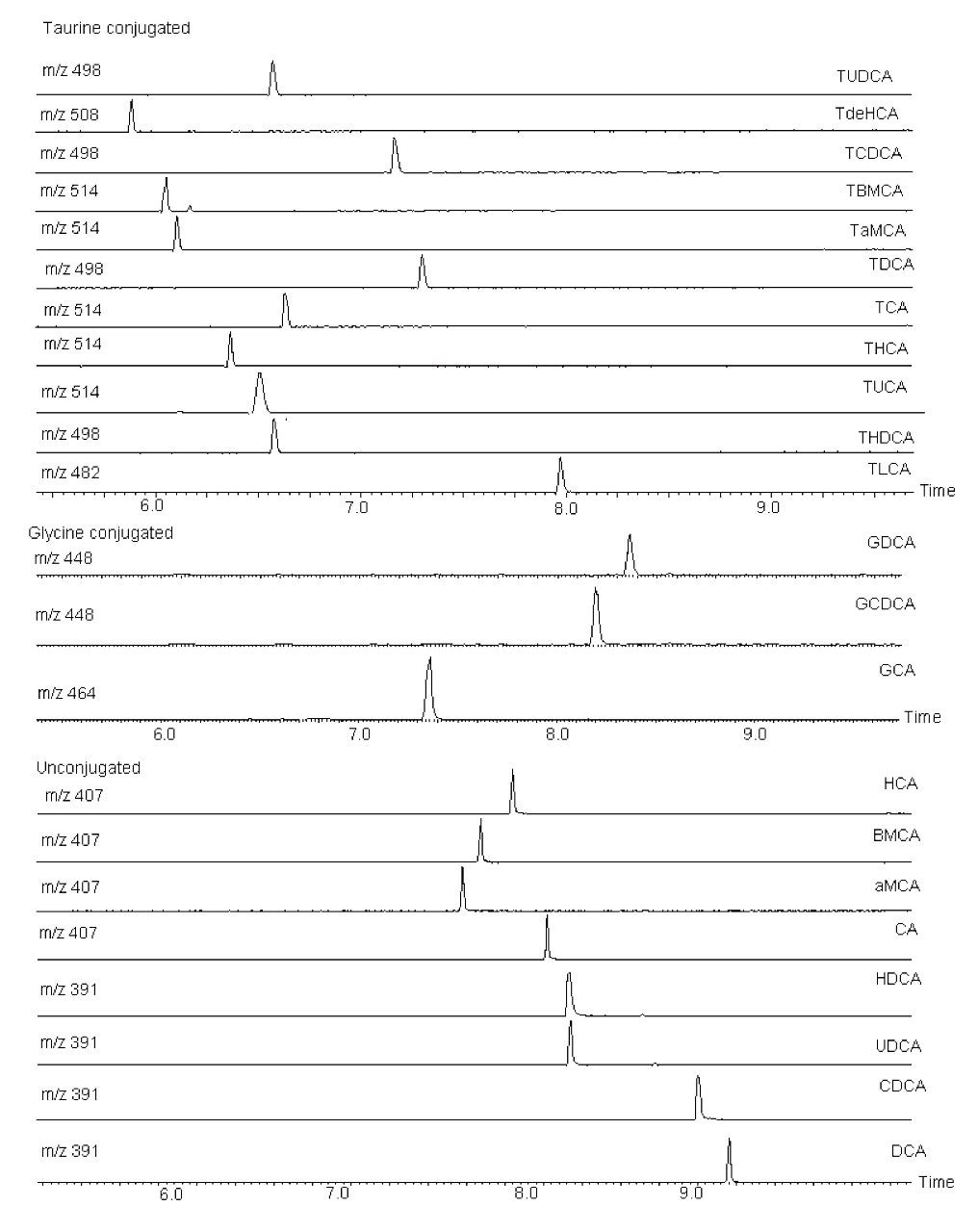
The UPLC-MS assay developed allowed for the reproducible separation and detection of 24 fully identified bile acids, plus 10 tentatively identified bile acids. All bile acids eluted within a 4 min window (6 to 10 mins) with good separation of individual bile acids, as shown in Figure 1. This method offers shorter analysis time than conventional HPLC methods (data not shown), and therefore allows significantly increased sample throughput.
Bile acids ionize strongly using negative mode ESI, producing a prominent precursor [M-H]- ion in the low energy data and informative fragmentation data in the high energy data. This allows conjugates to be easily distinguished by fragment ion analysis. Glycine conjugates give rise to a diagnostic ion at 74 m/z and taurine conjugates at 79.9, 106.0, and 124.0 m/z respectively, as shown in Figure 2. This information was obtained using MSE, facilitating the determination of the different conjugation classes of bile acids in a single run.
Importantly, in addition to bile acids, this assay allows for the detection of a wide range of endogenous metabolites, providing additional, complementary metabolite information. This is of great utility in metabonomics studies, as sample numbers may be large and it is desirable to obtain maximum information in a single analytical run. This provides reductions in sample volume, throughput time, and solvent consumption.
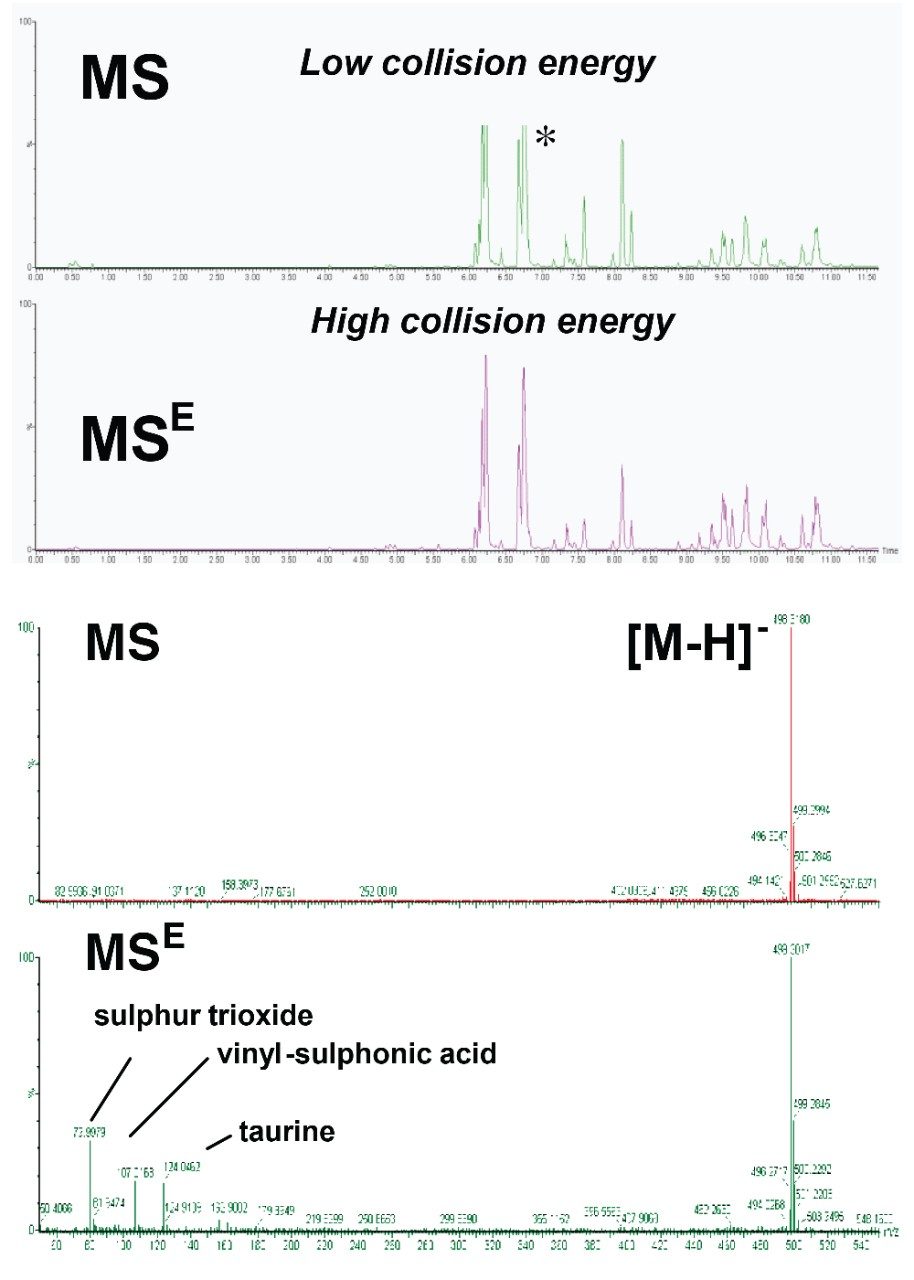
Figure 3 shows representative base peak intensity (BPI) UPLC-MS chromatograms of serum from a control animal and those treated with galactosamine, demonstrating varying degrees of severity of liver damage. Dramatic serum BA changes were visible after galactosamine treatment, as shown in Figure 3, with obvious increases in the taurine-conjugated BAs. These increases correlated with the extent of liver damage (as determined by histoscore) and also the clinical chemistry (AST and ALT).
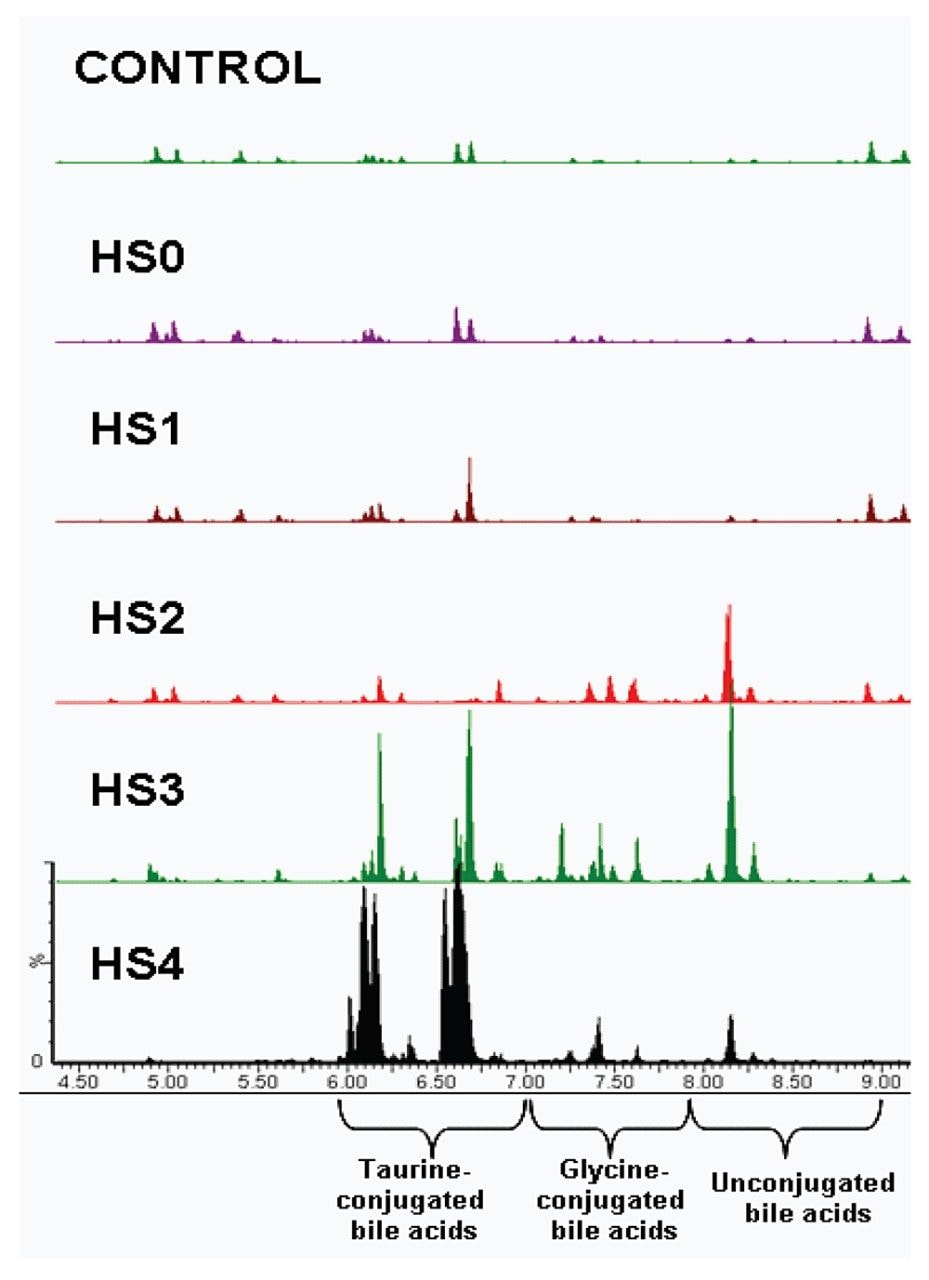
UPLC-MSE offers a reliable and reproducible approach for the analysis of bile acids in biological fluids. This was demonstrated with serum samples applied to a toxicity study, and could easily be extended to other sample types and studies. The employment of a 26-min gradient allowed for the analysis of a wide range of endogenous metabolites, providing both a targeted assay and a global metabolite profiling approach in the same analytical run. This approach enhances the information obtained, leading to a more comprehensive interpretation of metabolite changes.
720004007, June 2011