This application note describes the isolation of the isoflavone puerarin from the roots of the kudzu plant using a modular preparative HPLC system.
A modular preparative HPLC system provides flexibility without compromising performance for isoflavone isolation. Empower 2 Software offers total system control including fraction collection.
Kudzu (Pueraria lobata) is a climbing, woody, or semi-woody perennial vine with a tuberous root. The roots of kudzu contain a number of potentially useful isoflavones, including daidzein, daidzin, genistein, genistin, and quercetin. Kudzu is also a unique source of the isoflavone puerarin as shown in Figure 1. Consumption of kudzu root extracts is thought to reduce alcohol intake and reduce alcohol withdrawal symptoms. Antibacterial, anti-cancer, anti-inflammatory, and antioxidant effects have also been noted.1
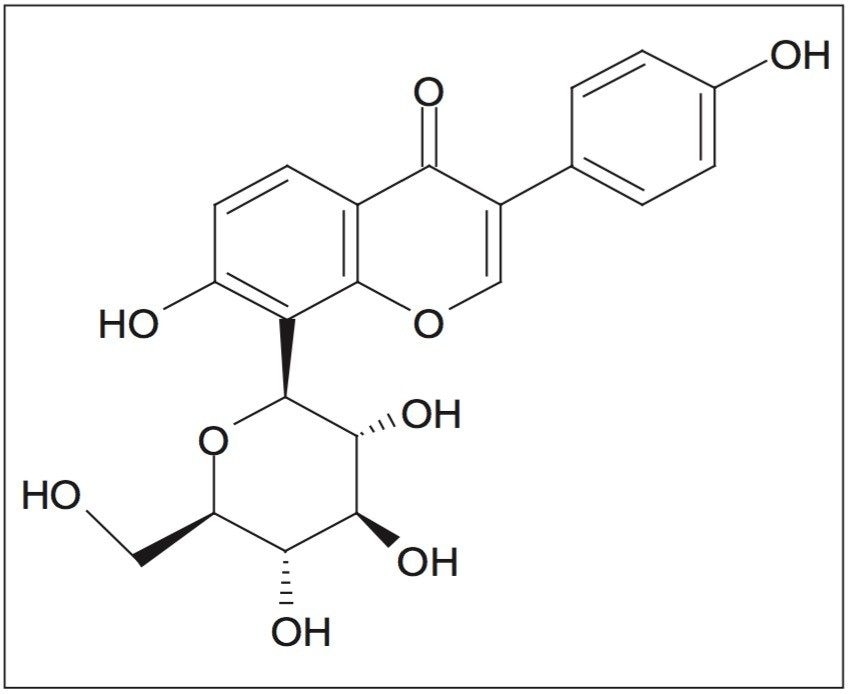
Generally, the goal in natural product purification is to isolate individual component compounds that may have biological activity. Sufficient amounts of these potentially active compounds need to be isolated in order to facilitate characterization of unknown compounds or for use in other studies, such as clinical trials and bioassays. The isolated compounds must be of high purity so that any results obtained can be attributed to the compound under study rather than small amounts of companion compounds. Many techniques for extraction, isolation, and purification of natural products have been previously described.2
Preparative reversed-phase high performance liquid chromatography (RP-HPLC) is a separation technique that is widely used in this endeavor. It is considered a rapid, reliable, and robust technique that has wide applicability over many classes of compounds. This application note describes the isolation of the isoflavone puerarin from the roots of the kudzu plant using a modular preparative HPLC system.
Kudzu root powder (50 g) was added to 250 mL of 9:1 water/methanol and shaken for one hour, allowed to stand overnight, and shaken for an additional hour. This extract was centrifuged at 3000 RPM for 20 minutes, passed through Whatman #1 filter paper, and used without further treatment.
Preparative chromatographic separations were carried out using a Waters Modular HPLC System as shown in Figure 2, which consisted of the following components:
|
Pump: |
2535 Quaternary Gradient Module |
|
Detector: |
2489 UV/Visible Detector |
|
Injector: |
2707 Autosampler configured with a 1-mL loop |
|
Collector: |
Fraction Collector III |
|
Columns: |
Initial Prep Trial – SunFire C18, 5 μm, 10 x 100 mm Final Prep Method – SunFire C18, 5 μm, 19 x 100 mm Column |

Analytical chromatographic separations, implemented for method development and final purity checks, were carried out using the Alliance HPLC 2695 and a 2998 PDA Detector with a SunFire C18, 5 μm, 4.6 x 100 mm Column. Both the analytical and prep systems, including fraction collection, were controlled using Empower 2 Software to collect, manage, process, and report chromatography data. Two initial analytical scale separations were developed (one gradient for purity checks and one isocratic for purification) with the conditions described on the next page.
|
Column temp.: |
Ambient |
|
Flow rate: |
1.5 mL/min |
|
Mobile phase A: |
Water + 0.1% formic acid |
|
Mobile phase B: |
Acetonitrile |
|
Gradient: |
95% A:5% B to 20:80 over 20 minutes |
|
Detection: |
UV at 251 nm |
|
Column temp.: |
Ambient |
|
Flow rate: |
1.5 mL/min |
|
Mobile phase A: |
88.5% Water + 0.1% formic acid |
|
Mobile phase B: |
11.5% Acetonitrile |
|
Detection: |
UV at 251 nm |
Preparative separations were geometrically scaled from the analytical methods and are described below.
|
Column temp.: |
Ambient |
|
Flow rate: |
7 or 25 mL/min (for 10 and 19 mm I.D. respectively) |
|
Mobile phase A: |
88.5% Water + 0.1% formic acid |
|
Mobile phase B: |
11.5% Acetonitrile |
|
Injection vol.: |
500 μL using Autosampler with 10-mm column or 2000 μL using Line D of the 2535 with 19-mm column |
|
Detection: |
UV at 251 nm |
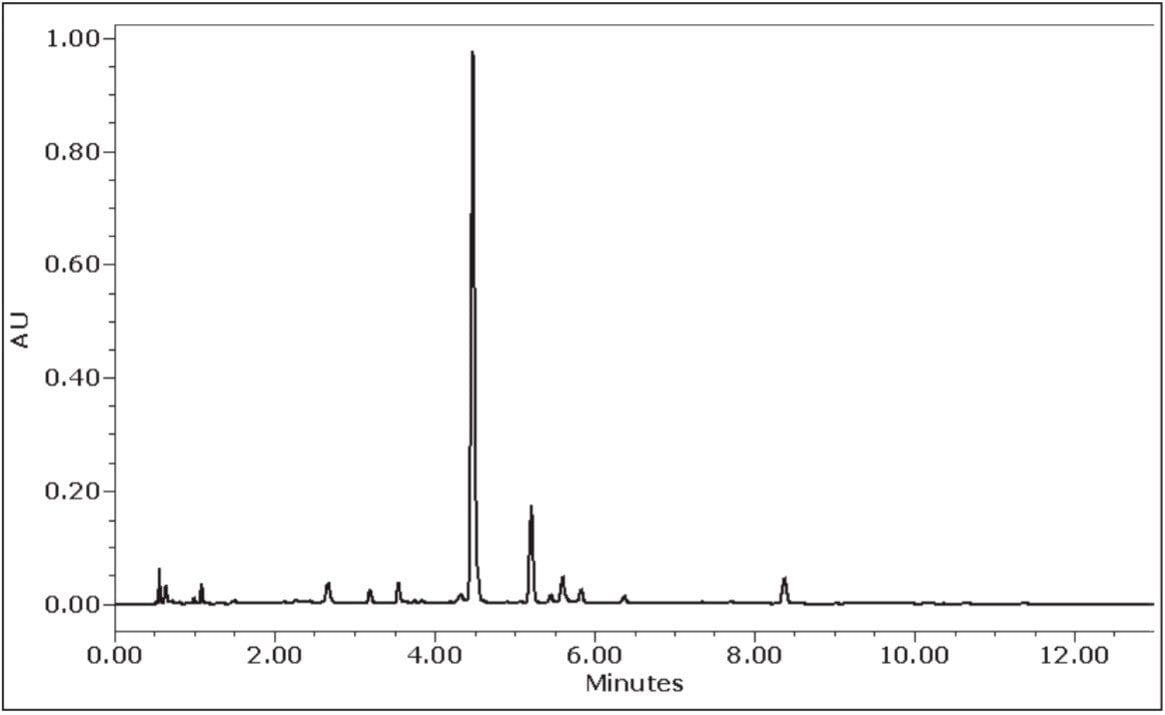
Gradient HPLC analysis of the prepared extract indicated a puerarin concentration of ~0.5 mg/mL and an overall purity of 59.6% by UV area percent as shown in Figure 3. Using a previously developed analytical isocratic HPLC method (data not shown), the separation was scaled to the 10-mm I.D. prep column using the Preparative OBD Column Calculator shown in Figure 4. The Preparative OBD Column Calculator, a free download, provides an easy-to-use tool that aids in analytical-to-preparative scaling calculations (www.waters.com/prepcalculator). The Preparative OBD Column Calculator was used to convert the analytical separation method to the preparatory separation methods described in this application note. The use of the Preparative OBD Column Calculator has been described in a previous application note.3

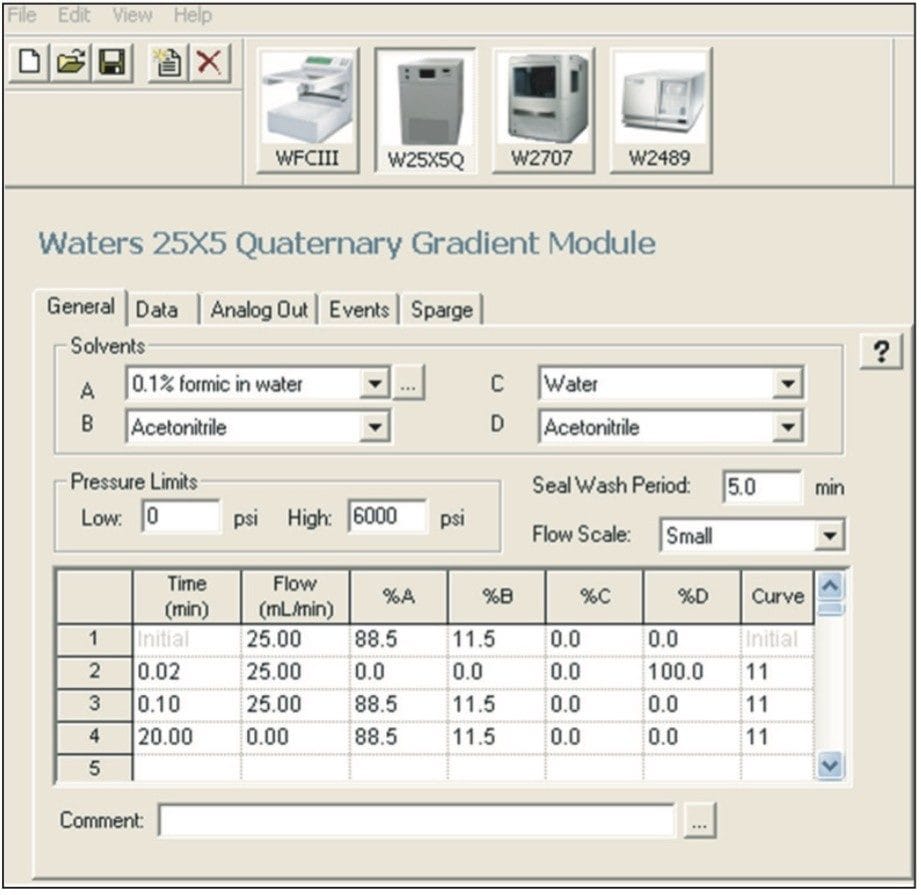
Using a 500-μL injection, acceptable reproducible preparative separations were achieved at 2.1% RSD retention time, as indicated in Figure 5. In order to increase the throughput, the method was further scaled to a 19-mm I.D. column. Injection volume scaling calculations indicated that a 2-mL injection was needed to keep the chromatography constant. Because the 2707 Autosampler was configured with a 1-mL loop, sample loading via the D line of the 2535 Quaternary Pump was used. In this loading technique, sample was introduced using a gradient table outlined in Figure 6. The gradient method was initiated via Empower 2 Software using the “Inject Immediate Sample Function.” Although the Autosampler is not used, it does not have to be removed from the system, which allows users to quickly choose sample introduction via the pump or autosampler.
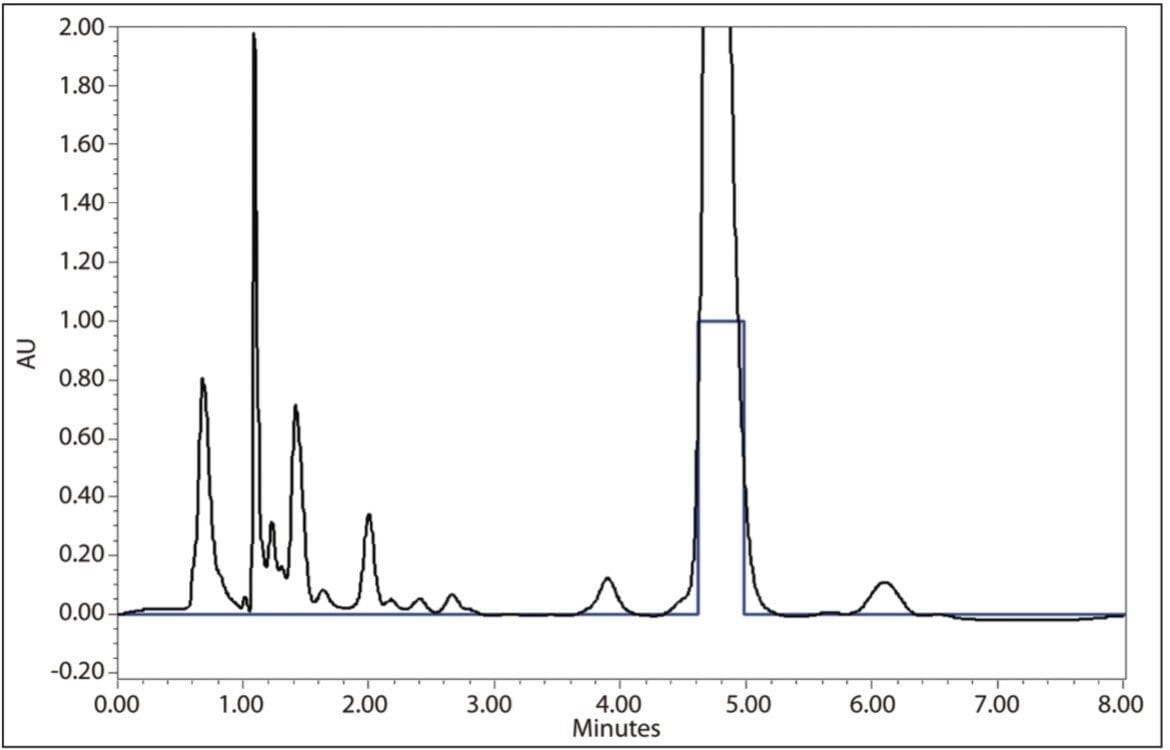
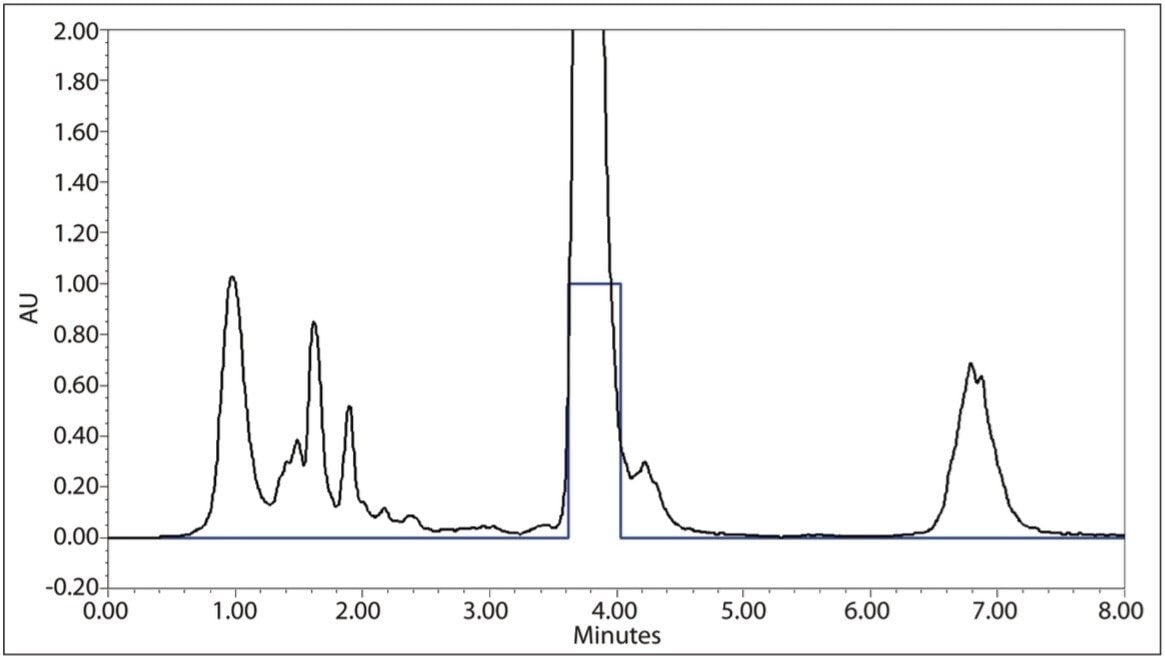
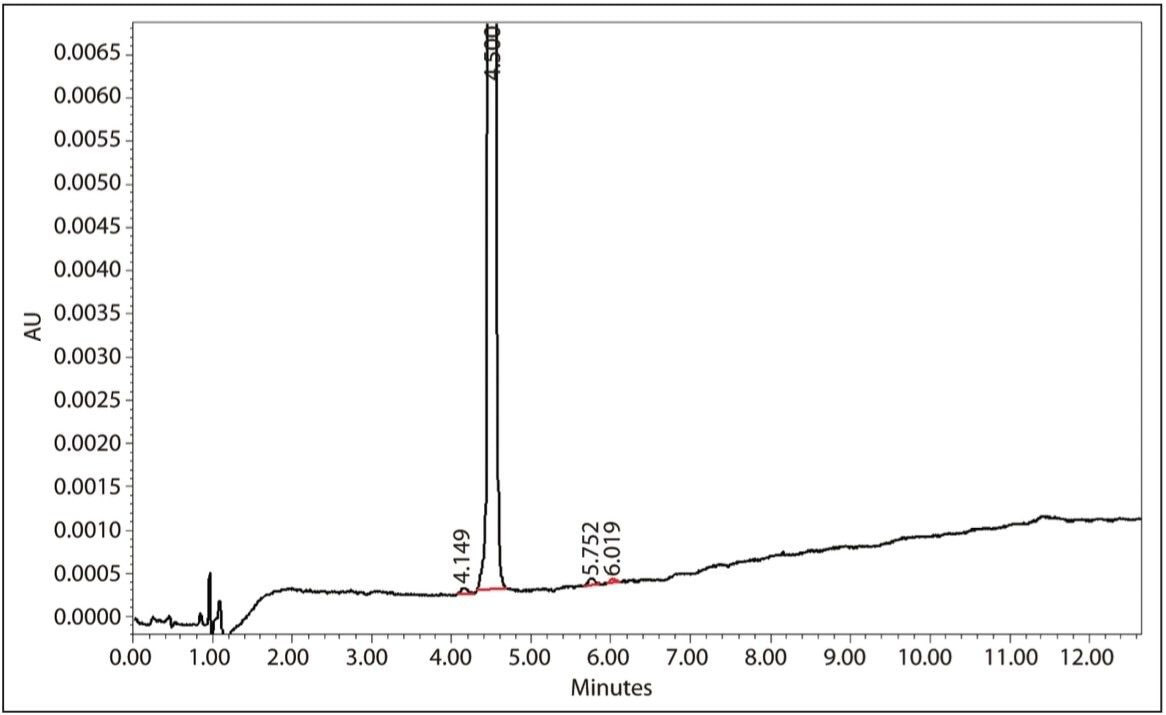
Acceptable, reproducible prep separations were again achieved with the 19-mm column shown in Figure 7. Fraction collection was set up using Empower 2 Software for threshold collection within a window. In this mode, a collection window is defined (in this case 3.0 to 6.0 minutes), and any peaks eluted outside of that window are not collected. Peaks eluting within the window must meet peak threshold criteria (in this case ~0.60 AU) to be collected. In addition, the fraction collector was set to collect multiple injections per position. Each peak is collected into the same bed position, as opposed to each peak being collected into a separate container. This allows for bulk-style purification and eliminates the need to pool collected fractions from identical sample injections. Ten separate injections totaling ~11.5 mg of puerarin were made on the 19-mm column with a total volume collected of 92 mL. Subsequent analysis of the collected fractions shown in Figure 8 had a puerarin concentration of 0.099 mg/mL with a UV area purity of >97.5%. The overall yield of puerarin from the kudzu extract was 79.6%.
To speed dry-down time, the system was used to concentrate the collected 92-mL fraction. The 92 mL collected was first diluted to 1 L with water. The column was then equilibrated with 99:1 water/acetonitrile. The entire diluted fraction was placed onto the column via the load valve embedded on the front of the pump. Once the entire sample was on the column, a simple step gradient to 100% acetonitrile was run and the eluted peak was captured in <10 mL, a 10X concentration factor. This small, mostly organic fraction was then dried to liberate crystalline puerarin.
Puerarin, an extract of kudzu root, was isolated and purified using Waters modular preparative HPLC system. Separations were analytically developed and geometrically scaled to two sizes of prep columns. The bulk purifications performed in an automated manner, eliminating the need for constant user intervention. Finally, the collected fractions were concentrated using the same system and column that was used to isolate the compound of interest.
720003797, November 2010