Improving Sensitivity and Analytical Method Greenness using MaxPeak™ Premier 1.0 mm Internal Diameter Columns
Kenneth Berthelette, Chris Collins, Thomas H. Walter
Waters Corporation, United States
Published on February 16, 2026
Abstract
Sensitivity is an important aspect of metabolomics studies, as some analytes are present at low concentrations. In this application note, we compare the results obtained using two different columns for the liquid chromatography / mass spectrometry analysis of citric and isocitric acids. Both columns were packed with the same batch of stationary phase but differed in the column hardware. The results showed improved peak areas using an inert MaxPeak Premier 1.0 mm internal diameter (ID) Column compared to a conventional stainless steel equivalent. Additionally, the Analytical Method Greenness Scores and mobile phase consumption were calculated for the method using the 1.0 mm column and a reference method using a 2.1 mm ID column.
Benefits
- Up to a 133% increase in peak area using a MaxPeak Premier 1.0 mm Column compared to a stainless steel 1.0 mm column
- A 77% reduction in mobile phase usage compared to a reference method using a 2.1 mm ID MaxPeak Premier Column
- A 21% lower AMGS compared to the reference method shows the reduced environmental impact of the method using the 1.0 mm ID column
Introduction
Accurate characterization of metal-sensitive analytes can be challenging due to interactions between the analyte and the system and column hardware. Most LC systems and columns employ stainless steel hardware, with titanium being used for some applications. Both stainless steel and titanium have a metal oxide layer on their surface,1 which carries a positive charge under acidic conditions.2 When negatively charged analytes like citric and isocitric acids are analyzed using this type of hardware, they can interact ionically with the metal oxide layer leading to peak tailing, and analyte loss through adsorption.3
One way to mitigate these interactions is to use inert hardware like that employed in MaxPeak Premier Columns and ACQUITY™ Premier Systems. Both incorporate MaxPeak High Performance Surfaces (HPS) Technology, which covers the metal oxide surfaces with an inert barrier.4 This technology has previously shown success in mitigating interactions with metal hardware for a variety of analytes including some cellular metabolites, radioligand therapy precursors, kinase inhibitors, and bisphosphonate drugs.3–7 Many cellular metabolites, particularly those with phosphate and carboxylate groups, are prone to adsorption on metal surfaces. Metabolomics workflows present several challenges, including achieving sufficient sensitivity to accurately characterize a metabolome. To achieve higher sensitivity, it may be advantageous to employ microflow columns with 1.0 mm ID. Smaller ID columns have been shown to improve sensitivity compared to 2.1 mm ID columns.8 The availability of microflow columns with MaxPeak HPS Technology not only solves the adsorption issue but can also reduce mobile phase usage and improve the overall “greenness” of a method.
This application note takes a previously published method and scales it down using a MaxPeak Premier 1.0 mm Column. The original method benefits from the improvements seen when employing HPS Technology for the analysis of citric and isocitric acids.3 In this work, a 1.0 mm ID HPS Column was compared to a 1.0 mm stainless steel column to confirm the benefits of HPS for these compounds. Additionally, the mobile phase consumption for the original method and the scaled method were compared. Lastly, the AMGS were calculated for both the original method and the 1.0 mm ID column method to compare their environmental impact.
Experimental
Sample Description
Citric and isocitric acid stock solutions were prepared at 1.0 mg/mL in water. Stock solutions were combined and diluted to a final concentration of 100 µg/mL each in water.
Method Conditions
Reversed-Phase LC Conditions
|
LC system: |
ACQUITY Premier QSM System with CHA and ACQUITY QDa™ II Mass Detector |
|
Detection: |
Single ion recording (SIR) @ 191.2 m/z |
|
Columns: |
ACQUITY UPLC™ CSH™ Phenyl-Hexyl Column, 1.0 x 100 mm, 1.7 µm (p/n: 186005402) |
|
ACQUITY Premier CSH Phenyl-Hexyl Column, 1.0 x 100 mm, 1.7 µm (p/n: 186011536) |
|
|
Column temperature: |
30 °C |
|
Sample temperature: |
10 °C |
|
Injection volume: |
3.0 µL |
|
Flow rate: |
0.1 mL/min |
|
Mobile phase A: |
0.1% Formic Acid in Water |
|
Mobile phase B: |
0.1% Formic Acid in Acetonitrile |
|
Gradient conditions: |
Initial condition 100% A; linear ramp to 25% B in 3.64 minutes; linear ramp to 95% B in 2.72 minutes. Hold for 0.91 minutes then return to starting condition and hold for 1.81 minutes. The total run time is 9.09 minutes. |
Data Management
|
Chromatography software: |
MassLynx™ V4.1 MS Software |
Results and Discussion
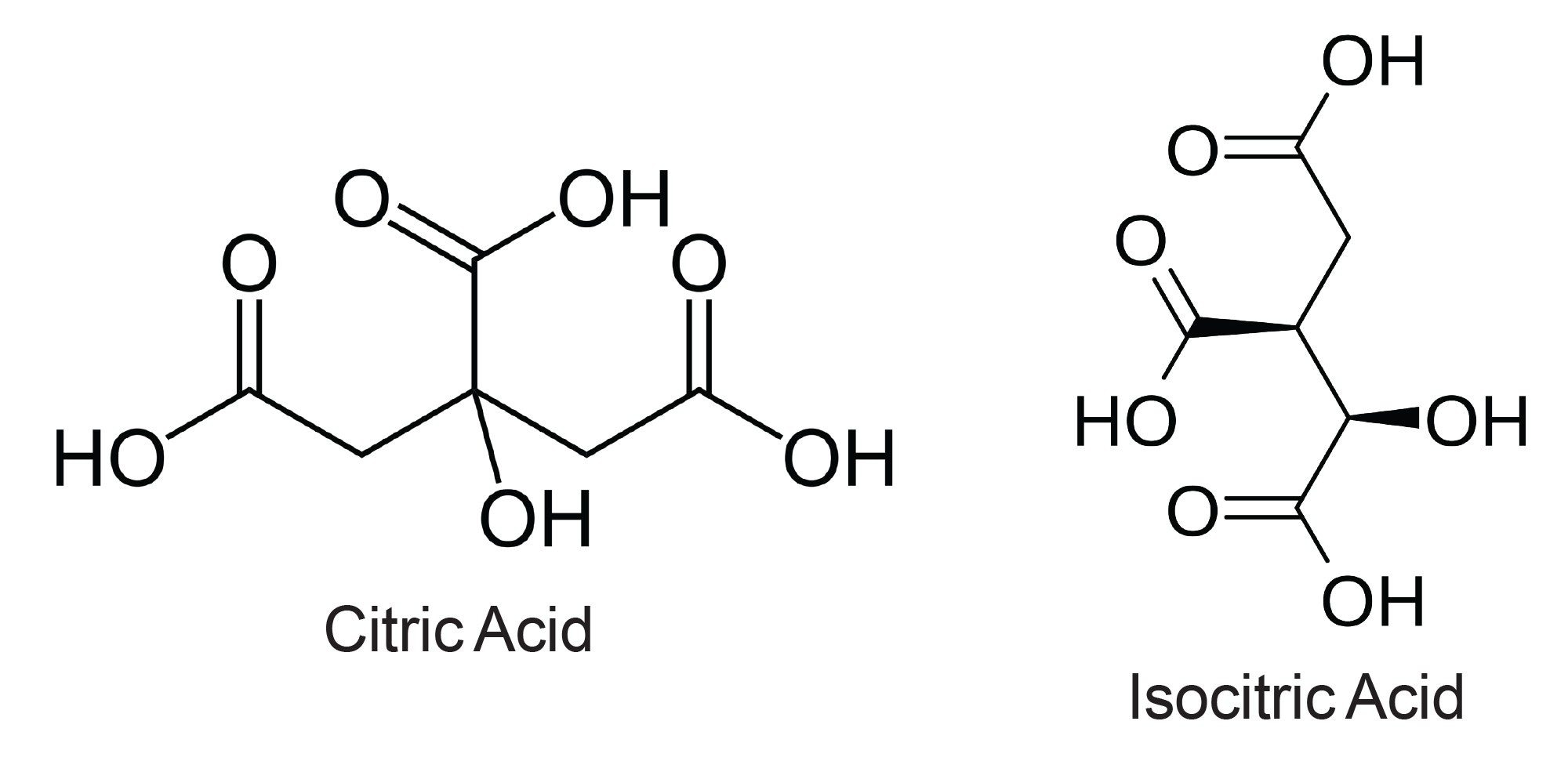
Citric and isocitric acids are structural isomers containing three carboxylic acid groups. They differ in the location of a hydroxyl group, either at the C2 or C3 position for isocitric and citric acid respectively, as shown in Figure 1.
LC analyses of these compounds commonly use an acidic mobile phase to minimize their ionic charge so that they are better retained on reversed-phase columns. These compounds are very polar and benefit from the increased retention provided by mixed-mode reversed-phase/anion-exchange columns like ACQUITY CSH Phenyl-Hexyl Columns. The positive charge of this stationary phase at low pH provides anion-exchange in addition to reversed-phase retention.
The reference method utilized a 2.1 x 100 mm ACQUITY Premier CSH Phenyl-Hexyl Column to improve recovery of both citric and isocitric acids. In order to create a microflow method, 1.0 mm ID columns were used along with appropriate method scaling. A comparison of the separations obtained using stainless steel and HPS 1.0 mm columns is shown in Figure 2.
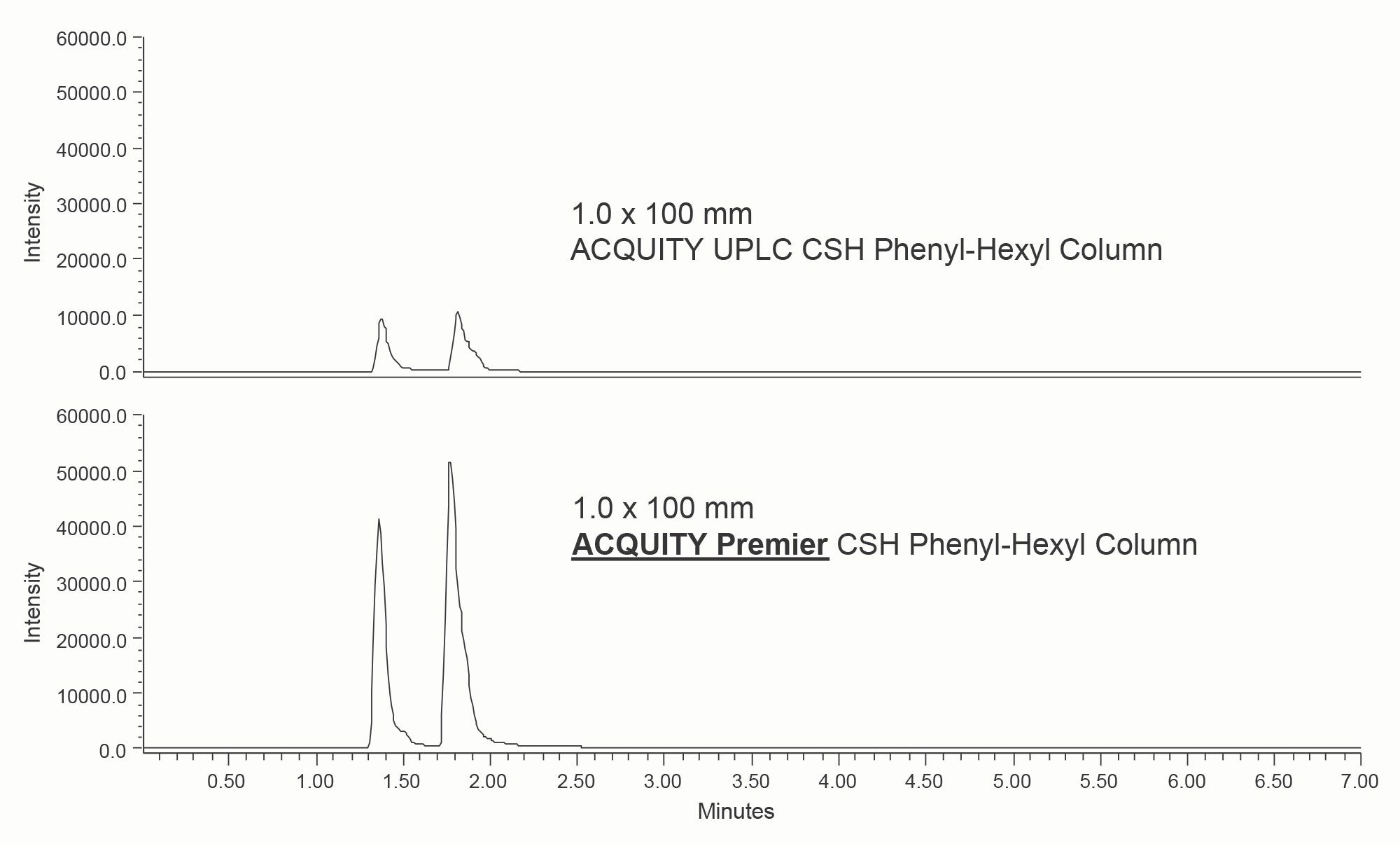
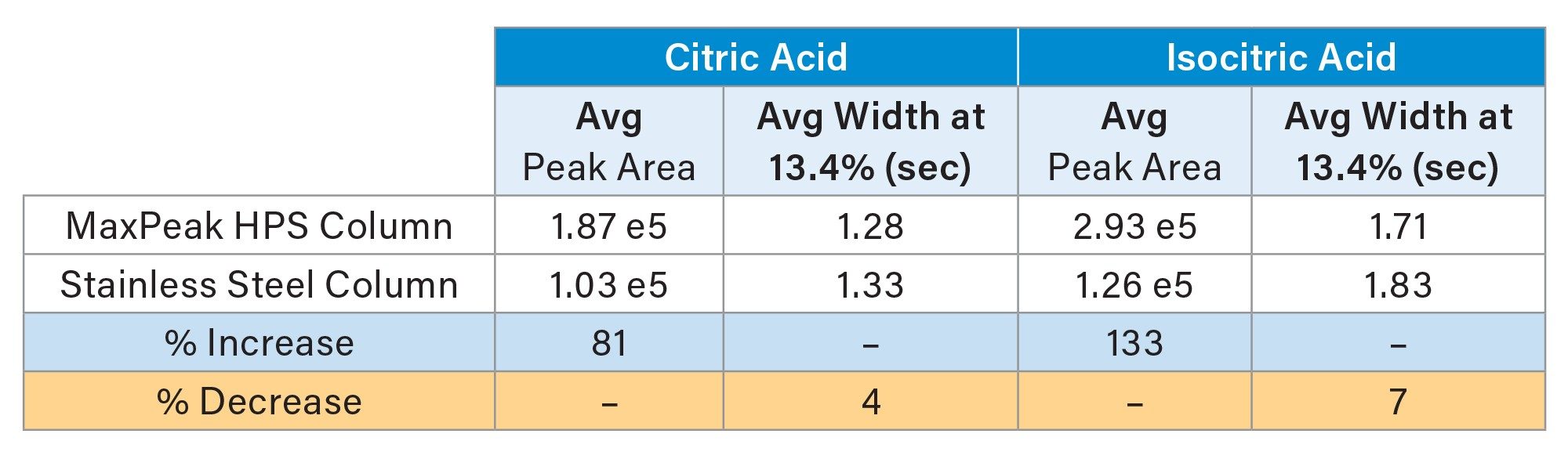
As shown, using an ACQUITY Premier 1.0 mm Column greatly improved the peak heights for both compounds. This is in agreement with the results seen in the original method using 2.1 mm ID columns. The average peak areas and peak widths at 13.4% (for 6 injections) are shown in Table 1. For citric acid, a 81% increase in peak area was realized by using an HPS column. A 133% higher peak area was obtained for isocitric acid. Smaller improvements were seen for the peak widths, with 4% and 7% decreases using the MaxPeak HPS Column.
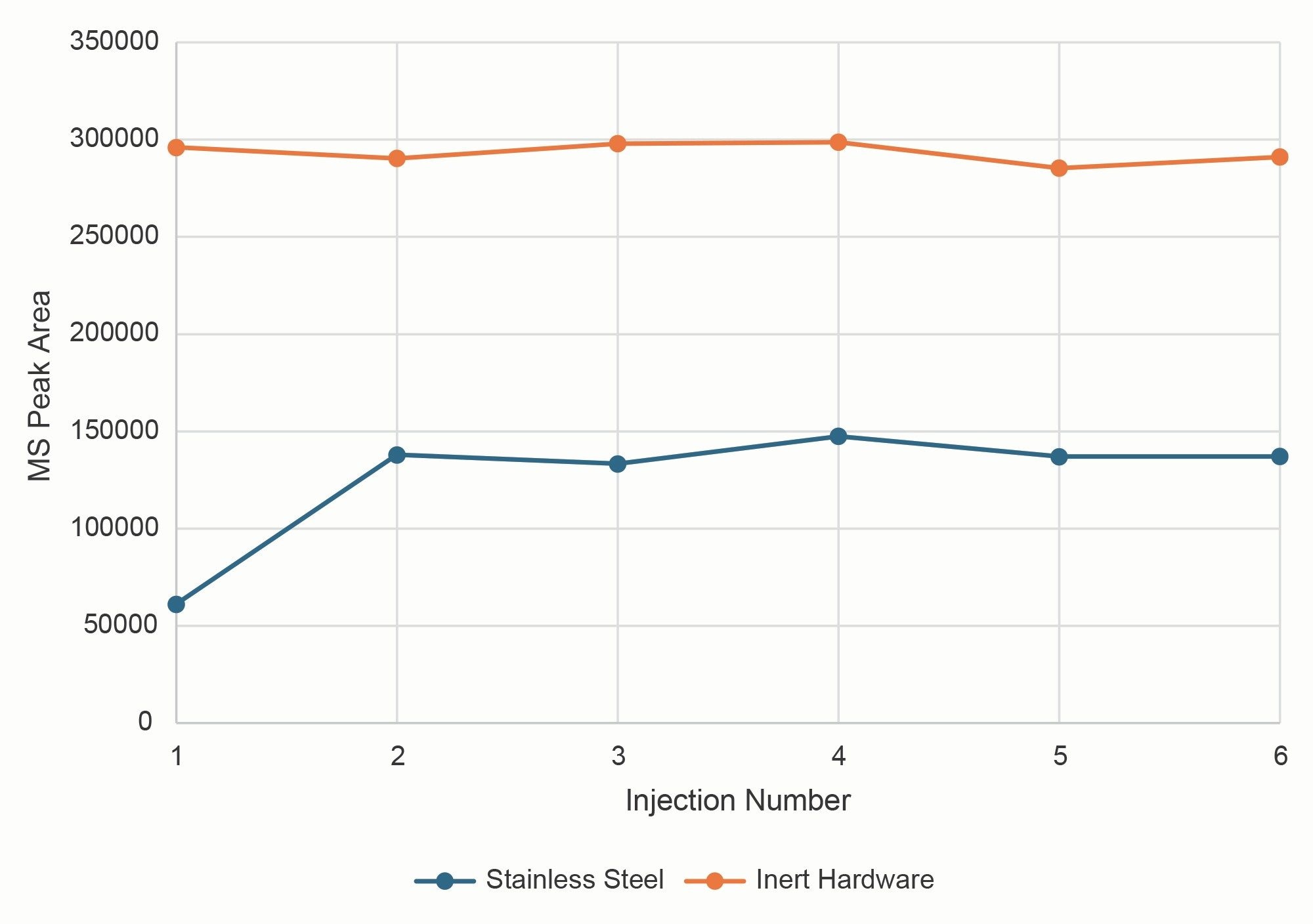
Examining the replicate injections of isocitric acid using the stainless steel column shows that the peak area was very low for the first injection, but increased in the subsequent injections. This is due to conditioning of the stainless steel hardware by the sample. The column made with inert hardware gave consistent peak areas from the first to last injection. A plot of peak areas across all six replicates for isocitric acid is shown in Figure 3. It’s notable that even though the peak area increased for injections 2–6 for the stainless steel column, the areas were still about 50% lower than those determined using the inert hardware column.
While the MaxPeak HPS Column provides significant improvements compared to stainless steel, the use of MaxPeak Premier 1.0 mm Columns offers additional benefits compared to MaxPeak Premier 2.1 mm Columns, namely decreased mobile phase consumption and improved analytical method greenness. Comparing the method described in this application note to the reference method, a calculation of mobile phase consumption per injection was performed. The results are shown in Table 2.
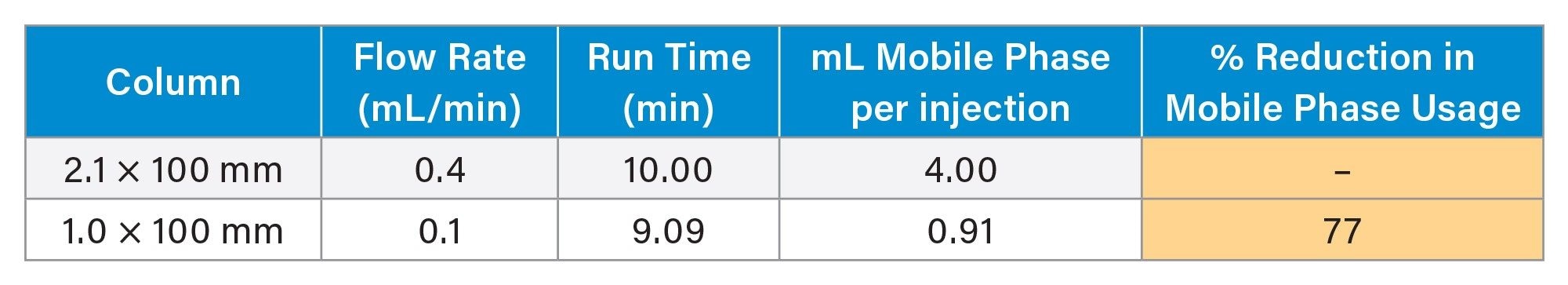
A 77% reduction in mobile phase usage was realized by switching to a 1.0 mm ID column. Over an entire sample set, this could amount to significant solvent savings, translating directly into decreased operating costs. The solvent savings also have an impact on the AMGS. The AMGS is a metric created as a means of comparing the environmental impact of analytical methods.9 Using the online calculator10 and inputting the two method conditions, the AMGS values for both methods were compared, as shown in Table 3. It is important to note that when using the AMGS calculator, there are input values for not only the mobile phase usage, but also for stock solution and working solution preparations. These can have an effect on both the solvent energy score and the solvent EHS score. In this case, when using the calculator, 1 injection was used for calculation, with two analytes of interest. Additionally, two stock preparations were created at 1 mL each in water. Lastly, one sample solution was prepared at 1 mL with water. Inputting these values along with the mobile phase and gradient parameters led to the values shown.
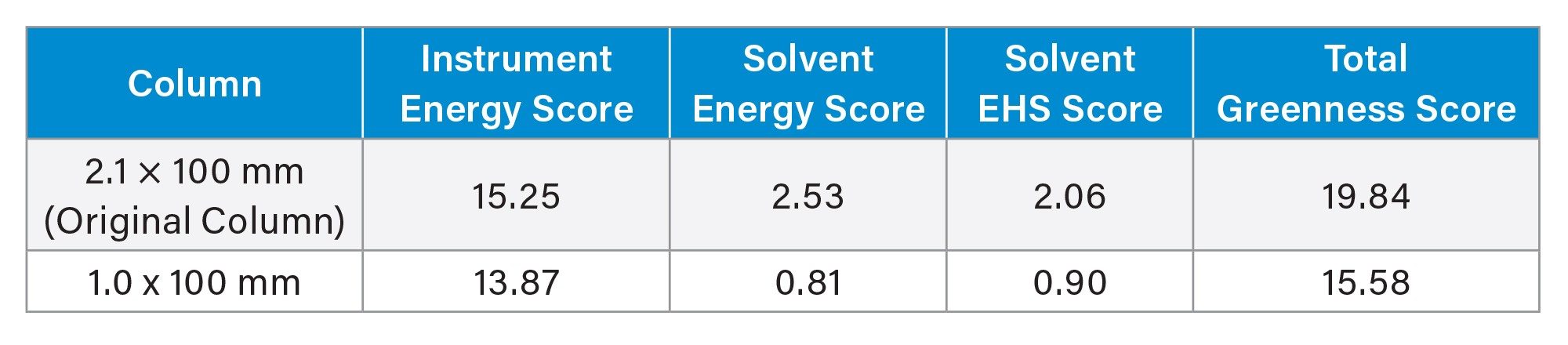
As expected, the instrument energy score is similar for the two methods. Given that the run times between the two methods are similar, the difference in energy needed to operate the system is small with only a 9% decrease in energy score using the 1.0 mm column. However, the solvent energy and solvent EHS scores are very different between the two columns. The 1.0 mm ID column has an approximately 56% lower solvent EHS score compared to the 2.1 mm ID column, and a 68% lower solvent energy score. These improvements are tied directly to the amount of solvent needed to run the methods. The total greenness score for the microflow method is 21% lower than that for the original method.
Using MaxPeak Premier 1.0 mm Columns can not only improve chromatographic results compared to stainless steel columns, but can also lead to greener methods due to lower solvent usage while lowering operating costs.
Conclusion
The use of MaxPeak Premier Columns improves the analysis of metal-sensitive analytes like citric and isocitric acids by mitigating the interactions between the analytes and the metal oxide layer of conventional stainless steel hardware. This technology is available in a variety of column configurations including 1.0 mm ID. These smaller ID columns require 77% less mobile phase than 2.1 mm ID columns, thus improving analytical method greenness.
References
- Collins K, Collins C, Betran C. Stainless Steel Surfaces in LC Systems, Part I – Corrosion and Erosion. LCGC 18 (6), (2000), 600-608.
- Sakamaki H, Uchida T, Lim LW, Takeuchi T. Evaluation of Column Hardware on Liquid Chromatography-Mass Spectrometry of Phosphorylated Compounds. J. Chromatogr. A. 1381,(2015), 125-131.
- Smith K, Rainville P. Utilization of MaxPeak High Performance Surfaces for Improved Separation and Recovery of Analytes Associated with the Tricarboxylic Acid Cycle. Waters Application Note. 720006727, 2020
- DeLano M, Walter TH, Lauber M, et al. Using Hybrid Organic-Inorganic Surface Technology to Mitigate Analyte Interactions with Metal Surfaces in UHPLC. Anal. Chem. 93, (2021), 5773-5781.
- Berthelette K, Aiello M, Collins C, Kalwood J, Walter TH. Analysis of Radioligand Therapy Components Using Reversed-Phase and HILIC Columns. Waters Application Note. 720006727, 2020
- Layton C, Rainville P. Advantages of Using MaxPeak HPS Technology for the Analysis of Targeted Cancer Growth Inhibitor Therapies. Waters Application Note. 720007565, 2021
- Aiello M, Berthelette K, Collins C, Walter TH. LC-MS Analysis of Underivatized Bisphosphonate Drugs Using Mixed-Mode Reversed-Phase/Anion-Exchange Chromatography. Waters Application Note. 720009039, 2025
- Rainville P, Wilson I, Plumb R, Johnson K. Comparison of 1- and 2- Millimeter ACQUITY UPLC Columns for LC-MS. Waters Application Note. 720001126, 2005
- Hicks MB, Farrell W, Aurigemma C, Lehmann L, Weisel L, Nadeau K, Lee H, Moraff C, Wong M, Huang Y, Ferguson P. Making the Move Towards Modernized Greener Separations: Introduction of the Analytical Method Greenness Score (AMGS) Calculator. Green Chem. 21, (2019), 1816-1826.
- ACS Green Chemistry Institute Pharmaceutical Roundtable website. Accessed 7-Jan-2026. https://acsgcipr.org/amgs/
Featured Products
720009221, January 2026