For research use only. Not for use in diagnostic procedures.
This application note describes an analytically validated bioanalytical clinical research method for the quantification of low concentration acylcarnitines in urine.
The amino acid derivative carnitine is involved in lipid metabolism, transporting long-chain acyl groups from fatty acids to the mitochondrial matrix, where they are broken down to form usable energy via the citric acid cycle. The analysis of these acylcarnitines in blood, urine, and plasma for clinical research studies is typically performed by tandem quadrupole MS coupled to gas chromatography, high-performance liquid chromatography, or capillary electrophoresis.2 In this application note, we describe an analytically validated bioanalytical clinical research method for the quantification of low concentration acylcarnitines in urine.3,4
Urine samples, stored at -80 °C, were left to thaw at 4 °C. A 30 µL aliquot of urine was centrifuged at 10,300 g for 10 minutes. Then, 20 µL of supernatant from each sample was diluted with 65 µL of water, 5 µL of acetonitrile, and 10 µL of the internal standard mixture. The internal standard mixture consisted of propionyl-L-carnitine-d3; butyryl-L-carnitine-d3, hexanoyl-L-carnitine-d3 and dodecanoyl-L-carnitine-d3 at 250 ng/mL and isovaleryl-L-carnitine-d9, octanoyl-L-carnitine-d3 and decanoyl-L-carnitine-d3 at 125 ng/mL. Tetradecanoyl-L-carnitine-d3 and hexadecanoyl-L-carnitine-d3 were also included at 125 ng/mL for testing of these analytes.
The UPLC-MS analysis was performed on an ACQUITY UPLC binary solvent manager, 2777 sample manager (CTC) and column manager (Waters, Milford, MA, USA) interfaced to a Xevo TQ-S Tandem Quadrupole Mass Spectrometer (Waters, Milford, MA, USA).
|
LC system: |
Open Architecture UPLC System |
|
Detection: |
Xevo TQ-S |
|
Vials: |
Waters certified vials |
|
Column: |
ACQUITY UPLC HSS T3 1.8 μm, 2.1 mm x 150 mm |
|
Column temp.: |
45 °C |
|
Sample temp.: |
4 °C |
|
Injection volume: |
2 μL |
|
Flow rate: |
0.5 mL/min |
|
Mobile phase A: |
0.1% formic acid |
|
Mobile phase B: |
0.1% formic acid in acetonitrile Gradient mode elution |
|
MS system: |
Xevo TQ-S |
|
Ionization mode: |
Positive ion |
|
Acquisition range: |
MRM mode |
|
Capillary voltage: |
2.75 kV |
|
Collision energy: |
14–26 eV |
|
Cone voltage: |
8–48 V |
|
Chromatography software: |
MassLynx Software |
|
MS software: |
MassLynx Software |
|
Informatics: |
TargetLynx Software |
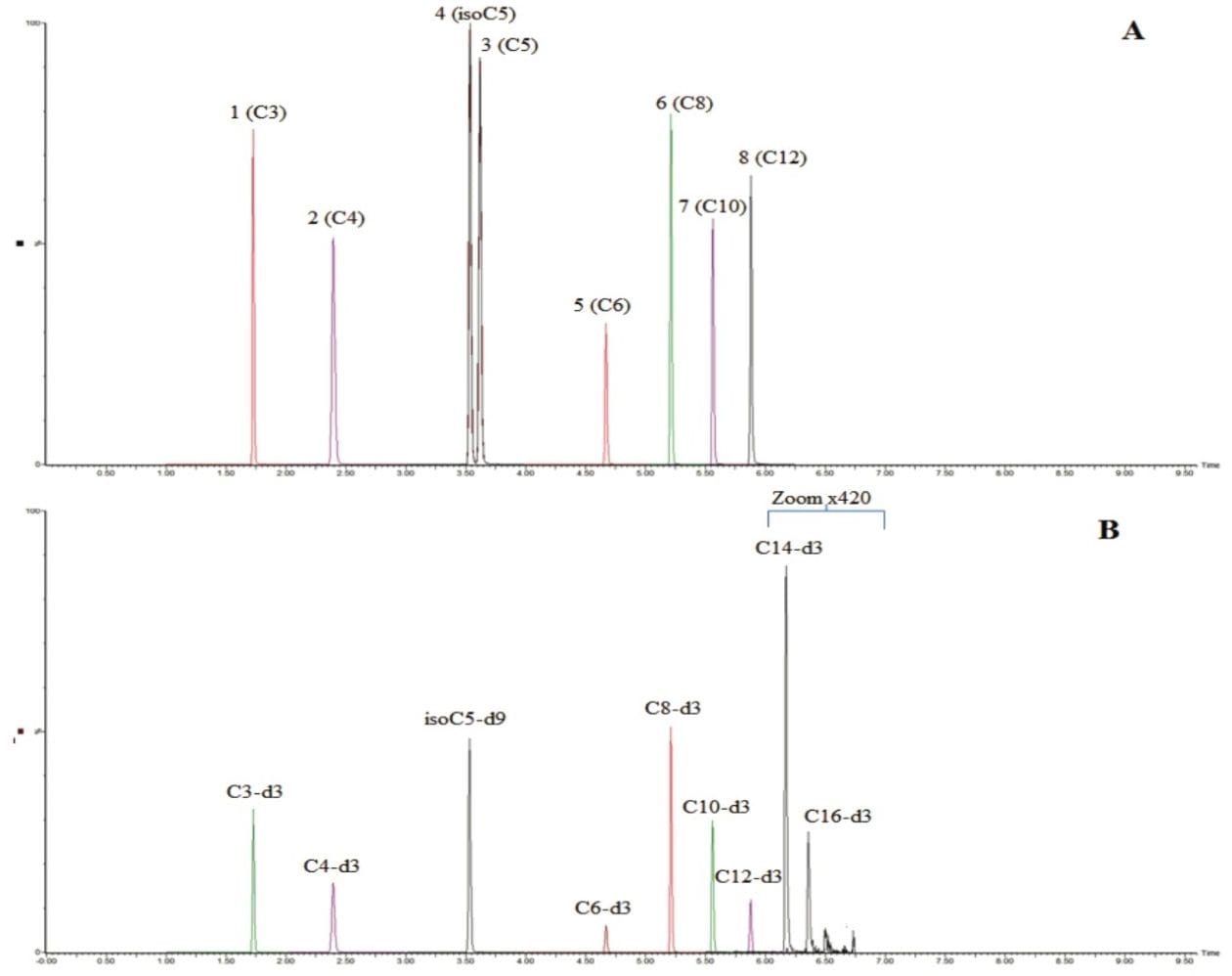
The chromatographic separation was developed to provide sufficient resolution of the analytes, with a run time of 10 minutes consistent with medium to high throughput analysis, while maintaining the separation between the two pairs of isomeric species isovaleryl-L-carnitine (isoC5) and valeryl-L-carnitine (C5), and isobutyryl-L-carnitine (isoC4) and butyryl-L-carnitine (C4). Due to their relatively low concentrations in urine, compared to carnitine itself and some other high concentration acylcarnitines, the analytes measured in this method, propionyl-L-carnitine (C3), butyryl-L-carnitine (C4), isovaleryl-L-carnitine (isoC5), valeryl-L-carnitine (C5), hexanoyl-L-carnitine (C6), octanoyl-L-carnitine (C8), decanoyl-L-carnitine (C10) and dodecanoyl L-carnitine (C12) need to be quantified using a method optimized for low concentration acylcarnitines. Dodecanoyl L-carnitine (C12), tetradecanoyl-L-carnitine (C14) and hexadecanoyl-L-carnitine (C16) are too low in concentration in the control urine to be quantified using the sample preparation method for this analysis, however, the internal standards are displayed in Figure 1B for the purposes of demonstrating retention time and could allow for testing of these analytes.
The samples were analyzed using a reversed-phased gradient separation with the methodology optimized for analyte resolution and reproducibility. The analtyes were detected and quantified by positive ion MRM with optimal MS conditions determined by the infusion of authentic analyte and stable isotope labelled internal standards to determine the maximum response for each transition. For quantification, one transition from each of these precursor ions was used to calculate unknown concentrations. The choice of quantification transition was determined from the ionization energy of each compound.
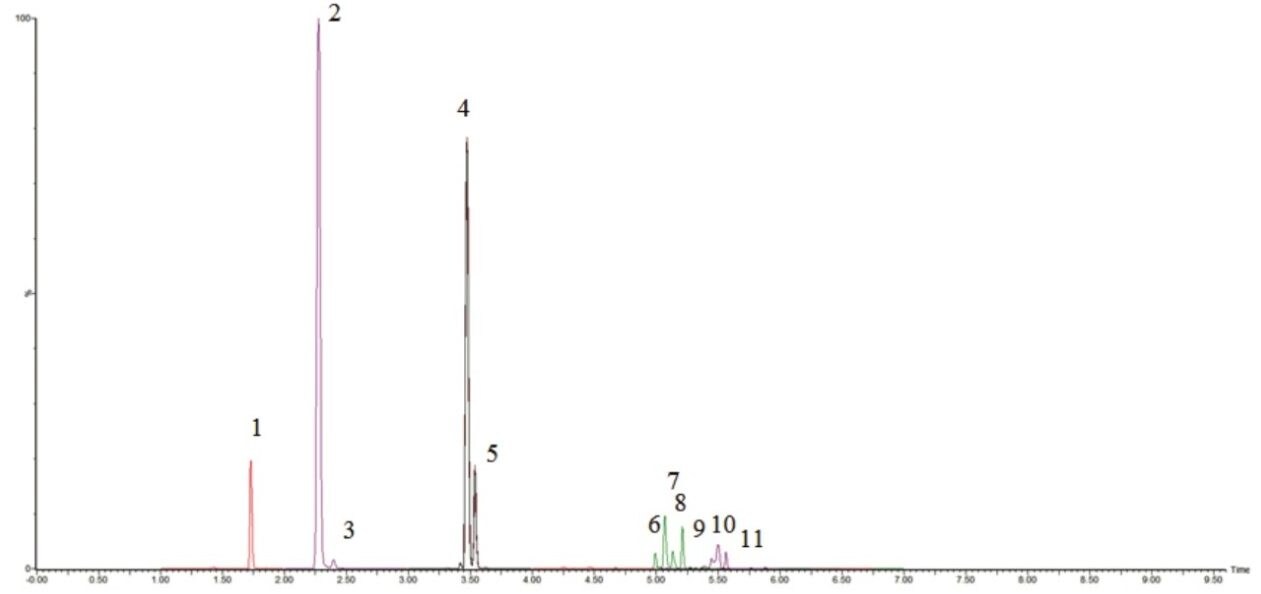
Typical chromatograms for the UPLC-MS/MS analysis of the acylcarnitine standards are shown in Figure 1A and 1B. Figure 2 shows a typical chromatogram of a urine sample diluted one part in five. All eight acylcarnitines examined were chromatographically resolved. The separation was shown to be highly reproducible, with retention time coefficients of variance for the acylcarnitine standards at the ULOQQC of <0.20% across the entire validation run (n=18).
The method was linear over the calibration ranges employed for the individual analytes, using a linear fit with a weighting of 1/X. The correlation coefficient (r2) values of the individual standard curves were determined to be between 0.988 and 0.999 for all analytes.
The method was assessed for accuracy linearity and bias over three days for the concentration ranges 7.5–100 ng/mL for propionyl-L-carnitine (C3); 3–40 ng/mL for butyryl-L-carnitine (C4), valeryl-L-carnitine (C5), isovaleryl-L-carnitine (isoC5), hexanoyl-Lcarnitine (C6) and dodecanoyl-L-carnitine (C12); 1.5–20 ng/mL for octanoyl-L-carnitine (C8) and decanoyl-L-carnitine (C10).
All of the analytes monitored showed excellent accuracy and precision. As an example, the coefficient of variance and mean concentration data obtained for the method data for day three are shown in Tables 1 and 2 respectively.

Table 1. Summary of the coefficient of variance for day three
Numbers rounded to three significant figures, or integers, as appropriate.

Table 2. Summary of the mean concentration data for day three
Numbers rounded to three significant figures, or integers, as appropriate.
The inter-day validation results are summarized in Table 3 and Table 4. Table 3 summarizes the inter-day coefficients of variance for each QC standard and Table 4 summarizes the inter-day mean concentration for each QC standard. The precision of the back calculated standards across the inter-day validation ranged from 0.89 to 9.75%, while the mean back calculated concentrations obtained for each of the standards gave mean bias values ranging from 3.25 to 8.20%. The Lower Limit of Quantification (LLOQ) was defined as 7.5 ng/mL for propionyl-L-carnitine (C3), 3 ng/mL for butyryl-L-carnitine (C4), valeryl-L-carnitine, (C5), isovaleryl-L-carnitine (isoC5) and hexanoyl-L-carnitine (C6), and 1.5 ng/mL for octanoyl L-carnitine (C8). However, for dodecanoyl-L-carnitine the LLOQ was 4 ng/mL and decanoyl- L-carnitine (C10) was 2 ng/mL, although the standards were tested to 3 ng/mL and 1.5 ng/mL respectively. The mean bias of the method ranged from -1.7% for decanoyl-L-carnitine, (C10) to +14.7% for dodecanoyl-L-carnitine (C12).
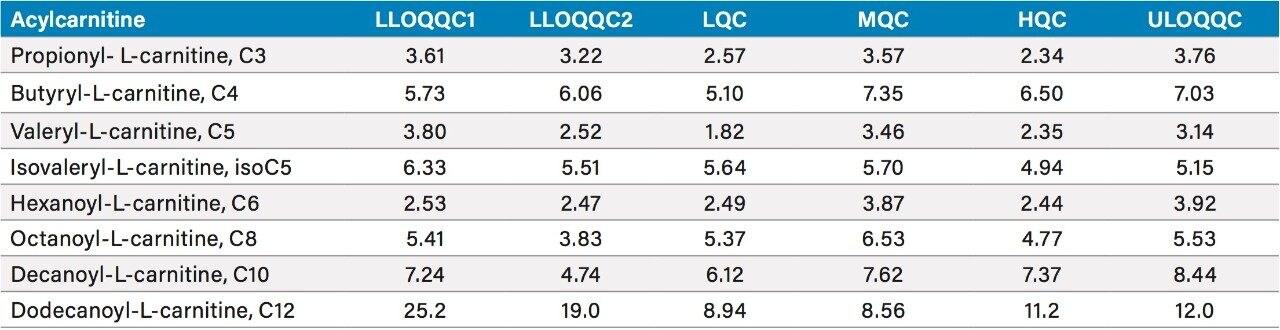
Table 3. Summary of the inter-day coefficient of variance
Numbers rounded to three significant figures, or integers, as appropriate.
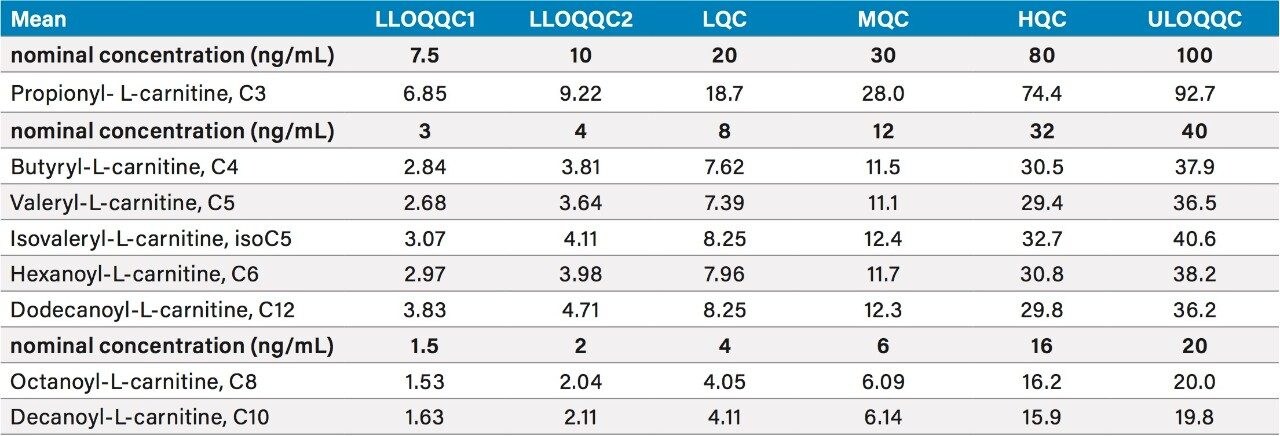
Table 4. Summary of the inter-day mean concentration data
Numbers rounded to three significant figures, or integers, as appropriate.
Carryover was assessed by running a double blank (water, containing no internal standard and no analytes) immediately after a upper limit of quantitation (ULOQ) calibration standard. The results obtained indicate that there was no significant carryover for the acylcarnitine standards. Carryover is the peak area of a compound found in the blank sample calculated as a percentage of the peak area of the compound in the lower limit of quantification quality control (LLOQQC). The highest percentage carry over at 14.27% was observed for dodecanoyl-L-carnitine. This is within the accepted limit of 20%. The internal standards carryover, at less than 0.98% for all compounds, was negligible.
Based on the use of stable isotope labelled internal standards, a quantitative clinical research method was validated over the range 7.5–100 ng/mL (37.5–500 ng/mL in urine) for propionyl-L-carnitine (C3) and 3–40 ng/mL (15–200 ng/mL in urine) for butyryl-L-carnitine (C4), valeryl-L-carnitine, (C5), isovaleryl-L-carnitine (isoC5) and hexanoyl-L-carnitine (C6). Dodecanoyl-L-carnitine (C12) ranged from 4–40 ng/mL (20–200 ng/mL in urine) while the octanoyl-L-carnitine (C8) range is from 1.5–20 ng/mL (7.5–100 ng/mL in urine) and decanoyl-L-carnitine (C10) from 2–20 ng/mL (10–100 ng/mL in urine). The methodology required only 30 µL of urine and exhibited excellent analytical sensitivity, robustness, and reproducibility.
720005974, May 2019