In this application note, we present the use of a simple sample extraction and dSPE cleanup where the resulting extract is analyzed by UPLC-MS/MS and/or GC-MS/MS for rapidly monitoring pesticides and mycotoxins in cannabis matrix to meet California regulations. With the variety of residues to be monitored as well as the continued possibility of new ones being added, method generation can be a tedious task.
The increased use of both medical and recreational cannabis in combination with its expanding legal acceptance in several US states1 has led to demanding cannabis safety and quality control testing. Analytical testing typically includes cannabinoids profiling/potency, mycotoxins, terpenes, residual solvents, metals, and pesticide residues analysis. Pesticides are of particular interest as they are widely used in the cultivation of cannabis plants to safeguard against harmful insects and to promote crop yields. The application of pesticides is strictly regulated,2 and their residues in cannabis products are closely monitored by state regulatory agencies in order to protect consumers. The number of regulated pesticides that are required to be monitored varies by state as do the action limits which range from 0.01 µg/g (10 ppb) to above 1 µg/g (1 ppm).
In addition to pesticides, cannabis intended for inhalation, ingestion, or topical application must also be tested for mycotoxins. Mycotoxins, including aflatoxins and ochratoxins, are naturally occuring toxins produced by certain strains of mold. This mold, or mycotoxin contamination, can occur during either cultivation or storage and the toxins produced present a serious health risk to consumers. Routine testing for mycotoxins at low levels is critical to ensure the health of consumers, particularly those who may already have compromised health. As with pesticides, a robust and rapid test is critical and single simultaneous test for pesticides and mycotoxins is ideal.
Multi-residue compound detection is routinely performed using tandem quadrupole mass spectrometry (MS/MS) in combination with liquid chromatography (LC) and gas chromatography (GC). Both LC-MS/MS and GC-MS/MS are commonly used for multi-residue pesticide analysis as some residues are only amenable to either LC or GC. Tandem quadrupole MS is the detector of choice as it provides high sensitivity and selectivity for simultaneous analysis of hundreds of pesticides at low ng/g (ppb) levels in a single analysis.
In this application note, we present the use of a simple sample extraction and dSPE cleanup where the resulting extract is analyzed by UPLC-MS/MS and/or GC-MS/MS for rapidly monitoring pesticides and mycotoxins in cannabis matrix to meet California regulations. With the variety of residues to be monitored as well as the continued possibility of new ones being added, method generation can be a tedious task. In this study, full analytical methods full analytical methods including LC, GC, and MS methods were utilized from Quanpedia eliminating the need for method development for the California pesticide and mycotoxin lists.
Standard compounds for 66 pesticides and 5 mycotoxins monitored on the California list were combined to produce a stock solution which was sequentially diluted to prepare the spiking solutions. The cannabis buds were first ground using a hand grinder. Aliquots of 0.5 g of ground material were weighed into 50mL centrifuge tubes and spiked with 0.10 µg/g (100 ppb) and 0.50 µg/g (500 ppb) of the acetonitrile spiking solutions. A 5 mL volume of acetonitrile was added and the samples were processed using a Geno Grinder for 3 minutes (1500 rpm). The mycotoxins were spiked at 0.02 µg/g (20 ppb) and 0.10 µg/g (100 ppb). The samples were then centrifuged at 5000 rpm for 5 minutes.
A 1 mL aliquot of the supernatant was added to a dSPE tube (2 mL centrifuge tube containing 150 mg MgSO4, 50 mg PSA, 50 mg C18, 7.5 mg graphitized carbon black), shaken for 1 minute, centrifuged, and the supernatant transferred to a sample vial for analysis by UPLC-MS/MS and/or GC-MS/MS. Extracted matrix that did not contain pesticide residues was used to generate matrix matched calibration curves. Prior to GC-MS/MS analysis, all samples were spiked with an internal standard mix (QuEChERS Internal Standard Mix for GC-MS Analysis from Restek).
LC separations were performed on Waters ACQUITY UPLC H-Class System and the Xevo TQ-S micro Tandem Quadrupole Mass Spectrometer. MassLynx MS Software (v4.2) was used for data acquisition and processing. GC separations were performed on the Xevo TQ-GC Tandem Quadrupole Mass Spectrometer using MassLynx MS Software (v4.2) for data acquisition and processing The Quanpedia Database and method generation software was used to automatically generate MRM acquisition and TargetLynx processing methods for both LC-MS/MS and GC-MS/MS.
|
UPLC system: |
ACQUITY UPLC H-Class |
|
Separation mode: |
Gradient |
|
Column: |
XBridge C18 2.5 μm, 2.1 × 150 mm |
|
Solvent A: |
5 mM ammonium formate with 0.020% formic acid in water |
|
Solvent B: |
Methanol |
|
Flow rate: |
0.400 mL/min |
|
Column temp.: |
50 °C |
|
Injection volume: |
5 μL |
|
Time (min) |
%A |
%B |
Curve |
|---|---|---|---|
|
0 |
98% |
2% |
– |
|
0.2 |
98% |
2% |
6 |
|
4 |
30% |
70% |
6 |
|
10 |
30% |
70% |
6 |
|
12 |
1% |
99% |
6 |
|
15 |
1% |
99% |
6 |
|
15.01 |
98% |
2% |
1 |
|
17 |
98% |
2% |
1 |
|
MS system: |
Xevo TQ-S micro |
|
Ionization mode: |
ESI+/ESI |
|
Capillary voltage: |
3.0 kV (+); 2.5 kV (-) |
|
Cone voltage: |
Various V |
|
Collision energy: |
Various eV |
|
Desolvation temp.: |
550 °C |
|
Source temp.: |
150 °C |
|
Desolvation gas: |
800 (L/hr) |
|
Cone gas: |
50 (L/hr) |
|
GC system: |
Xevo TQ-GC |
|
Column: |
Rxi-5MS 20 m × 0.18 mm × 0.18 μm |
|
Carrier gas: |
Helium |
|
Injection type: |
Pulsed splitless |
|
Injector temp.: |
280 °C |
|
Pulse time: |
1.5 min |
|
Pulse pressure: |
45 psi |
|
Inlet liner: |
Single taper splitless with wool |
|
Flow rate: |
2 mL/min |
|
Injection volume: |
1 μL |
|
Rate (°C/min) |
Temp. (°C) |
Hold (min) |
|---|---|---|
|
— |
60 |
0.45 |
|
18.7 |
330 |
2.25 |
|
Total run time = |
17.14 min |
|
|
Ionization mode: |
EI+, 70 eV |
|
|
GC interface temp.: |
300 °C |
|
|
Source temp.: |
250 °C |
|
|
MS resolution: |
IntelliStart Custom Resolution settings were used |
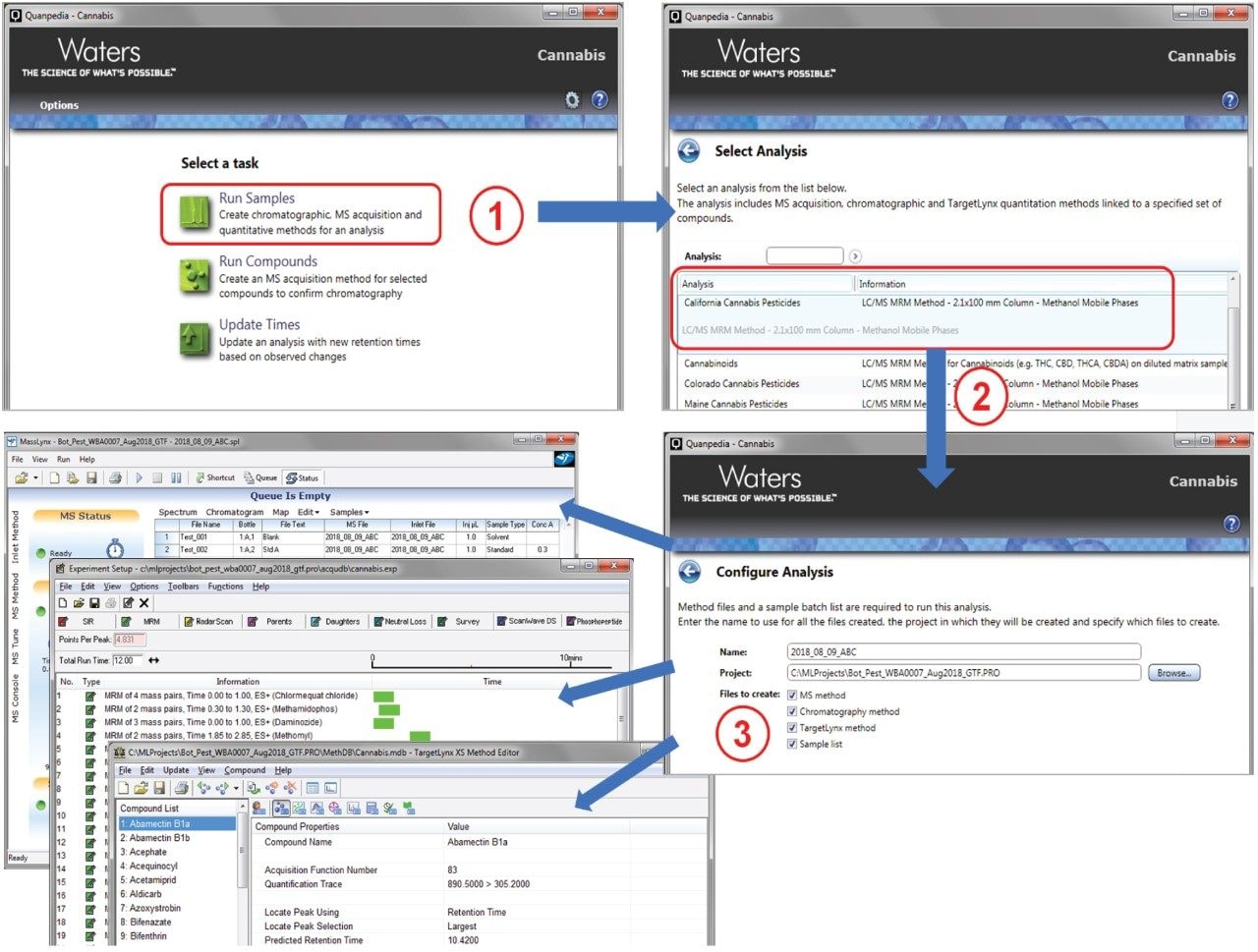
Waters Quanpedia method database was used to automatically create the LC, GC, MS, and data processing methods (Figure 1) for the various target analytes to be monitored using the MRM transitions as listed in Appendix Tables 1 to 3. Users can quickly generate pre-defined LC-MS/MS, and GC-MS/MS methods in just three steps, which eliminates the level of potential error and the complexity involved in method development for large numbers of target analytes. Another advantage is that Quanpedia greatly decreases the amount of work, time, and resources required for laboratories to set up methods. Quanpedia also contains functionality to quickly adjust retention times associated with a method, eliminating the lengthy process of manually adjusting MRM time windows due to retention time shifts. This UPLC-MS/MS method contained 67 compounds (62 pesticides and 5 mycotoxins) and the GC-MS/MS method contained 54 compounds, fully covering the California requirements for pesticide and mycotoxin residue analysis.
Currently, US states and Canada have defined different testing requirements for pesticide residue testing in cannabis. The list of pesticides varies with each state. Furthermore, the composition and complexity of the matrix varies widely across different cannabis strains (or cultivars). The combination of long lists of pesticides with variable and complex matrices presents a significant challenge in method development.
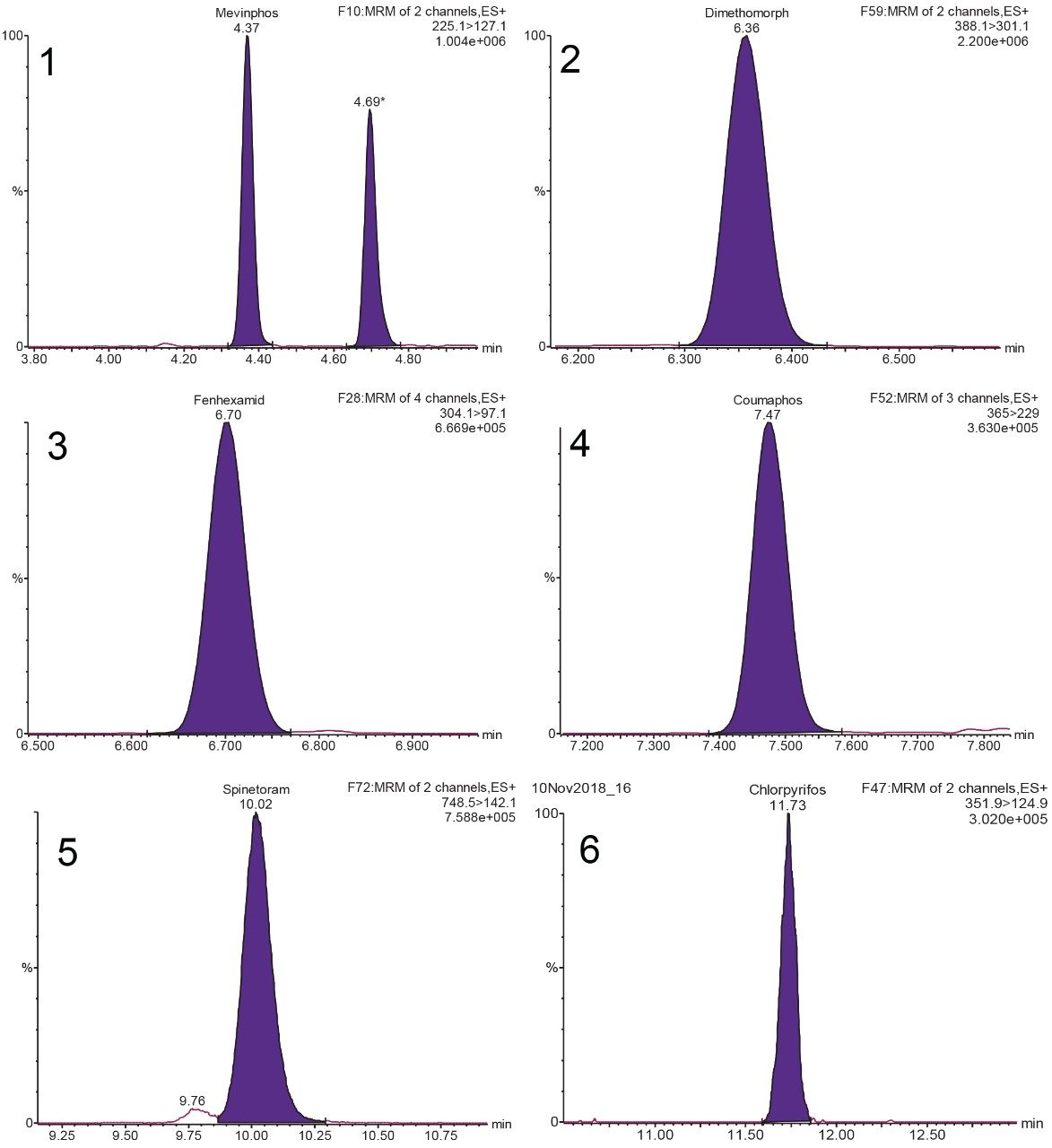
The State of California monitors 66 target pesticides in cannabis.3 This list completely encompasses the Oregon pesticide4 list minus MGK-264, and it has additional pesticides including spinetoram, captan, chlordane, pentachloronitrobenzene, chlorpyrifos, coumaphos, dimethomorph, fenhexamid, and mevinphos. Representative MRM chromatograms for selected pesticides are displayed in Figure 2. Chromatogram 1 shows the cis and trans-isomers of mevinphos.
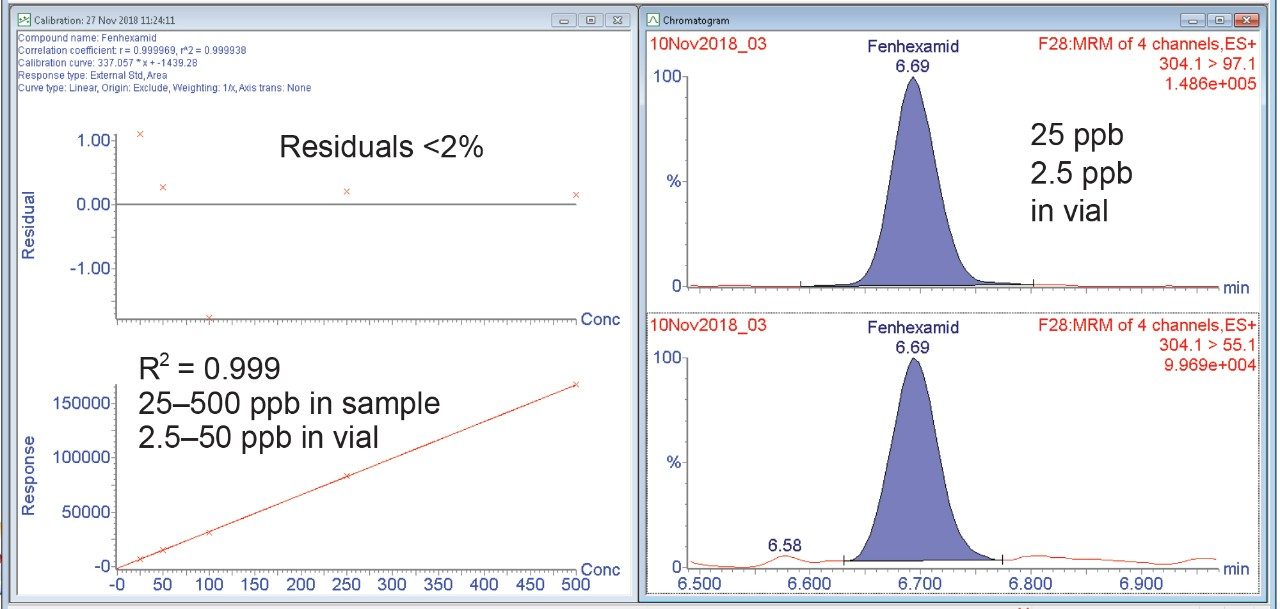
Matrix matched calibration curves were generated using blank extracted cannabis. An example of the quantitation curve for the pesticide fenhexamid is shown in Figure 3. Linear calibration curves (R2>0.990) for all pesticides were obtained over the range tested 0.025 to 0.50 µg/g (25 to 500 ppb in sample or 2.5 to 50 ppb in vial concentration).
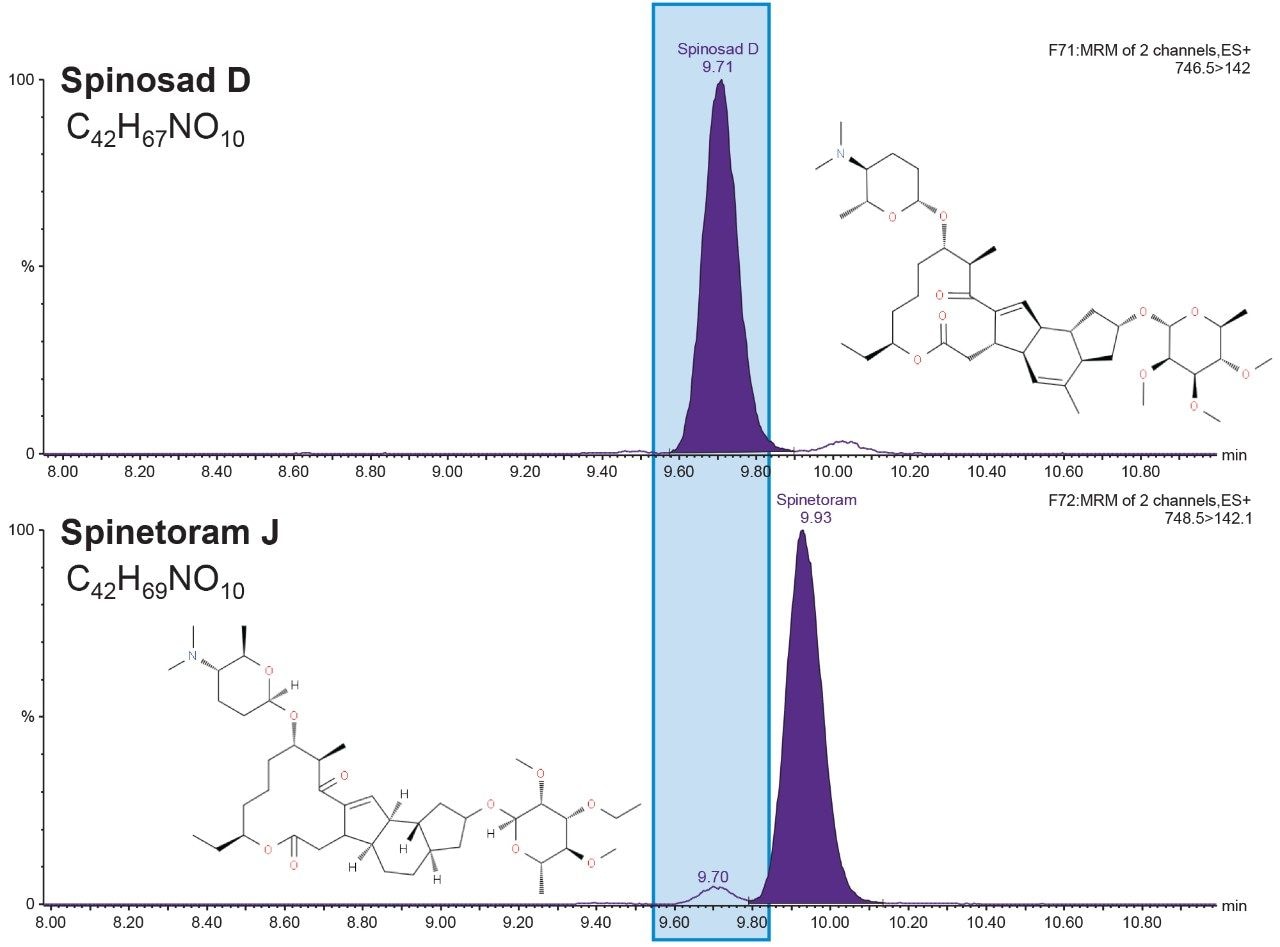
Multi-pesticide residue analysis requires careful attention to the MRM transitions of co-eluting compounds since there can be signal interferences which can lead to inaccurate quantitation. The compounds spinetoram J and spinosad D share common structural features and produce the same primary and secondary ion fragments during collision induced dissociation (CID). The precursor m/z for spinosad D is 746.5 and that of spinetoram J is m/z 748.5. The isotopic form of spinosad D that contains two carbon-13 atoms gives rise to a signal in the same MRM channel used for analysis of spinetoram J (Figure 4). When both pesticides co-occur in a mixture, accurate quantitation of each individual pesticide requires chromatographic separation as shown in Figure 4.
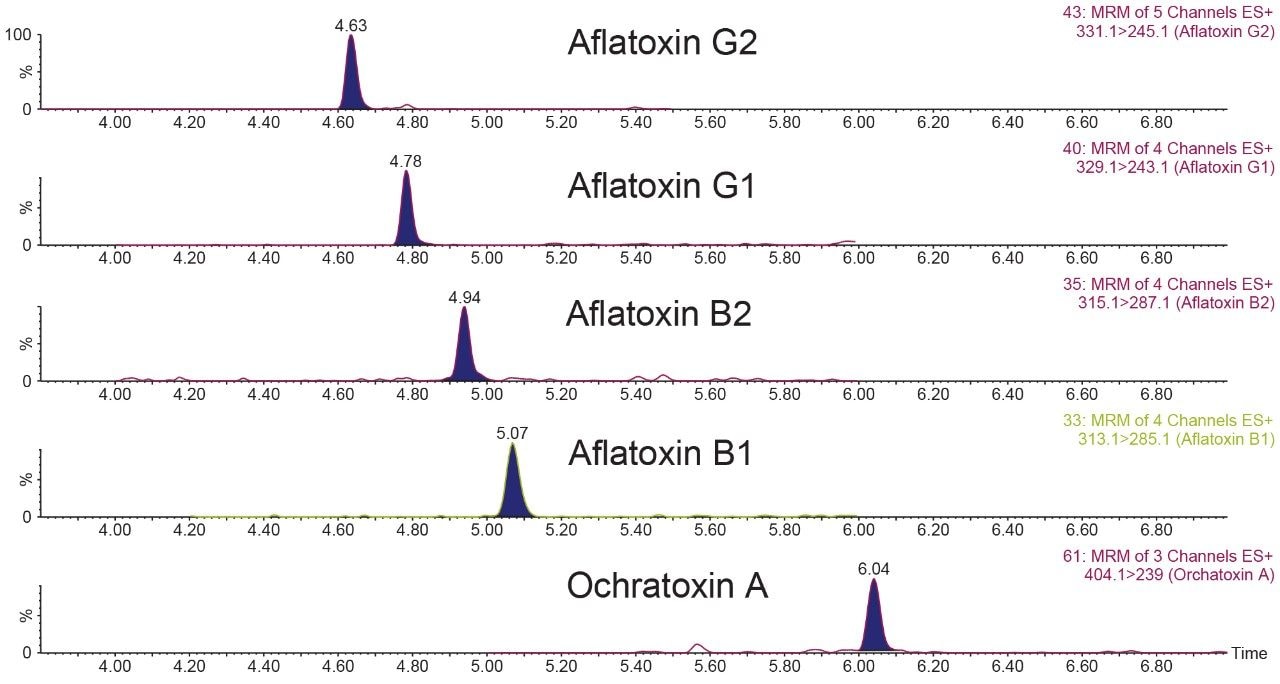
The LC-MS/MS analysis of mycotoxins can be combined with the analysis of pesticide residues in a single analytical injection, allowing trace level detection of aflatoxins G2, G1, B2, B1, and ochratoxin A. The calibration curves for all mycotoxins were linear (R2>0.990) over the range tested 0.005 to 0.10 µg/g (5 to 100 ppb in sample or 0.5 to 10 ppb in vial concentration) in matrix matched calibration curves. Figure 5 shows the chromatograms of cannabis matrix spiked at 0.02 µg/g which is the action level set by the State of California for mycotoxins testing.
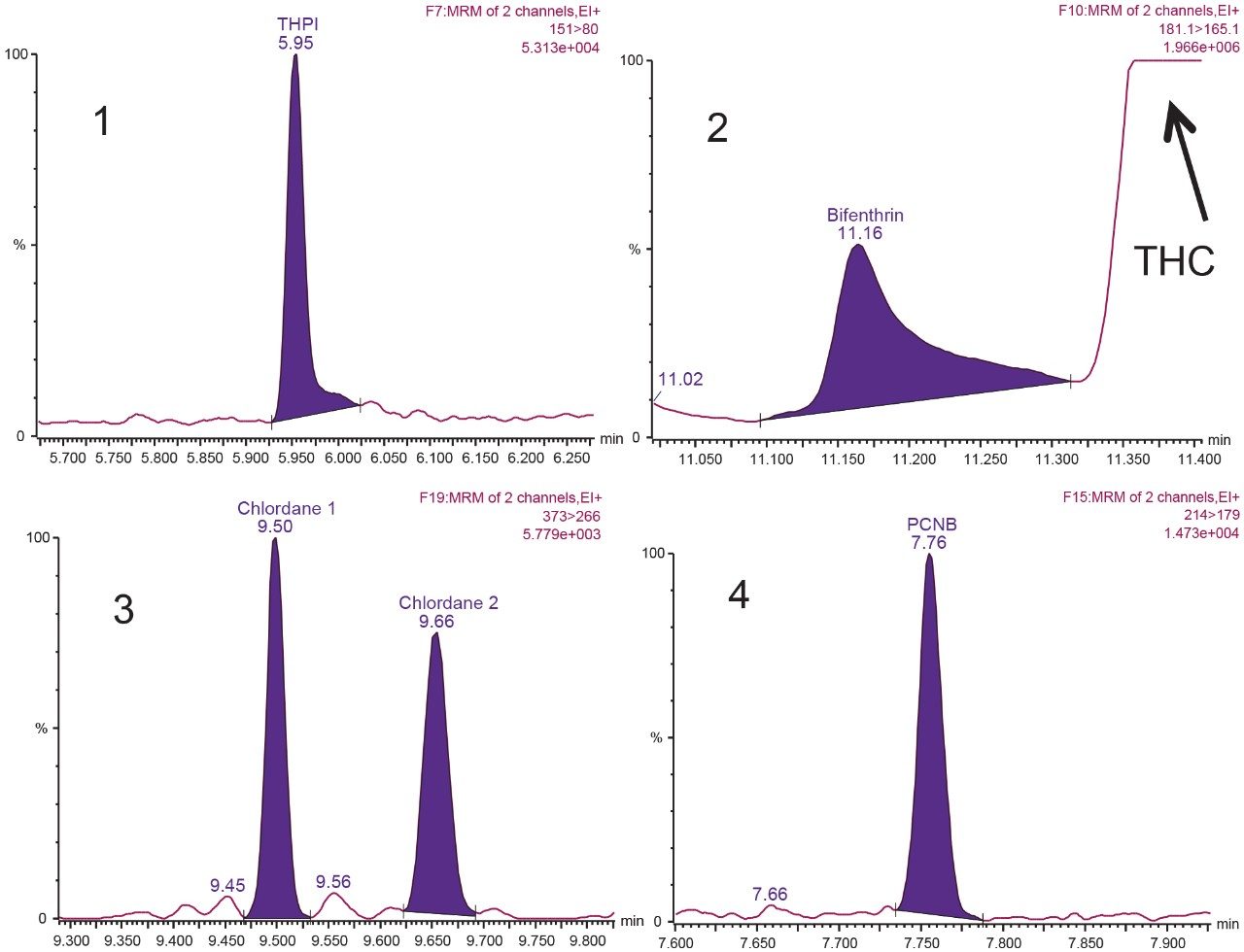
Analysis for pesticide residues in the cannabis flower extracts also required GC-MS/MS to fully cover the California pesticide regulations. Compounds like chlordane, captan (analyzed as its degradant THPI), and pentachloronitrobenzene (PCNB) require GC-MS/MS due to poor ionization using electrospray ionization in LC-MS/MS. Conversely, compounds such as bifenthrin ionize well using both LC-MS/MS and GC-MS/MS, but experienced matrix interference to reach the required levels using LC-MS/MS. Additionally, there was a large subset of compounds that worked well using both techniques. Therefore, analysis on both systems allows for increased confidence in results and the GC-MS/MS data can be used as an added confirmatory technique.
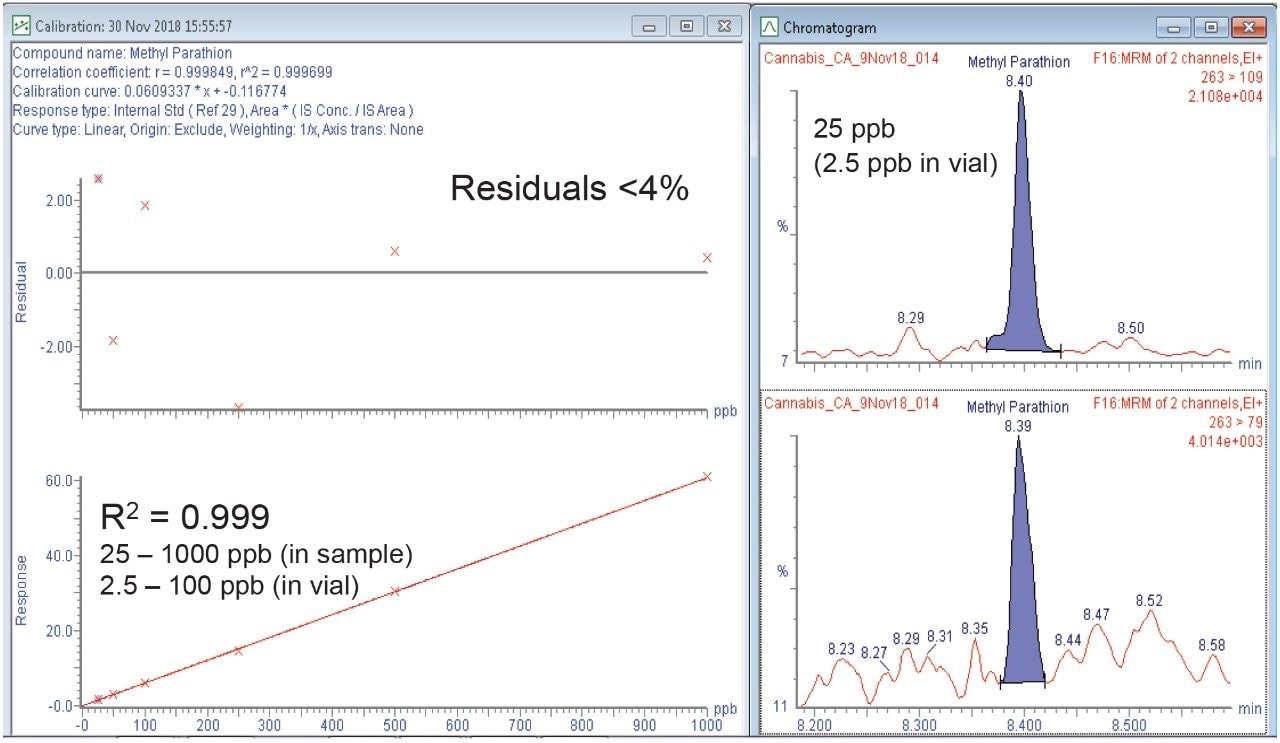
Matrix matched calibration curves were bracketed around injections of sample extracts. Continuing calibration checks were also run throughout the analysis sequence to ensure system performance was maintained through the runs. Linearity over the range of 0.025 to 1 µg/g (25 to 1000 ppb) was excellent with R2 values >0.995 and residuals were within 20%. Figure 6 highlights an example of methyl parathion post spiked into the cannabis flower extract showing excellent linearity, residuals, and sensitivity. Figure 7 demonstrates an example of the pesticides that required GC-MS/MS analysis at the action limit extracted in cannabis flower samples. It is important to note that bifenthrin elutes close to the region where THC and other cannabinoids elute and therefore its peak shape is broadened due to the matrix.
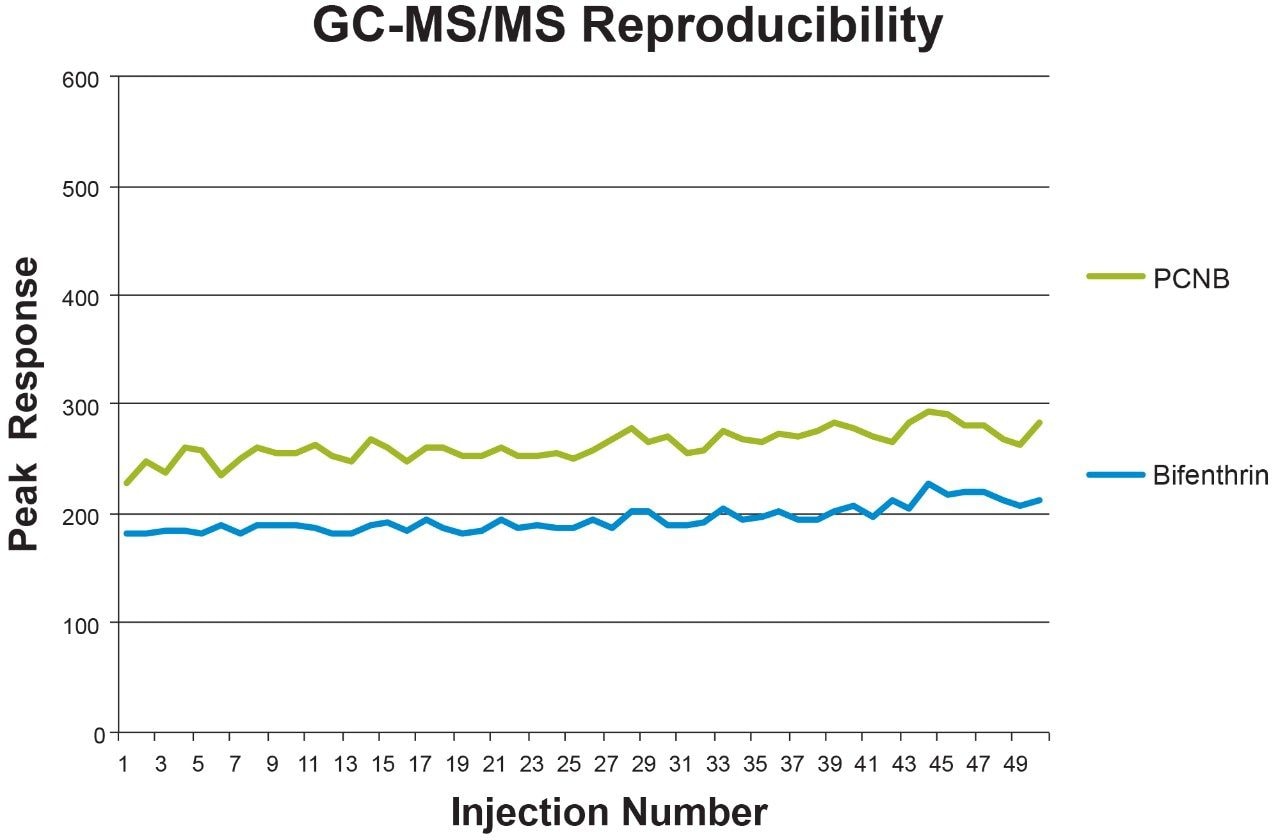
Cannabis is a very challenging matrix that can quickly create a loss of response from the inlet liner and source components becoming dirty. It is essential to have a system that is robust enough to hold up to challenging matrices such as cannabis. Reproducibility of the GC-MS/MS response was assessed by injecting 50 replicates of cannabis matrix post spiked with the California pesticide suite at 0.1 µg/g. Figure 8 highlights three of the GC pesticides analyzed over the series of injections. The %RSD of the peak response were below 15% for the majority of the pesticides across the 50 replicate injections.
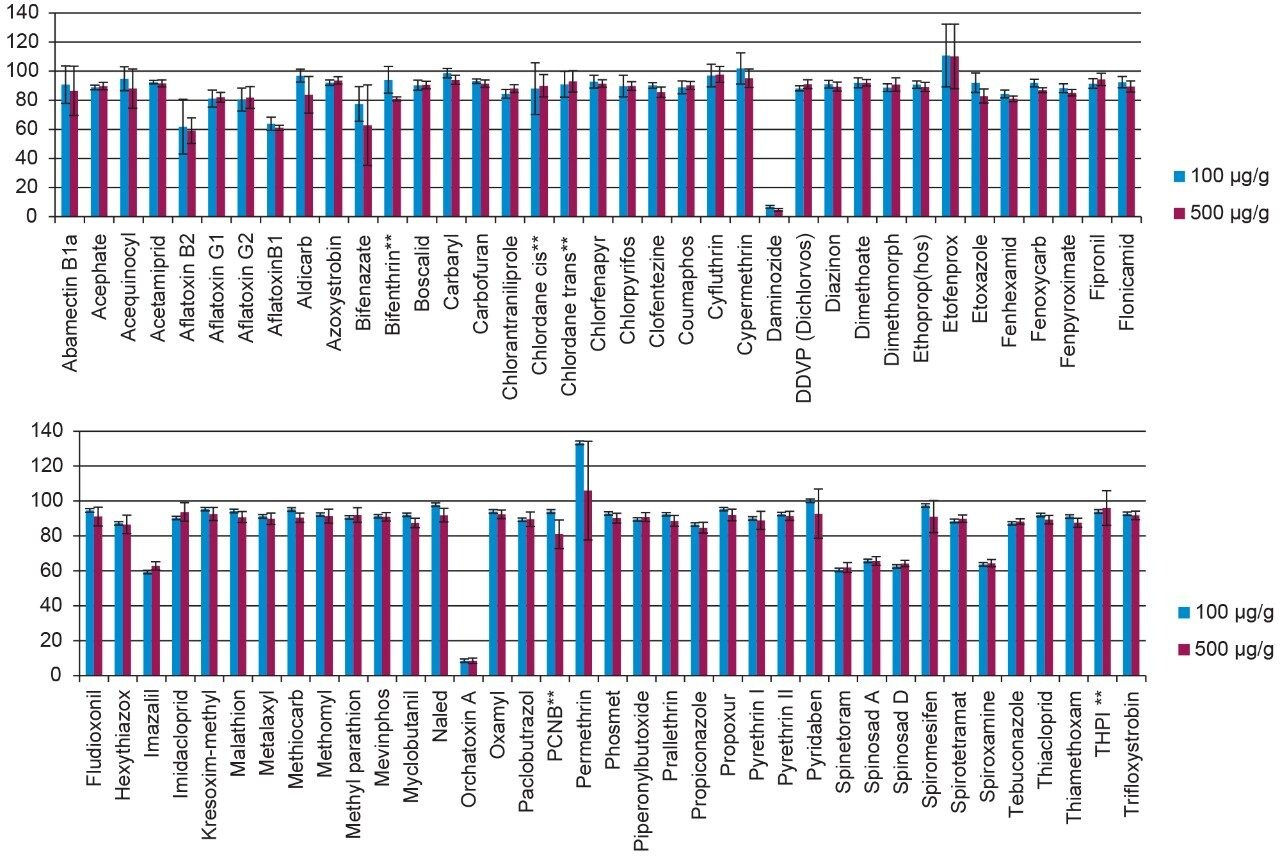
Method recovery was assessed by spiking pesticides at the 0.1 µg/g and 0.5 µg/g levels in cannabis flower and comparing the response to that observed from spiked matrix blanks (matrix matched standards). The mycotoxins were spiked at 0.02 µg/g and 0.10 µg/g. As shown in Figure 9, the recoveries observed for most pesticides were in the range of 80% to 120%. The dSPE cleanup provided significant reduction of matrix suppression for most compounds (data not shown). An example of the benefits of using dSPE to reduce suppression effects can be seen in a previous study using the Oregon list pesticides.4 Compounds that eluted in the same range as the cannabis resin constituents such as THCA show greater ion suppression compared to compounds that eluted before the resin constituents.
This simple sample extraction and dSPE cleanup method followed by UPLC-MS/MS and GC-MS/MS analysis provides a rapid, sensitive, and robust workflow for determination of the California pesticide list and mycotoxins in challenging cannabis matrix. Matrix suppression was significantly reduced using dSPE cleanup for many pesticides; thereby improving the data quality. This method is capable of meeting the action levels for the California pesticide list and mycotoxins in cannabis matrix.
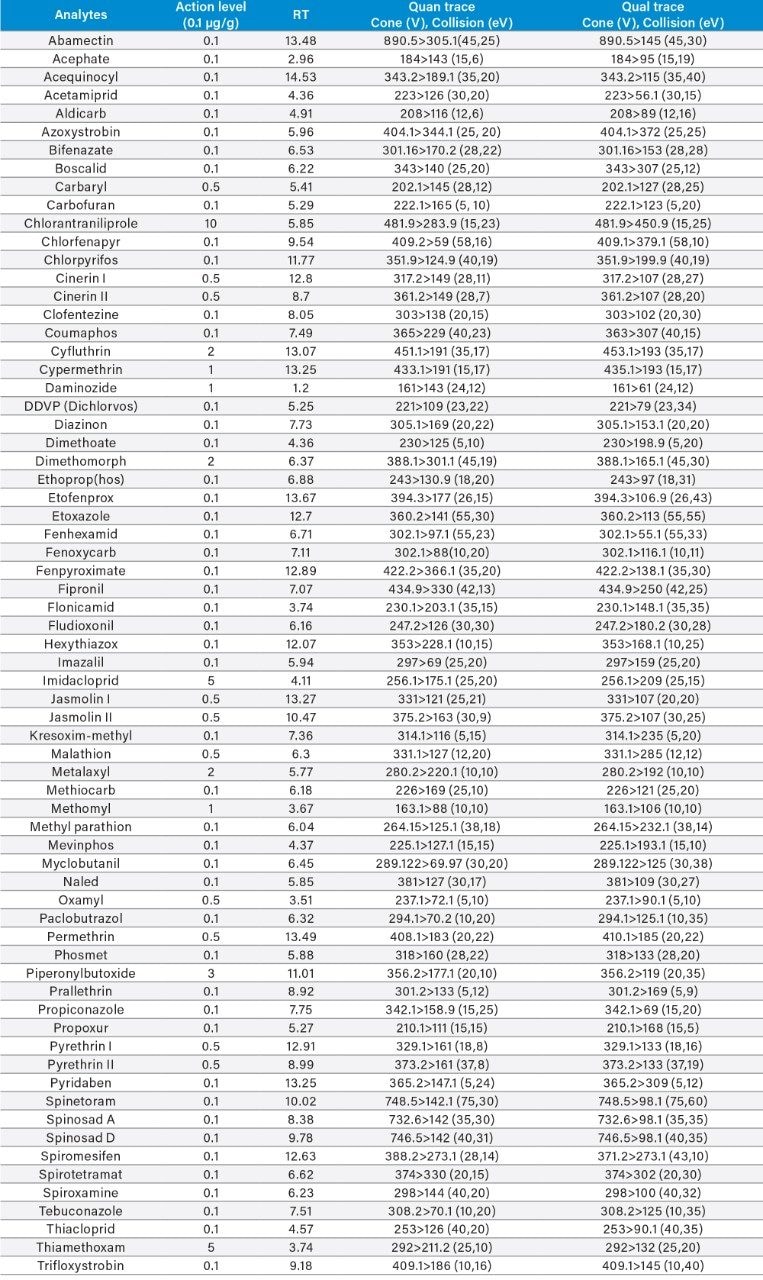
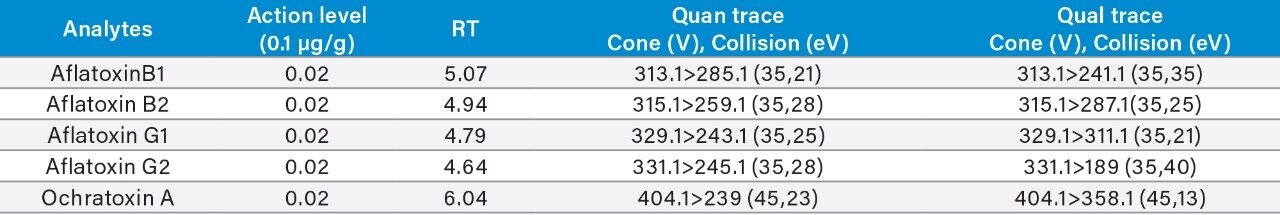
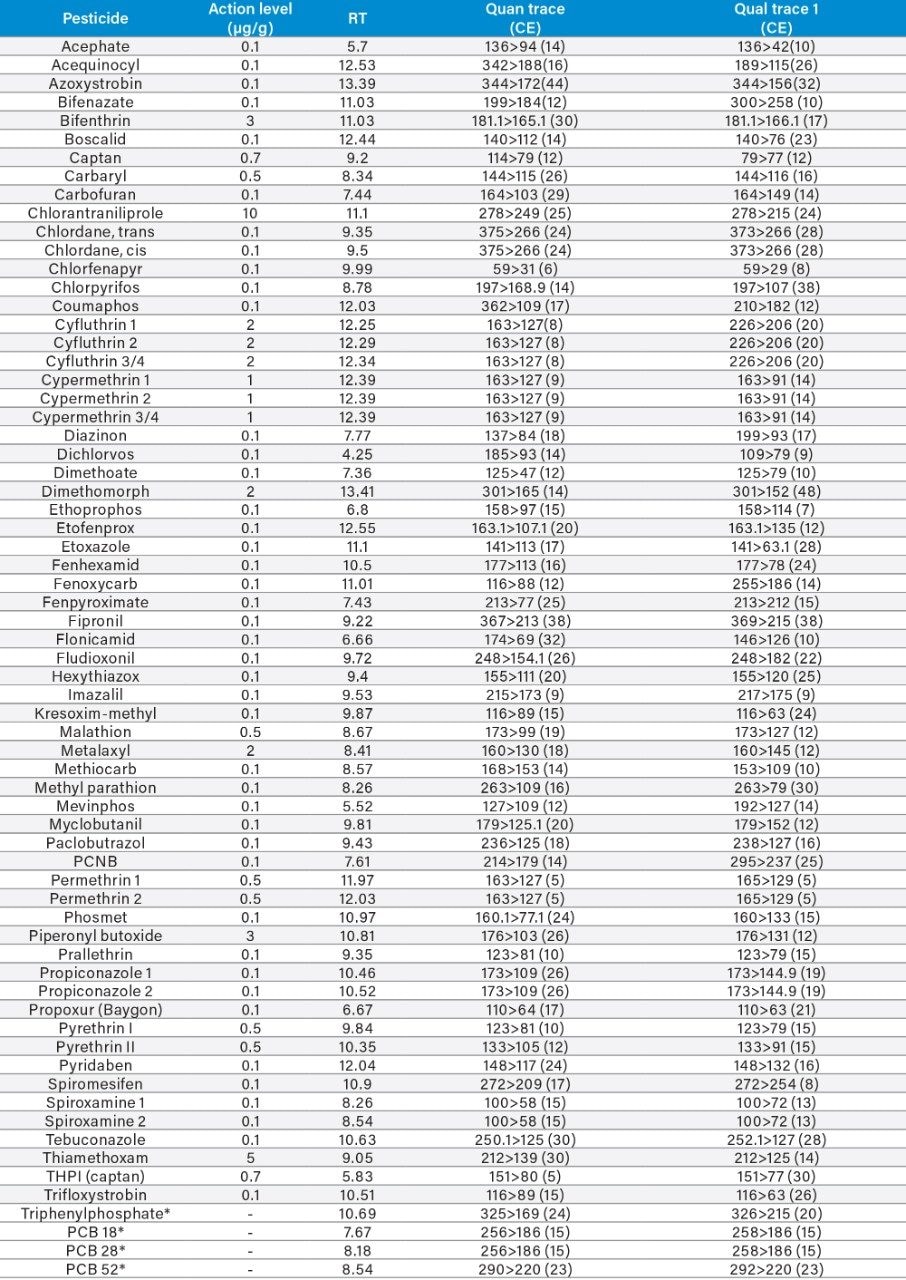
Appendix Table 3. GC-MS/MS retention times and MRM transitions for the GC amenable pesticides on the California list.
* Indicates compound used as internal standard.
720006465, Revised April 2021