
This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates an easy method setup for targeted analysis of pesticides in green tea, as well as concurrent MRM/full scan acquisition for monitoring of matrix co-extractives.
Concurrent MRM and full-scan data acquisition using RADAR Technology allows for the identification of co-extractives with minimum impact on the sensitivity of the method.
Tea represents one of the most widely consumed beverages in the world, and pesticide analysis of teas is legally required to ensure consumer safety.1 GC-MS/MS analyses using highly selective and specific MRM transitions are required for full coverage of all GC amenable pesticides at the trace levels required for many different method types. As green tea is a complex matrix, co-extractives, or matrix interferences, may impact MRMs. In this technical note, we demonstrate an easy method setup for targeted analysis of pesticides in green tea, as well as concurrent MRM/full scan acquisition for monitoring of matrix co-extractives.

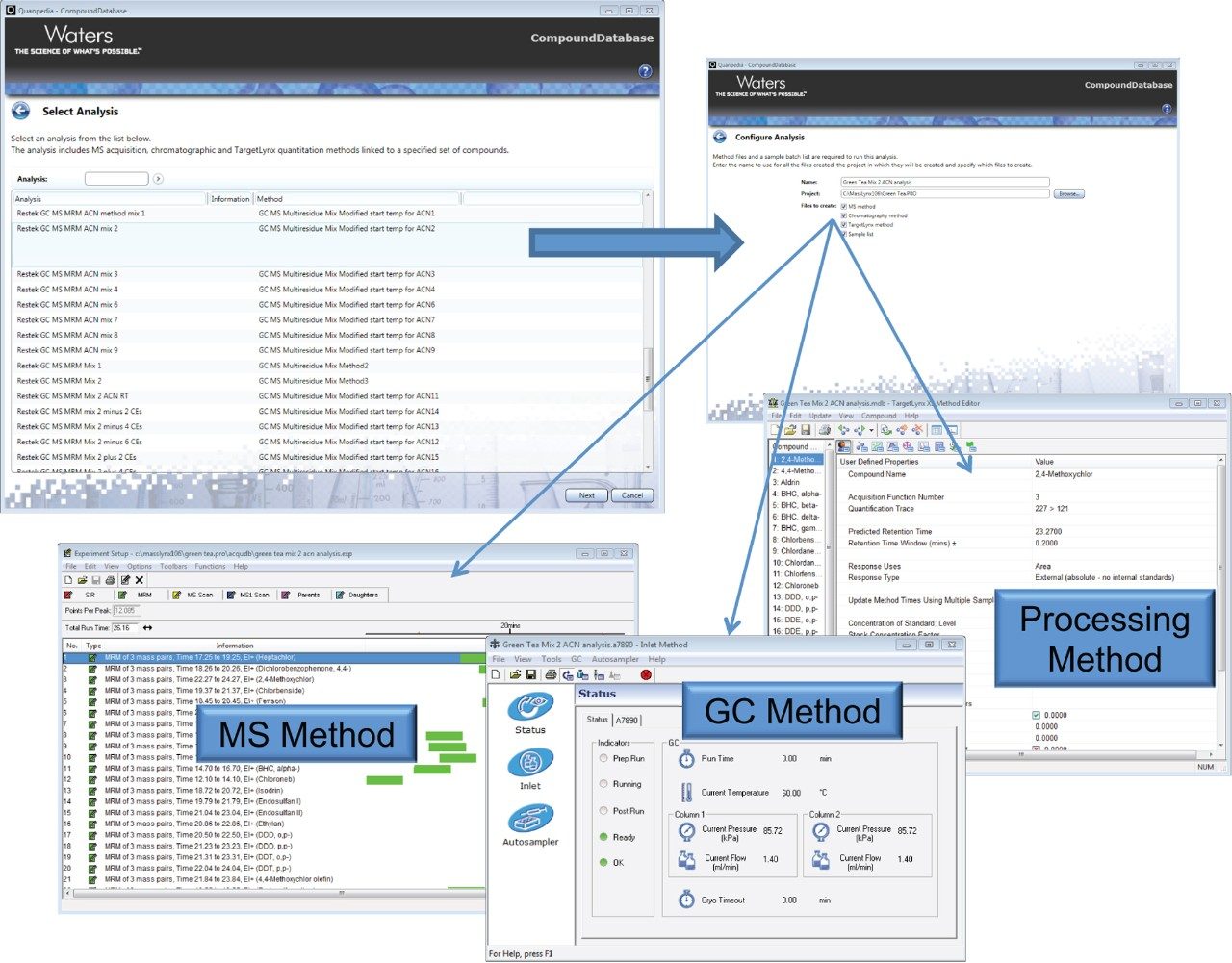
Green tea samples were prepared using Waters DisQuE QuEChERS CEN extraction followed by solid phase extraction (SPE) pass-thru clean-up using Oasis PRiME HLB. Following extraction and clean-up, a matrix matched calibration curve and QC samples were prepared for 1 µL injections using a splitless injection mode. Data were acquired using EI+ at a potential of 70 eV. Concentration determination is described here as both ppb (spike into volume of final extract, i.e. matrix matched), and mg/kg in tea, which is 0.005 × the ppb value, based on the sample preparation used (2 g of tea reconstituted as per QuEChERS extraction). The GC temperature program, MRM transitions and processing method were automatically generated using Quanpedia (Figure 1) which eliminated the potential of transcription error on input of the GC and MRM conditions. Utilizing Quanpedia also eliminates the need for extensive method development and simplifies the workflow for routine analysis.
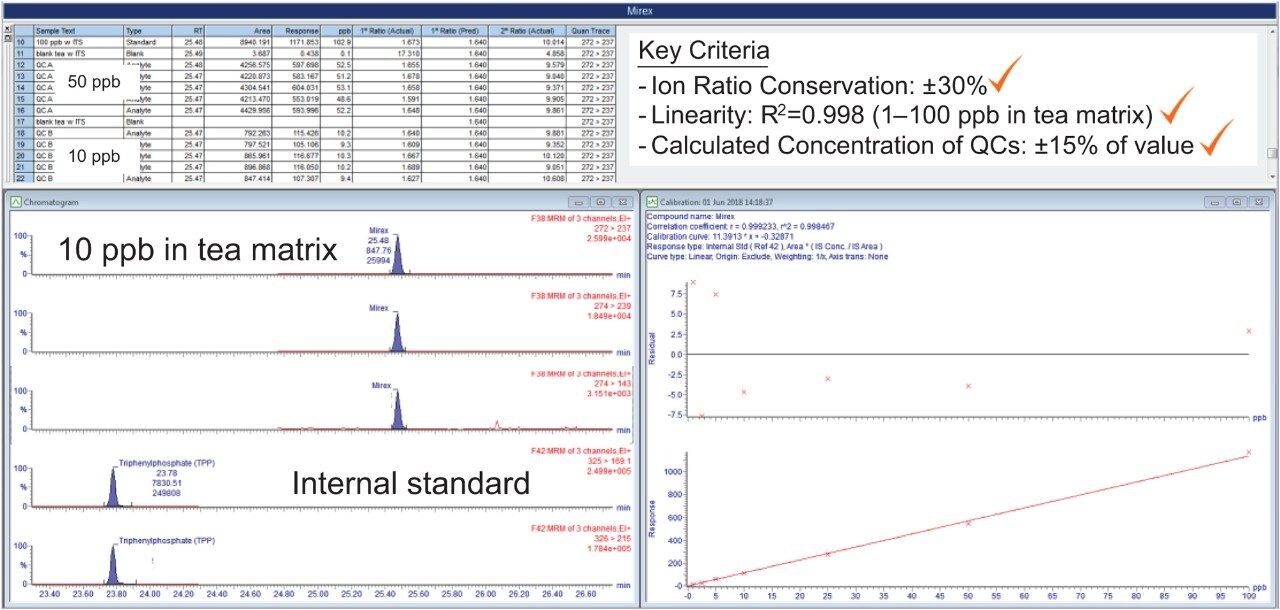
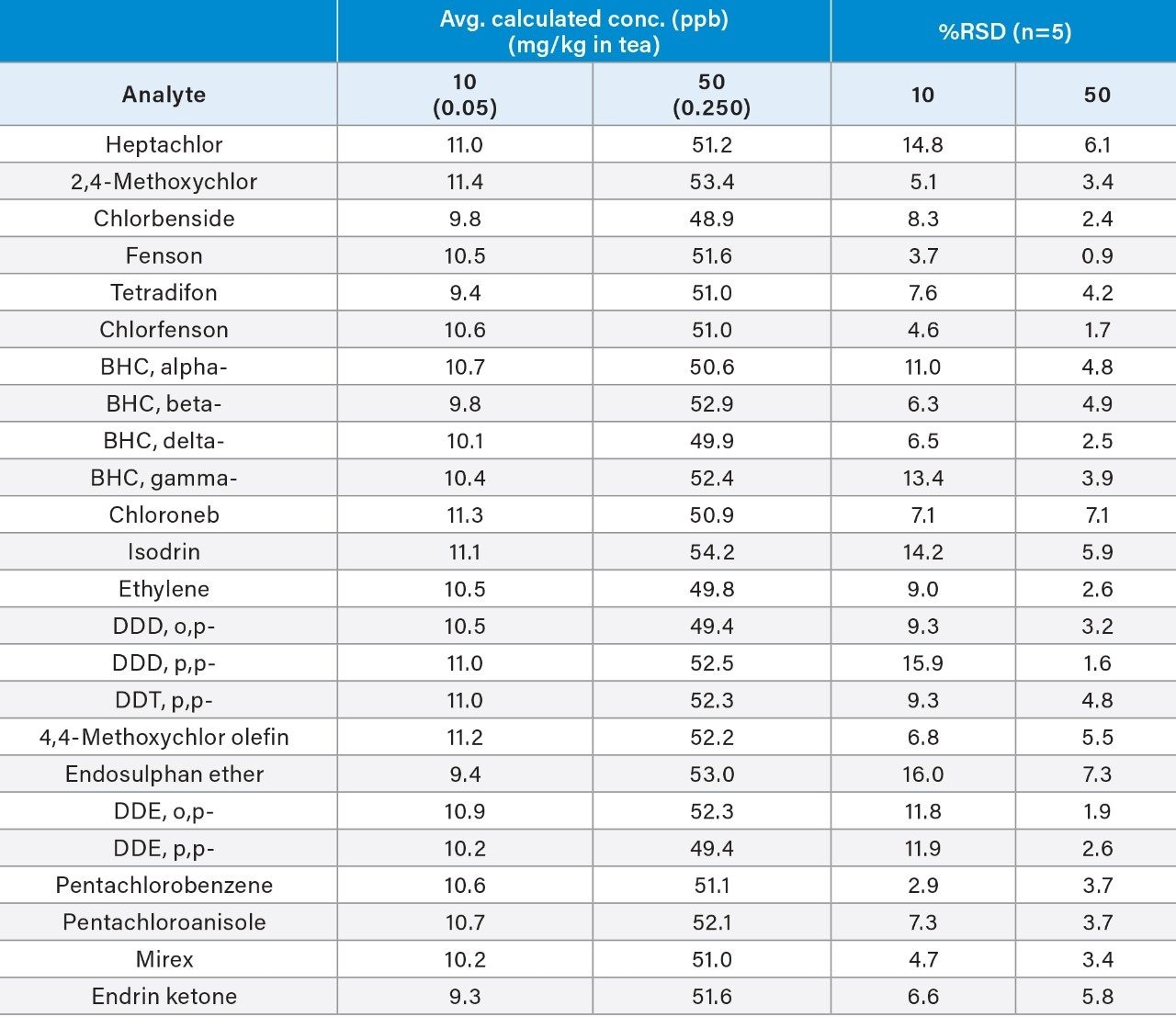
In order to assess the Xevo TQ-GC System’s ability to meet regulatory recommendations, matrix matched calibration curves were prepared. As shown in Figure 2, the calibration curve and the QC injections met the key criteria of ion ratio conservation of ±30%, linearity of R2 ≥0.998 (1 to 100 ppb, which calculates to 0.005 mg/kg to 0.5 mg/kg in tea matrix), back calculated residuals of <20%, and and calculated concentrations of the QCs within ±15% of the true value. Table 1 provides a summary of the QC sample data for various organochlorine pesticides.
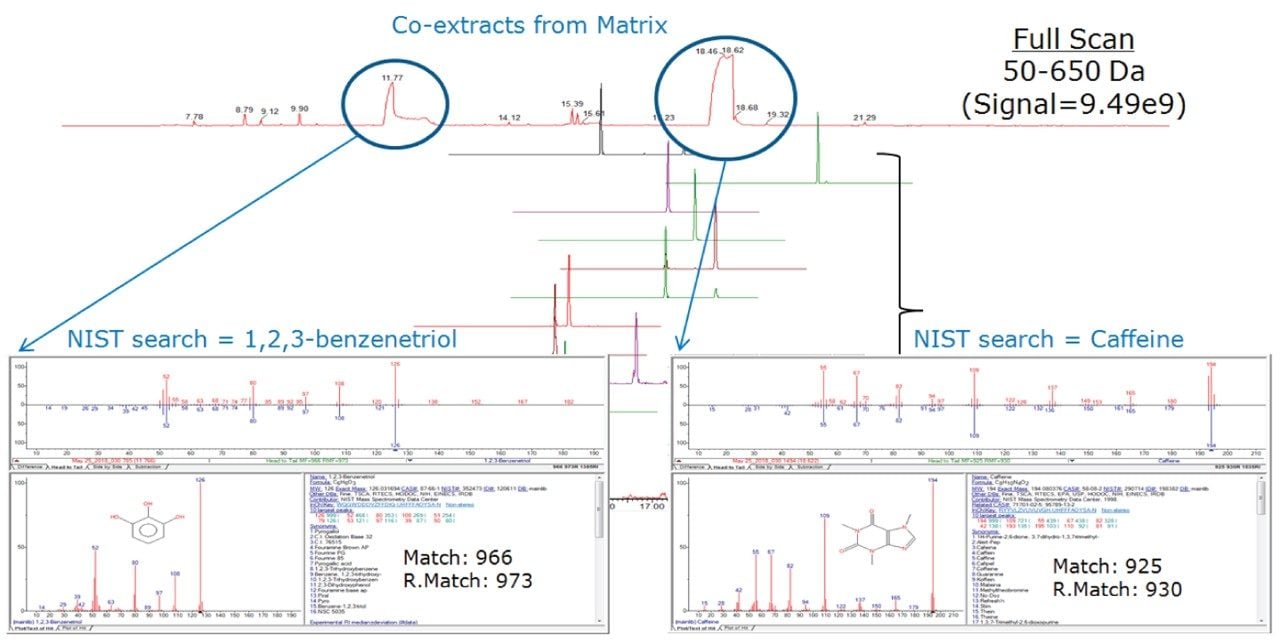
Following quantitative demonstration of the targeted MRMs, the use of concurrent MRM and full scan (RADAR) data was assessed. The full scan acquisition was from m/z 50–650 with a scan time of 0.5 sec. Green tea sample extracts were spiked at 0.1 mg/kg with more than 200 pesticides (in nine separate mixtures to prevent interactive effects as provided in the GC-MS Multiresidue Kit from Restek Corporation). The full scan data was interrogated to determine the identity of two large matrix interference peaks. In Figure 3 the identification of 1,2,3-benzenetriol and caffeine are shown using the NIST Library. Caffeine is a known stimulant which occurs naturally in green tea, and the phenolic compound 1,2,3-benzenetriol (also referred to as pyrogallol or pyrogallic acid) is also found in green tea.2
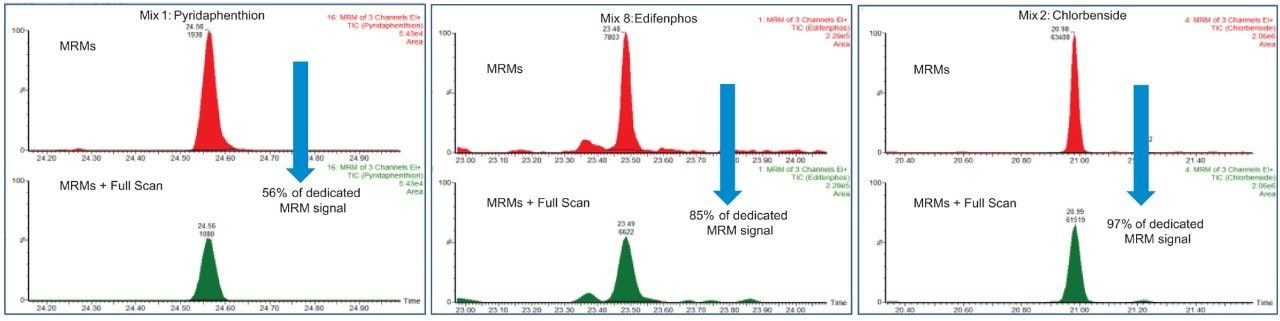
Using the same samples, an assessment of the loss in signal due to the combined acquisition of full scan and MRM was also performed. This was achieved by running one spiked sample with just MRM followed by the same sample with RADAR enabled. The loss of signal is dependent on the number of transitions contained in the method and when the transitions occur relative to one another. Figure 4 shows that the signal conservation when using RADAR ranged from 56 to 97% for the three illustrated compounds, representing the approximate range observed for the pesticides across mixes. This means that low limits of detection can still be achieved when utilizing RADAR which allows further sample information to be analyzed.
The Xevo TQ-GC has simplified workflows that enables users to produce results that meet regulatory recommendations. The added functionality of RADAR allows full scan and MRM data to be collected simultaneously. This data can be interrogated to identify change to understand extraction interferences at the time of the original analysis, or as part of a retrospective data mining investigation. The combination of MRM and Full scan in a single method acquisition does not impact sensitivity and is valuable for use in method optimization.
720006377, September 2018