
In this feasibility study, a method for botanical authentication was developed using the MS data collected by the ACQUITY QDa Mass Detector. The accuracy of this authentication method was evaluated in a blind test with four commercial black cohosh samples. The advantages of MS over using a evaporative light scattering detector (ELSD) are also highlighted.
Botanical ingredients are widely used in dietary supplements, herbal medicines, cosmetics, and personal care products. Potential contamination or misidentification of plants has been a health concern due to the lack of standardization of production. The testing of botanical ingredients and processed products for authenticity is a challenging task due to their complex phytochemical constituents, the variation in chemical profiles from different locations, plant anatomy, age and harvest season, and the similar phytochemical profile of closely related species. Many analytical techniques are used in botanical authenticity testing.1 Liquid chromatography (LC) coupled with mass spectrometry (MS) is one of the most effective tools.2-7 However, LC-MS has not been widely employed in analytical labs for routine authenticity testing due to the relatively high cost of mass spectrometers and the high level of expertise required.
Waters ACQUITY QDa Mass Detector is an affordable and easy-to-use mass spectrometer designed for chromatographers. It offers a practical solution for bringing LC-MS to the routine analysis lab environment. In this application note, the feasibility of using mass detection for authenticity testing is demonstrated in a study for North American (NA) black cohosh (Actaea racemosa). In this feasibility study, a method for botanical authentication was developed using the MS data collected by the ACQUITY QDa Mass Detector. The accuracy of this authentication method was evaluated in a blind test with four commercial black cohosh samples. The advantages of MS over using a evaporative light scattering detector (ELSD) are also highlighted.
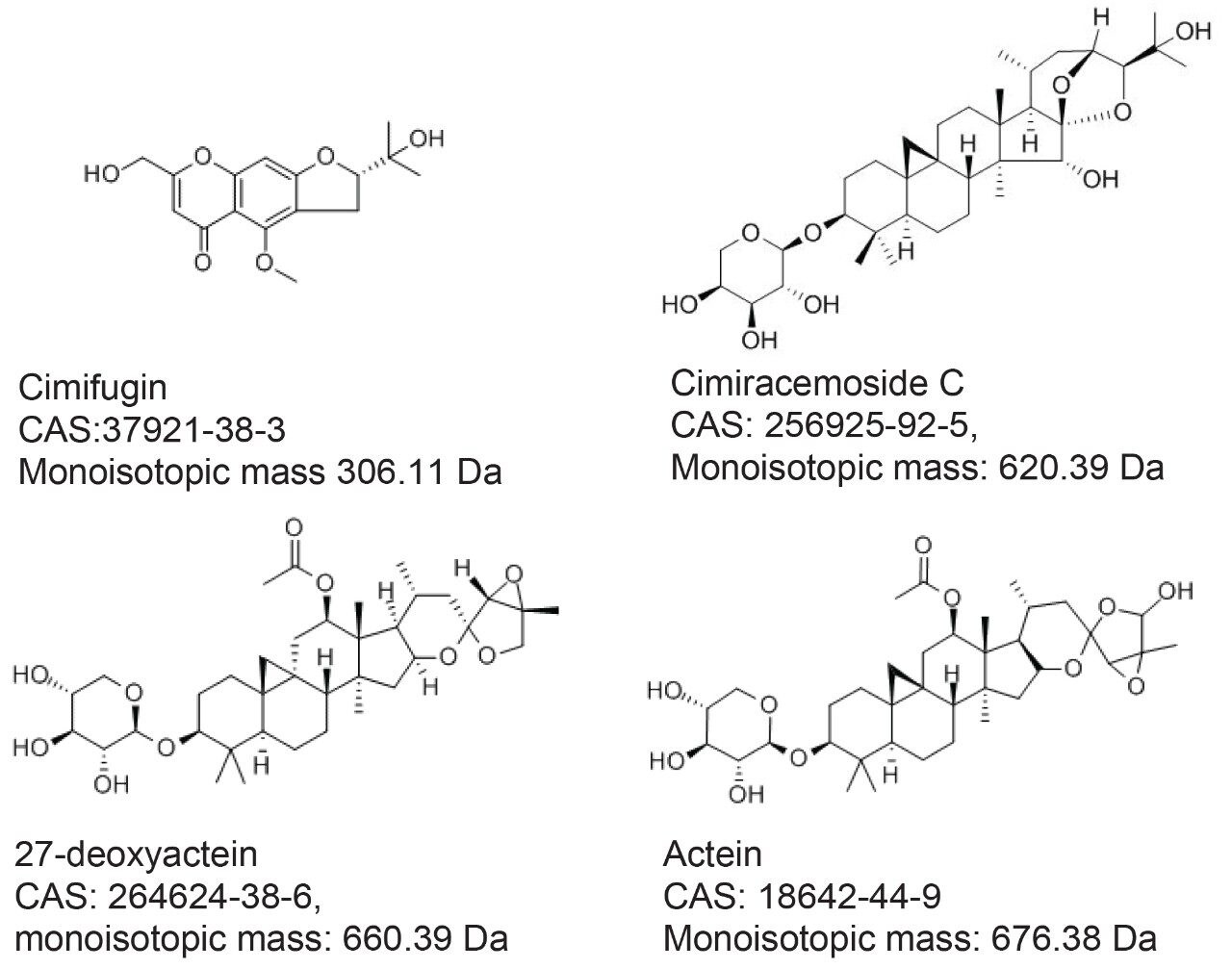
Three authentic NA black cohosh, Actaea racemosa extracts (NA1-3), three Asian black cohosh, Actaea cimicifuga extracts (A1-3), and four commercial black cohosh samples (U1-4) were provided by a collaborator. These extracts were diluted with 70% methanol to about 5 mg/mL. Four standards: cimifugin, cimiracemoside C (cimigenol-3-alpha-L-arabinoside), 27-deoxyactein (23-epi-26-deoxyactein), and actein were purchased from ChromaDex (Irvine, CA). These standards were prepared in 70% methanol at about 5 μg/mL. The standard structures, CAS Registry numbers, and monoisotopic masses are shown in Figure 1. Homemade black cohosh samples, M-5 and M-10, were prepared by mixing NA black cohosh sample (NA1) and Asian black cohosh (A1) at 95:5 and 90:10 mass ratios, respectively. Sample solutions were filtered by 0.2 micron PTFE membrane prior to the analysis.
|
UPLC system: |
ACQUITY UPLC H-Class |
|
Column: |
ACQUITY BEH C18, 1.7 μm, 2.1 × 100 mm, (p/n 186002352) |
|
Column temp.: |
50 °C |
|
Eluent A: |
De-ionized water with 0.1% formic acid |
|
Eluent B: |
Acetonitrile/methanol (v/v 7/3) with 0.1% formic acid |
|
Injection volume: |
10 μL |
|
Run time: |
9 min |
|
Reconditioning: |
2.5 min |
|
MS system: |
ACQUITY QDa (with Diverter Valve) |
|
Software: |
Empower 3 |
|
Detection: |
ESI+, MS scan |
|
Scan: |
200 to 1000 Da |
|
Capillary voltage: |
1.5 kV |
|
Cone voltage: |
10 V |
|
Probe temp.: |
300 °C |
|
Sampling rate: |
5 Hz |
|
Diverter valve events: |
Switch on/flow to QDa at 0.8 min; switch off/flow to waste at 9 min |
|
Detector: |
2424 ELS |
|
Gain: |
250 |
|
Pressure: |
45.0 psi |
|
Drift tube temp.: |
55 °C |
|
Nebulizer: |
Heat at 50% power level |
|
Data rate: |
10 pps |
|
Filter time constant: |
Normal |
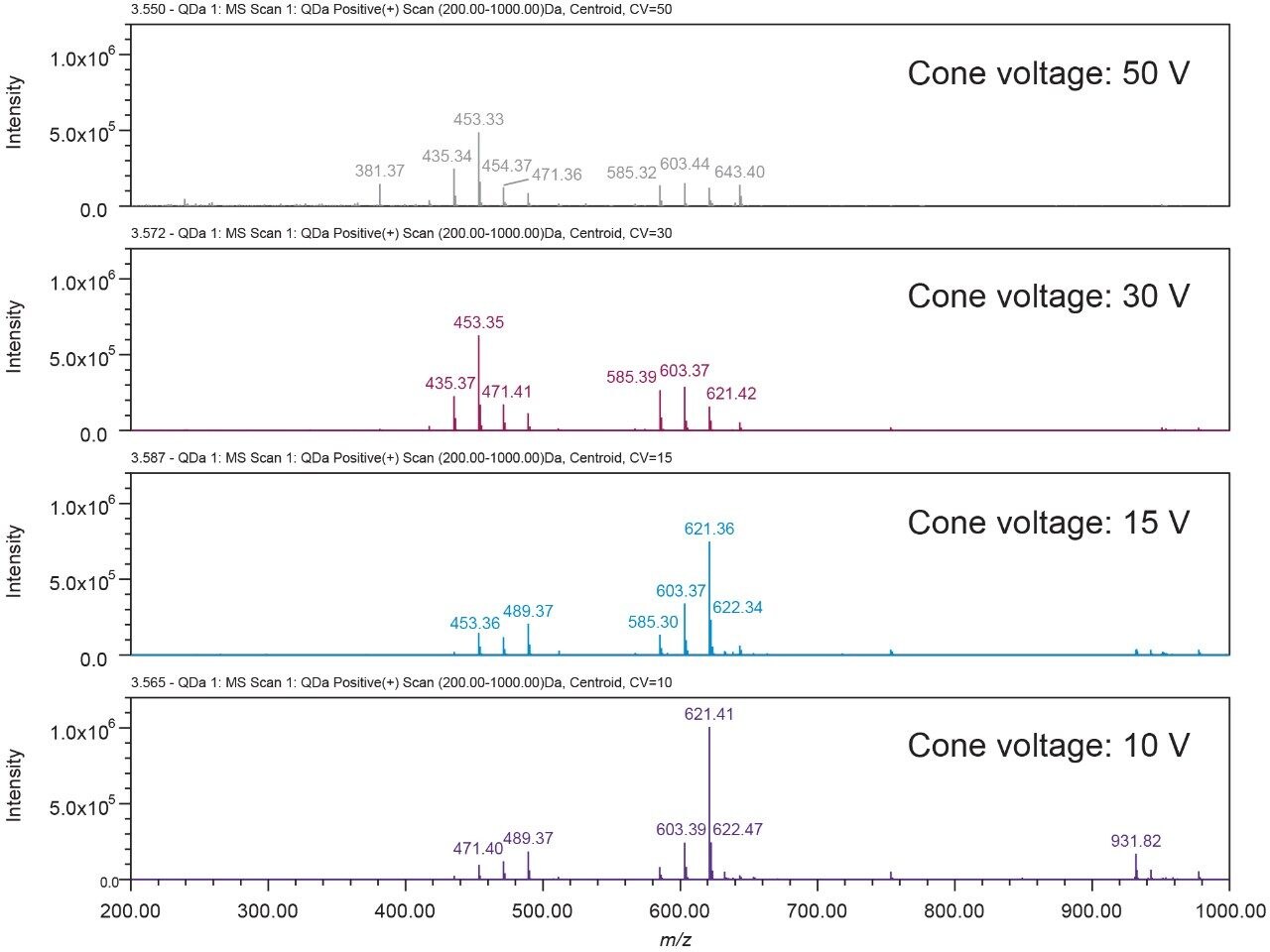
The ACQUITY QDa Mass Detector’s default setting is suitable for many applications. For this study, in order to get the maximum molecular ion intensity, the MS instrument parameters, such as the probe temperature, capillary voltage, and cone voltage were optimized. Figure 2 shows the effects of the cone voltage on the cimiracemoside C mass spectrum. The MS parameters that generated the maximum molecular ion intensity were selected and used, as described in the Experimental section. UPLC conditions6 were used in this study with minor optimization.
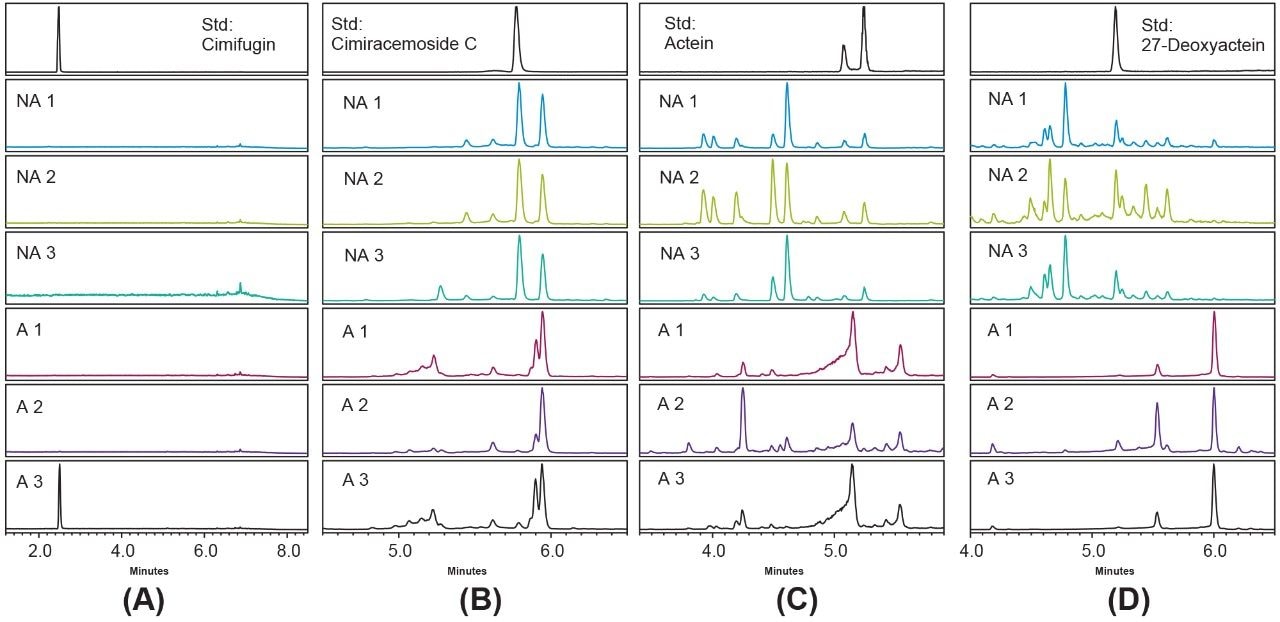
Authentic black cohosh, or NA black cohosh, is manufactured from the roots and rhizomes of Actaea racemosa. The potential contamination or misidentification of the black cohosh plant has been a health concern for many years due to a lack of standardization of production. Adverse events of hepatotoxicity associated with the use of black cohosh products have been reported, and it was suspected that unauthentic black cohosh might have contributed to some of the incidents.8 Contamination or misidentification often occurs with the Chinese species of Actaea such as A. heracleifolia, A. dahurica, and A. cimicifuga, and NA Actaea species growing in the same area as NA black cohosh, such as A. pachypoda, A. rubra, and A. podocarpa. Many chemical constituents of black cohosh have been used as biomarkers for authenticity testing.2-6 Among these markers, the most common ones, such as cimifugin, cimiracemoside C, actein, and 27-deoxyactein, were screened in this UPLC-MS study for suitable marker(s) to be used with the ACQUITY QDa Mass Detector (see Figure 3).
The molecular ions ([M+H]+) of cimifugin, cimiracemoside C, and 27-deoxyactein, and the dehydration ions ([M+H-H2O]+) of actein were the dominant ions (base peak) in their respective mass spectra (data not shown). The mass-to-charge ratios (m/z) of these base peak ions were used to extract the ion chromatograms from the MS scan data, respectively. The resulting extracted ion chromatograms (XICs) are shown in Figure 3. After inspection of the XICs in Figure 3, the cimiracemoside C (m/z 621 Da, retention time 5.8 min in Figure 3B) was selected as the marker for NA black cohosh because it showed the most simple and distinctive chromatographic pattern in the NA black cohosh as compared to the pattern from the Asian black cohosh.
In order to make this authenticity method suitable for routine analysis, a new authenticity data processing protocol was designed. In this protocol the marker’s distinctive chromatographic and mass spectral patterns, or fingerprints are used to evaluate sample authenticity (Figure 4). This authenticity data processing protocol was implemented in Empower 3 CDS Software using the existing functions. No special chemometric software program was used. Details of how this protocol was developed and implemented in Empower 3 Software are discussed in a second application note.9
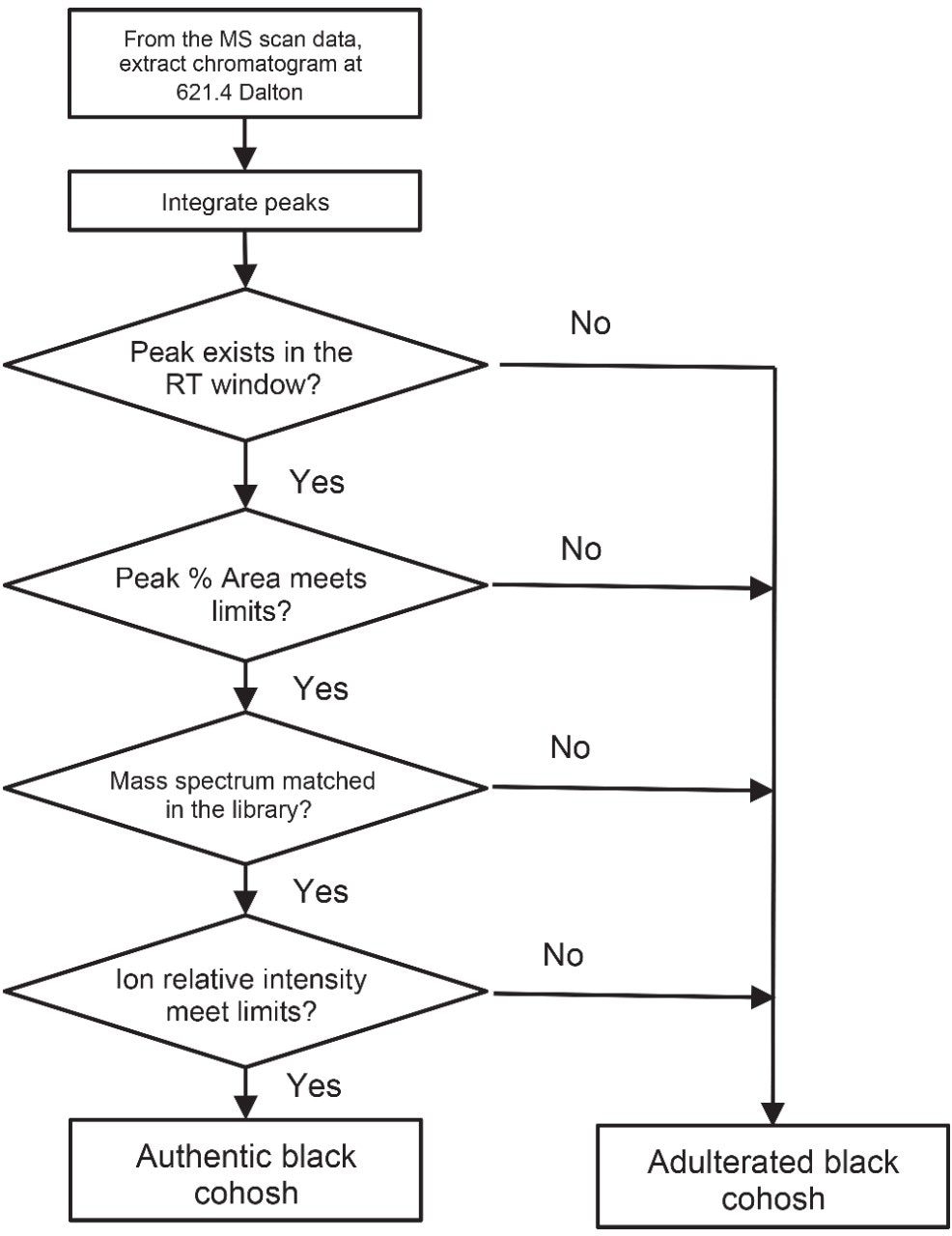
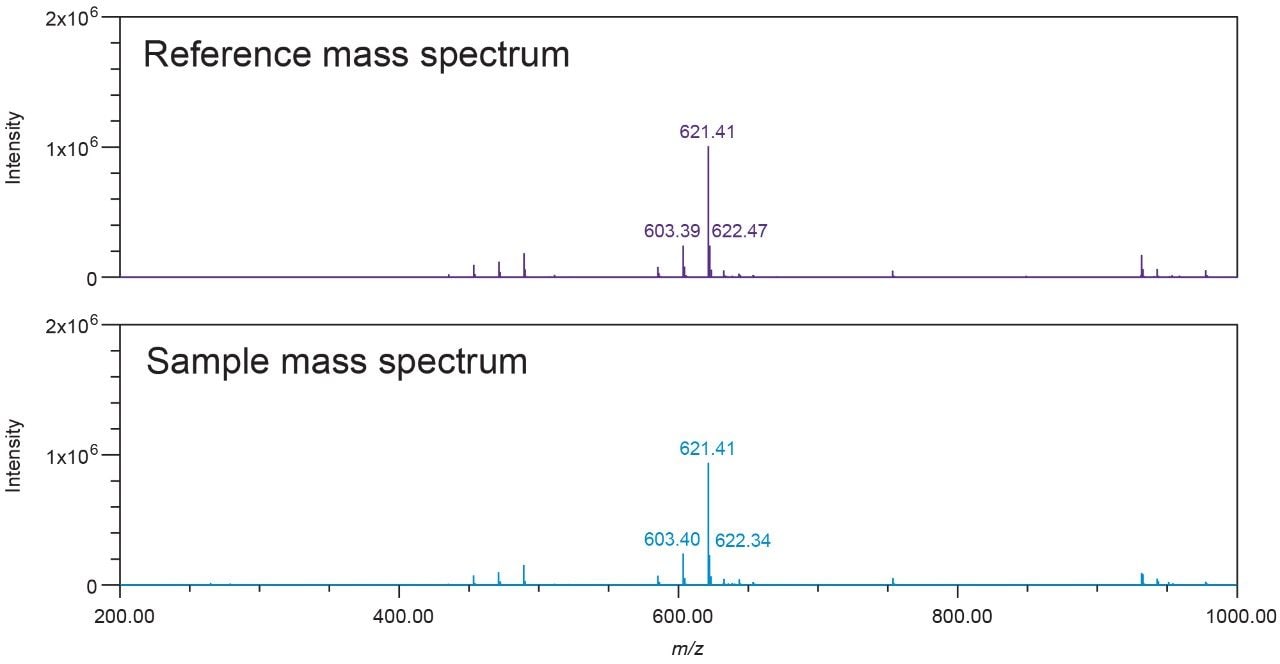
To support this authenticity data processing protocol, a black cohosh MS library was created in Empower 3 Software. The cimiracemoside C mass spectra from the NA black cohosh samples were extracted (with baseline subtraction) from the MS scan data, and stored in the NA black cohosh MS library. Empower's MS Library Match function was used to check whether a sample’s mass spectrum matched with any of the reference mass spectra in the library. An MS library can be custom built in Empower 3, exported and imported. Figure 5 shows an MS Library Match result. UV/Vis spectra can also be used for authenticity testing. However, UV/Vis spectra are usually less specific compared to mass spectra. In this case, the cimiracemoside C is UV transparent. So, it is not useful to use the UV/Vis spectral library for black cohosh authenticity testing.
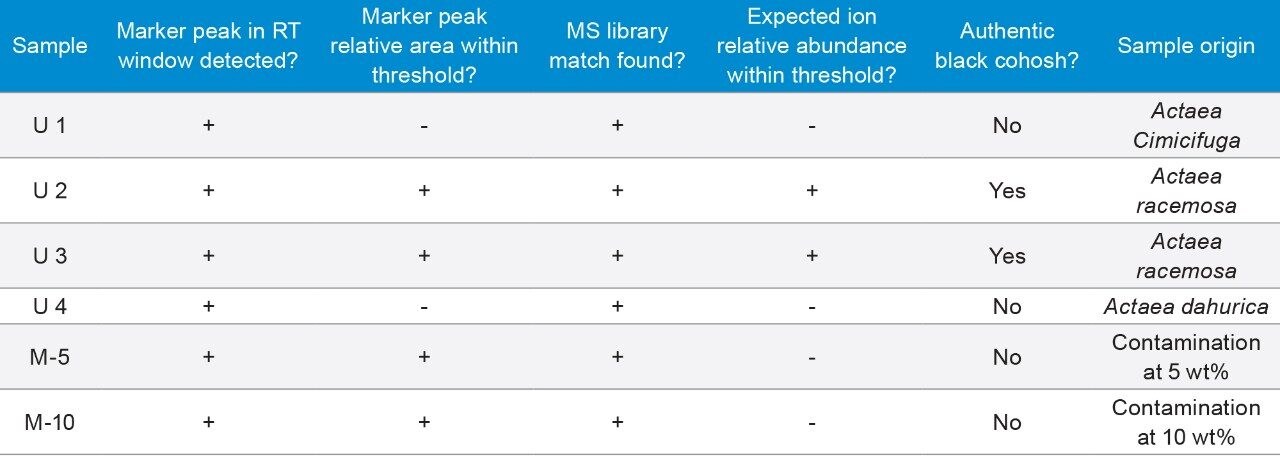
Four commercial black cohosh samples (U1-4) and two homemade black cohosh samples (M-5 and M-10) were tested by this method. The commercial sample origins were revealed after the evaluation was completed. Table 2 is a summary of the authenticity test results for these samples. The authentic NA black cohosh (U2 and U3) and the inauthentic black cohosh (U1 and U4) samples were correctly determined. The contaminated samples were also correctly detected, including the 5 wt% contaminated black cohosh sample (M-5). Details of how the homemade black cohosh samples were prepared can be found in the Experimental section.
Evaporative light scattering detector (ELSD) is commonly used for the black cohosh identification.10 A comparison of the MS and the ELSD data for authenticity testing was made with an NA black cohosh sample (NA 1), an Asian black cohosh sample (A1), and four unknown black cohosh samples (U 1-U 4), as shown in Figure 6. UPLC conditions were the same for all chromatograms. From the UPLC-MS chromatograms (Figure 6A), it was easily determined that Samples U2 and U3 were authentic black cohosh, and that Samples U 1 and U 4 were not. On the contrary, it was difficult to tell from the ELSD chromatograms (Figure 6B) which of the unknown samples was authentic black cohosh. Figure 6 clearly demonstrates that MS is a better technique for differentiating complex botanic samples than ELSD.
In this application note, a method for black cohosh authentication has been developed using the ACQUITY UPLC H-Class System and the ACQUITY QDa Mass Detector. Specifically, four black cohosh constituents: cimifugin, cimiracemoside C, 27-dexoyactein, and actein were screened, and cimiracemoside C was selected as a marker. A new automated data processing protocol that is suitable for routine authenticity testing was developed. In this data processing protocol, the marker’s chromatographic and mass spectral data were evaluated for authenticity. This authenticity method was tested with four commercial black cohosh samples and two homemade contaminated NA black cohosh samples. All authentic and inauthentic or contaminated samples were correctly identified. It should be noted that due to the limited number of training or reference samples used in the method development, this black cohosh authenticity method may need further improvement or validation.
This application note demonstrates that the ACQUITY QDa Mass Detector is well suited for botanical authentication, which is an essential test for product safety and quality. The cimiracemoside C ion chromatogram collected by mass detection had less interfering peaks, and showed clear differences between the NA and Asian black cohosh samples. The cimiracemoside C mass spectrum contained a characteristic mass spectral pattern that was used for authenticity testing. The ACQUITY QDa is simple to use and affordable, and it can be easily implemented in analytical QC labs for routine botanical authentication. The adoption of mass detection for authenticity testing can greatly improve the speed and confidence for botanical authentication, and help to ensure the safety and high quality of dietary supplements, herbal medicines, cosmetics, and personal care products on the market.
720006247, May 2018