In this application note, we demonstrate that the ACQUITY UPLC M-Class System with 2D Technology (300 μm I.D., high capacity configuration) is capable of providing high chromatographic resolving power required for the quantitative HCP analysis and the separation of other complex peptide samples.
To provide the experimental procedure used for testing the chromatographic performance of the ACQUITY UPLC M-Class System with a 300-μm ID configuration.
The ACQUITY UPLC M-Class System with 2D Technology (300-μm I.D., high capacity configuration) has been recently used for the analysis of low-abundance host cell proteins (HCPs) that are typically present in the concentration range of 1–100 ppm (ng/mg) in a highly-purified biotherapeutic protein.1-5 To successfully quantify these levels of HCPs, the dynamic range of the separation needs to be extended. In order to extend the dynamic range of the separation to detect and quantify low level HCPs, an orthogonal 2D reversed-phase (RP/RP) approach is used where sample is loaded at pH 10 and separated at pH 2.5.6
The microscale 2D-LC separation system consists of a 1st dimension reversed-phase (RP) column (XBridge Peptide BEH, C18, 300Å, 5 μm, 1.0 x 50 mm) for fractionation of the peptide mixture under basic conditions (pH 10), an ACQUITY UPLC M-Class Symmetry C18, 2D HCP Trap Column (5 μm, 300 μm x 25 mm) for retaining the peptides eluted in each fraction from the 1st dimension column, and an ACQUITY UPLC M-Class HSS T3 Column (100Å, 1.8 μm, 300 μm x 150 mm) analytical column operating under acidic conditions (pH 2.5) for high-resolution peptide separations (Figure 1). Fractions are eluted from the 1st dimension column using discontinuous step gradients of increasing organic concentration.
While a typical 2D-LC experiment involves 3, 5, or 10 fractionation steps, the sample complexity determined the number of elution steps required. As peptides elute from the 2nd dimension, a quadruple time-of-flight mass spectrometer is used to detect the peptides and their fragments by alternating collision cell energy between a low and elevated energy state (MSE data acquisition mode).
Here we demonstrate the chromatographic performance of the ACQUITY UPLC M-Class System with 2D Technology for the five-step fractionation of two peptide mixtures: MIX-1 MassPREP Digestion Standard and ENL Digestion Standard.
|
2D-LC system: |
ACQUITY UPLC M-Class System with 2D Technology (300-μm ID, high capacity configuration) using on-line dilution ACQUITY UPLC M-Class 300-μm scale tubing kit (p/n 205001432) |
|
First LC dimension (operating at pH 10.0) |
|
|
Column: |
XBridge Peptide BEH C18, 300Å, 5 μm, 1.0 x 50 mm (p/n 186003615) |
|
Trap column: |
ACQUITY UPLC M-Class Symmetry C18 2D HCP Trap Column, 5 μm, 300 μm x 25 mm (p/n 186007499) |
|
Flow rate: |
10 μL/min |
|
Mobile phase A: |
20 mM ammonium formate in DI water (pH=10) |
|
Mobile phase B: |
Acetonitrile (ACN) |
|
Diluting solution: |
0.1% TFA in DI water, 90 μL/min flow rate |
|
Step-elution gradient: |
A 5-step elution gradient was used for the fractionation of the peptide mixture at pH 10. The percentages of ACN in each step were: 11.4, 14.7, 17.4, 20.7, and 50.0%, respectively. |
|
Time(min) |
Flow (μL/min) |
%B |
|---|---|---|
|
0.0 |
10 |
0.0 |
|
0.5 |
10 |
0.0 |
|
1.0 |
10 |
11.4 |
|
6.0 |
10 |
11.4 |
|
7.0 |
10 |
0.0 |
|
20.0 |
10 |
0.0 |
|
Second LC dimension (operating at pH 2.5) |
||
|
Column: |
ACQUITY UPLC M-Class HSS T3 Column, 100Å, 1.8 μm, 300 μm x 150 mm (p/n 186007472) |
|
|
Column temp.: |
60 °C |
|
|
Flow rate: |
10 μL/min |
|
|
Mobile phase A: |
0.1% FA in DI water (pH=2.5) |
|
|
Mobile phase B: |
0.1% FA in ACN |
|
Time(min) |
Flow (μL/min) |
%B |
|---|---|---|
|
0 |
10 |
3 |
|
40 |
10 |
45 |
|
41 |
10 |
90 |
|
42 |
10 |
90 |
|
43 |
10 |
3 |
|
50 |
10 |
3 |
|
MS system: |
SYNAPT G2-S HDMS Mass Spectrometer equipped with a narrow bore ESI probe (p/n 186007529) |
|
|
Ionization mode: |
ESI+ |
|
|
TOF resolution: |
Resolution mode (~22,000) |
|
|
Capillary voltage: |
2.6 kV |
|
|
Cone voltage: |
40 V |
|
|
Source offset: |
60 V |
|
|
Source temp.: |
100 °C |
|
|
Desolvation temp: |
150 °C |
|
|
Desolvation gas flow: |
500 L/h |
|
|
Lockspray: |
0.2 μM GFP in 50% ACN, 0.1% FA, sampled every 4 min |
|
|
Acquisition parameters: |
m/z range: 100–1990 0.5 sec scans, 50 min runtime |
|
|
MSE settings |
Trap |
Transfer |
|
Low energy scans (precursors) |
4V |
4V |
|
High-energy fragmentation scans |
20-45V ramp |
10V |
*Observation: A Xevo G2-XS Mass Spectrometer using similar MS conditions is suited for this application as well.
MassLynx Software v4.1 (SCN 916)
A tryptic digest mixture derived from four different proteins (MassPREP Digestion Standard MIX-1, p/n 186002865) was diluted with 1 mL of 20 mM ammonium formate (pH 10) to prepare a solution containing 1 μM yeast alcohol dehydrogenase (ADH), 1 μM rabbit glycogen phosphorylase b (PHO), 0.4 μM yeast enolase 1 (ENL), and 0.6 μM bovine serum albumin (BSA). In addition, a stock solution containing 1 μM of the ENL MassPREP Digestion Standard (p/n 186002325), was prepared by adding 1 mL of 20 mM ammonium formate to the ENL glass vial (1 nmol). The digest was then diluted 10 fold to prepare a solution containing 100 nM ENL digest. The injection volume for each sample was 50 μL.
Two samples, a tryptic digest mixture derived from four different proteins (MassPREP Digestion Standard MIX-1) and an enolase digest (ENL MassPREP Digestion Standard) were studied using 2D-RP/RP chromatography with the configuration shown in Figure 1.
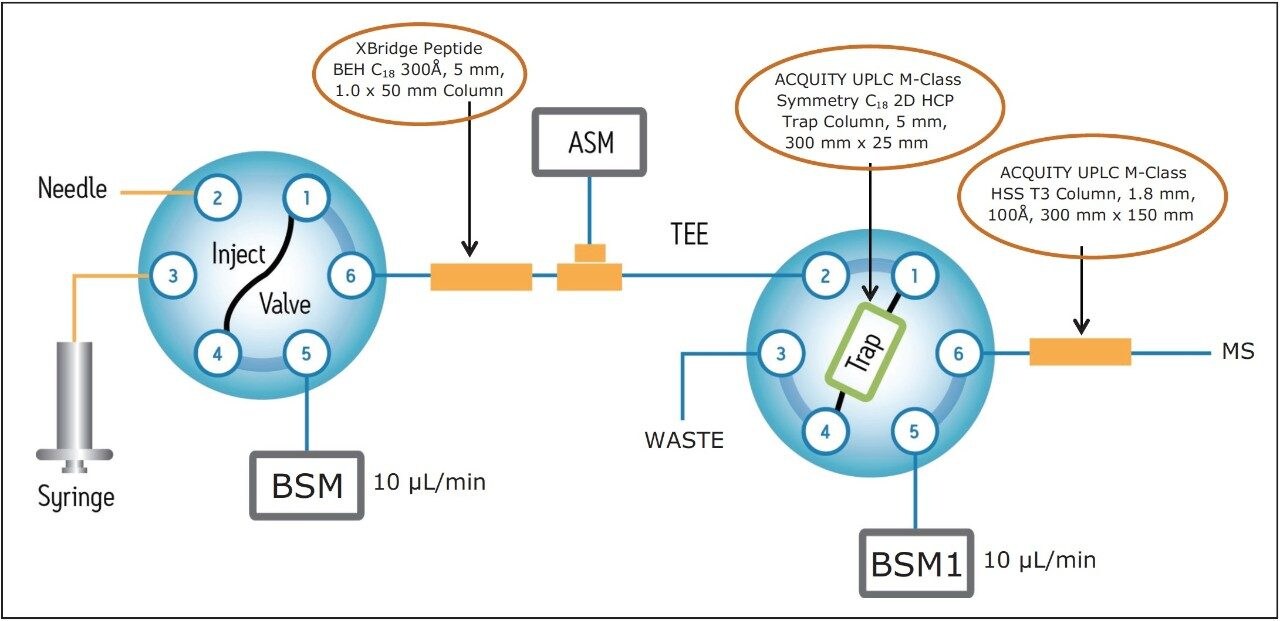
Figure 2 displays the base peak chromatograms (2A) and several extracted mass chromatograms (2B) recorded for the 2D-LC RP/RP five-step fractionation experiment performed on the MIX-1 digest using the parameters listed in the Experimental section.
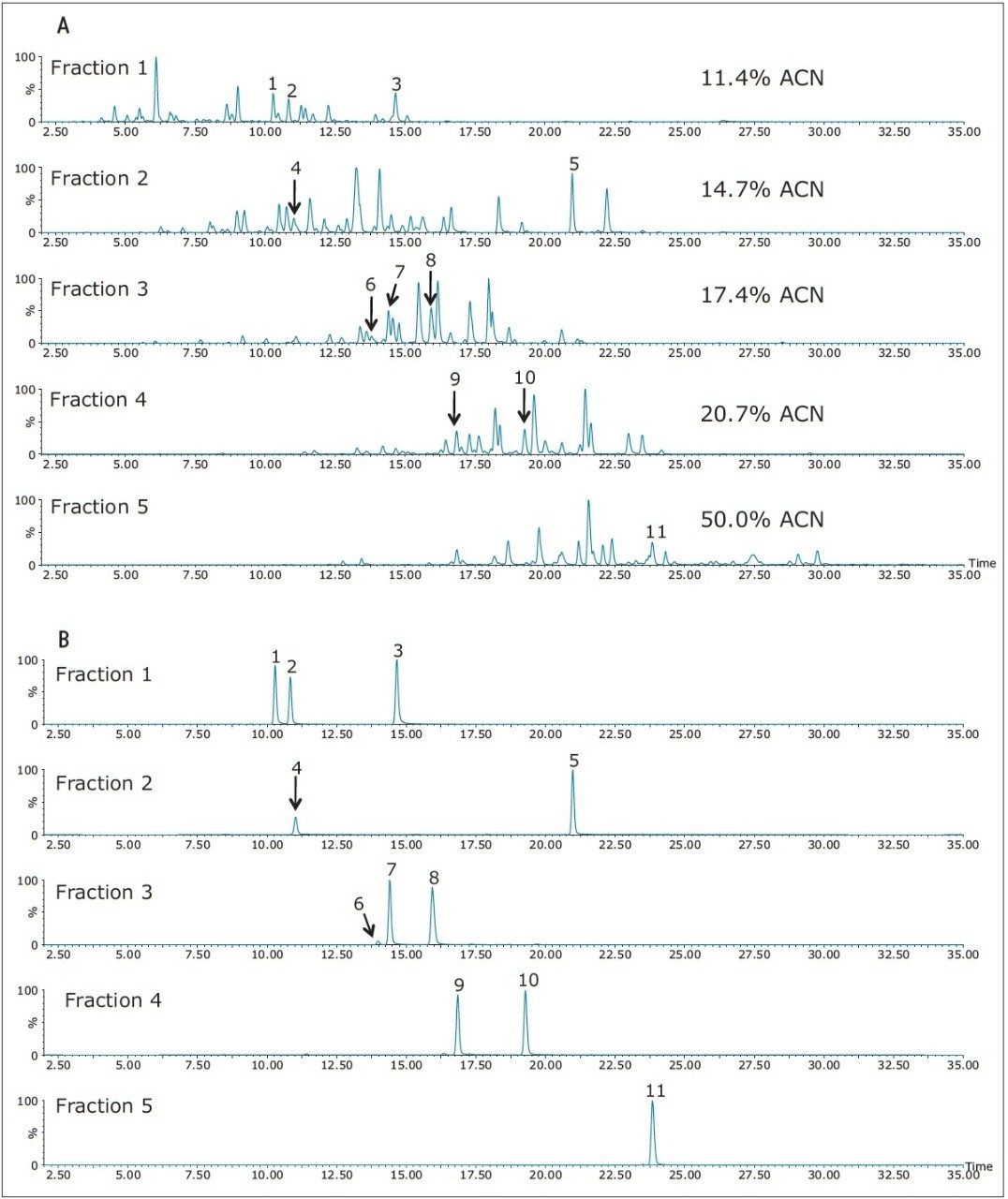
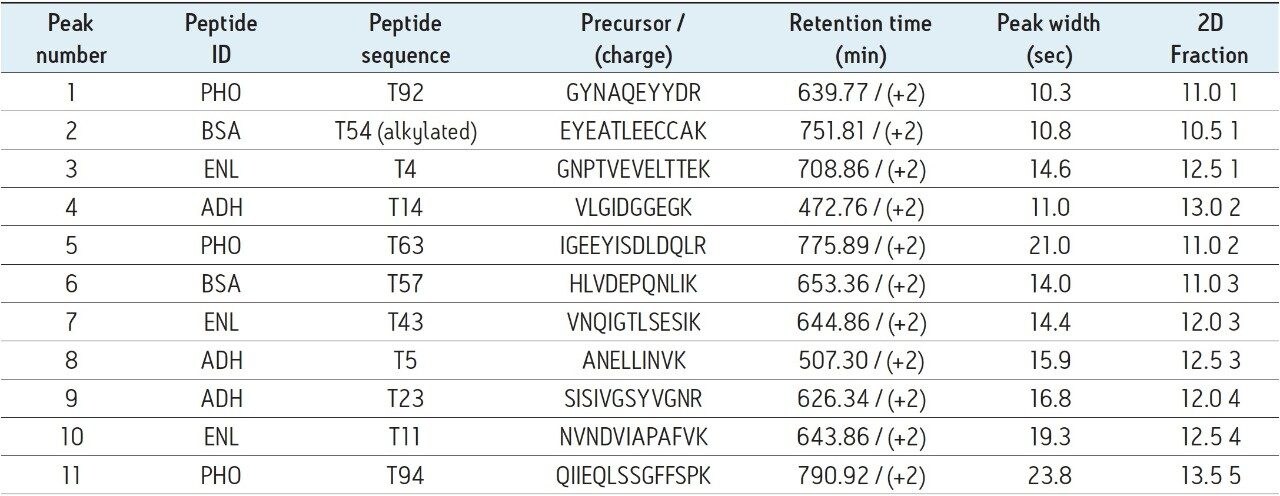
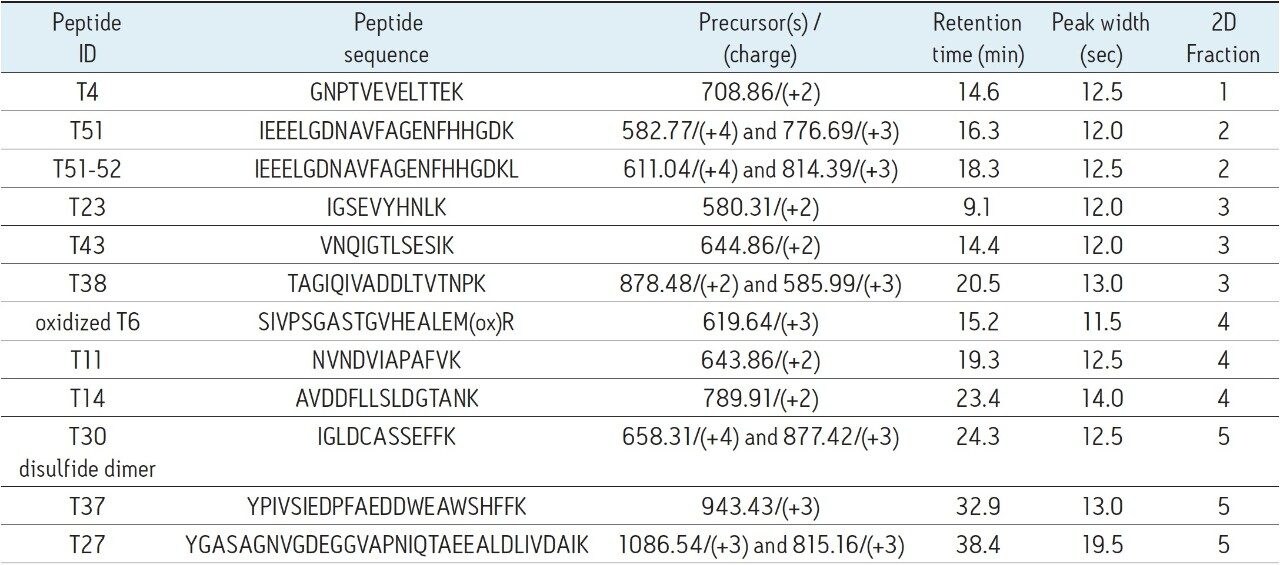
The elution times and peak widths of 11 peptides (from four different proteins) highlighted in this figure are listed in Table I along with peptide sequences and their corresponding monoisotopic precursor ions. The same 2D-LC fractionation experiment was repeated for the ENL digest and the base peak chromatograms produced for this sample are presented in Figure 3. The retention times and peak widths of 12 ENL peptides eluting in different fractions are shown in Table II.
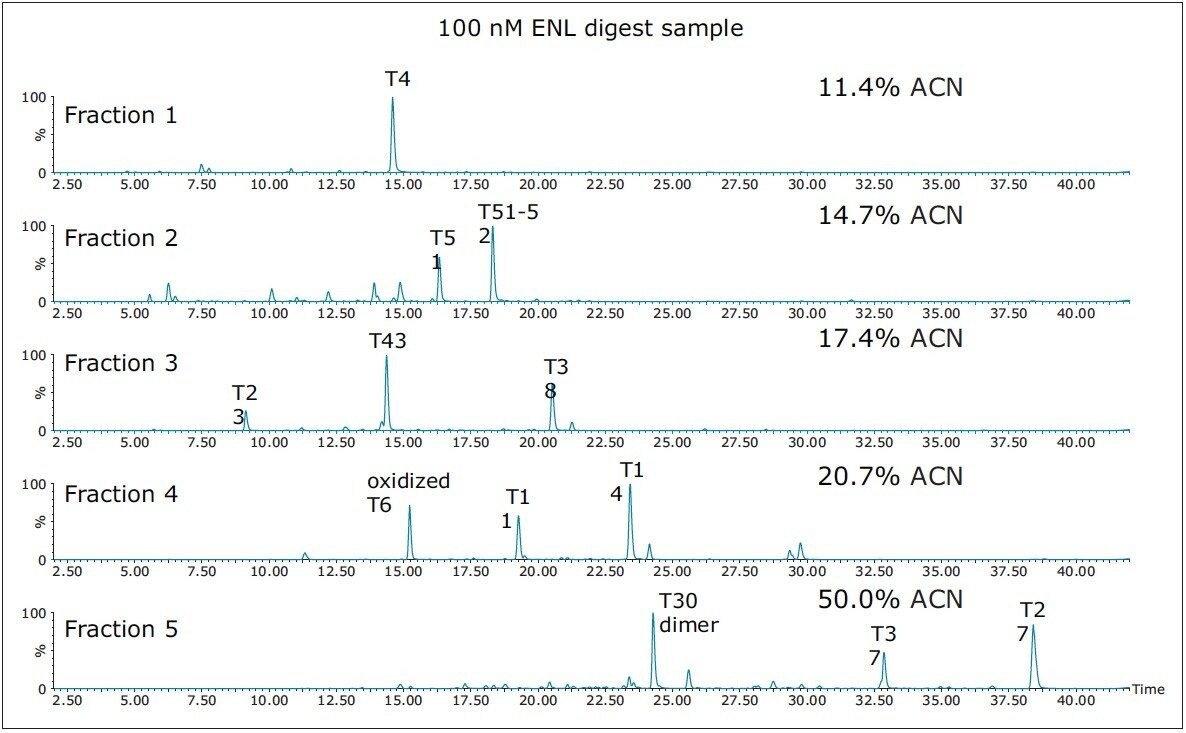
Based on the peak width values recorded at 10% of peak height for the peptides listed in Table I and II, the average peak capacity of the 2nd dimension separation is estimated to be 200. Considering that the 2D-LC experiment is comprised of a five-step fractionation process, the theoretical peak capacity of the 2D-LC HCP setup is 1,000, clearly demonstrating the capability of this 2D-LC system to generate a high resolution separation.
Here we demonstrate that the ACQUITY UPLC M-Class System with 2D Technology (300 μm I.D., high capacity configuration) is capable of providing high chromatographic resolving power required for the quantitative HCP analysis and the separation of other complex peptide samples.
720005333, March 2015