
In this application note, a simple HPLC separation of five sources of Goldenseal was performed in five minutes, and seven compounds were identified.
Natural product herbal supplements are widely used as remedies for physical ailments. Depending on sample origin and processing, product composition can range widely in the number, type, and amount of natural product compounds present. As consumers are growing in concern to the side effects of chemicals used in cosmetics and personal care products, the use of herbal plants in cosmetics,1 and personal care products,1,2 is growing in demand. Accurate sample characterization from numerous sources is useful to control supplement quality. However, the sample complexity and/or variability require highly efficient columns that do not sacrifice analysis speed. CORTECS 2.7 μm Columns contain solid-core particles which produce high peak capacity separations with reduced back pressure, making them ideal for use on traditional HPLC instruments. Using CORTECS 2.7 μm Columns, therefore, allows easier analysis of natural product mixtures.
Goldenseal is a plant native to southeastern Canada and the northeastern United States. Traditionally, it has been used to support digestion, mucous membranes, bile secretions, and many other bodily functions, as well as a topical treatment. In this application note, five different sources of Goldenseal which span four different manufacturers, two different parts of the plant, and two different formulation matrices are analyzed. This variety of sources was tested to determine the differences between each sample and if there were any compounds common to all the tested sources. Each sample was analyzed using a CORTECS C18+, 2.7 μm, 3.0 x 50 mm Column on an Alliance HPLC System with both UV and an ACQUITY QDa Detector in order to get fast and reliable mass data for peaks present in the sample, providing additional and crucial information for the characterization of the natural products.
20 mg of powdered sample was removed and placed into a 10 mL centrifuge tube. 2.5 mL of 90:10 methanol:water with 0.1% acetic acid was added. Samples were sonicated for 15 minutes, and centrifuged at 4000 rpm for 5 minutes. The supernatant was then removed and placed into a separate vial. Extraction was performed three additional times. Extracted liquid filtered through a 0.1 μm nylon filter prior to injection.
Two drops of liquid Goldenseal was added to 10 mL of 90:10 methanol:water with 0.1% acetic acid. Sample filtered through a 0.1 μm nylon filter prior to injection.
|
System: |
Alliance HPLC |
|
Column: |
CORTECS C18+, 2.7 μm, 3.0 x 50 mm (p/n 186007400) |
|
Mobile phase A: |
0.1% formic acid in water |
|
Mobile phase B: |
0.1% formic acid in acetonitrile |
|
Gradient: |
7–30% B in 5.0 minutes, return to 7% B in 0.1 minutes, hold for 1.0 minutes |
|
Flow rate: |
1.0 mL/min |
|
Column temp.: |
30 °C |
|
Detection (UV): |
300 nm |
|
ACQUITY QDa setting: |
ESI+ mode, full scan from 150–1250 amu |
|
Injection volume: |
1.0 μL |
|
Sample vials: |
LC-MS Certified Max Recovery Vials (p/n 600000670CV) |
|
Data management: |
Empower 3 CDS |
Five commercial sources of Goldenseal were acquired for characterization. Table 1 outlines the source of each sample as well as details regarding the part of the plant used and the sample format.
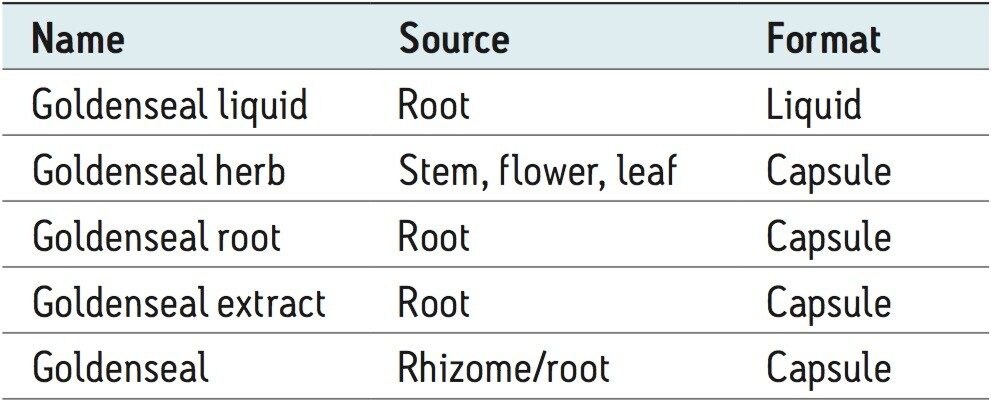
After sample preparation, the samples were injected and both UV and mass data were collected. Figure 1 shows the full scale separation of the five samples.
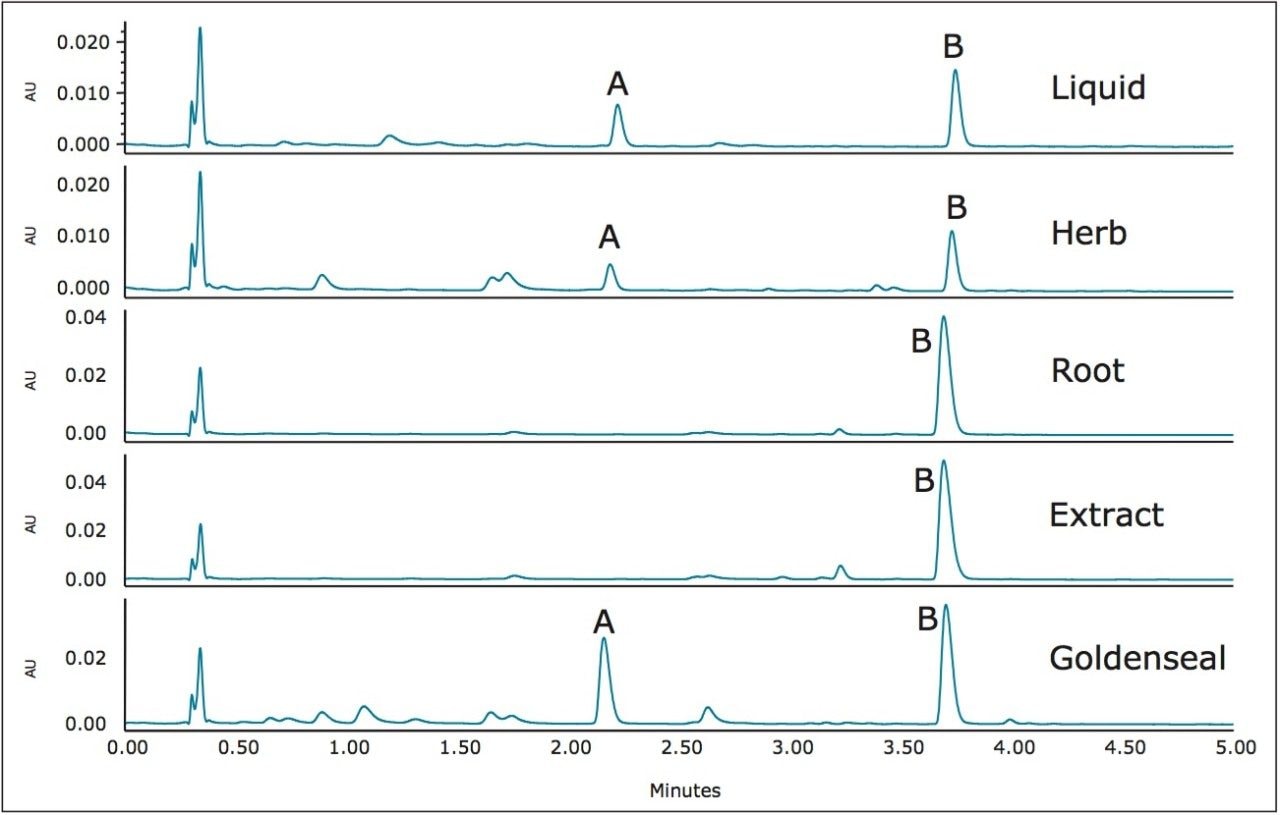
The chromatograms were then scaled to show the low level constituents that exist in the samples. Figure 2 shows the zoomed in chromatograms of the five samples.
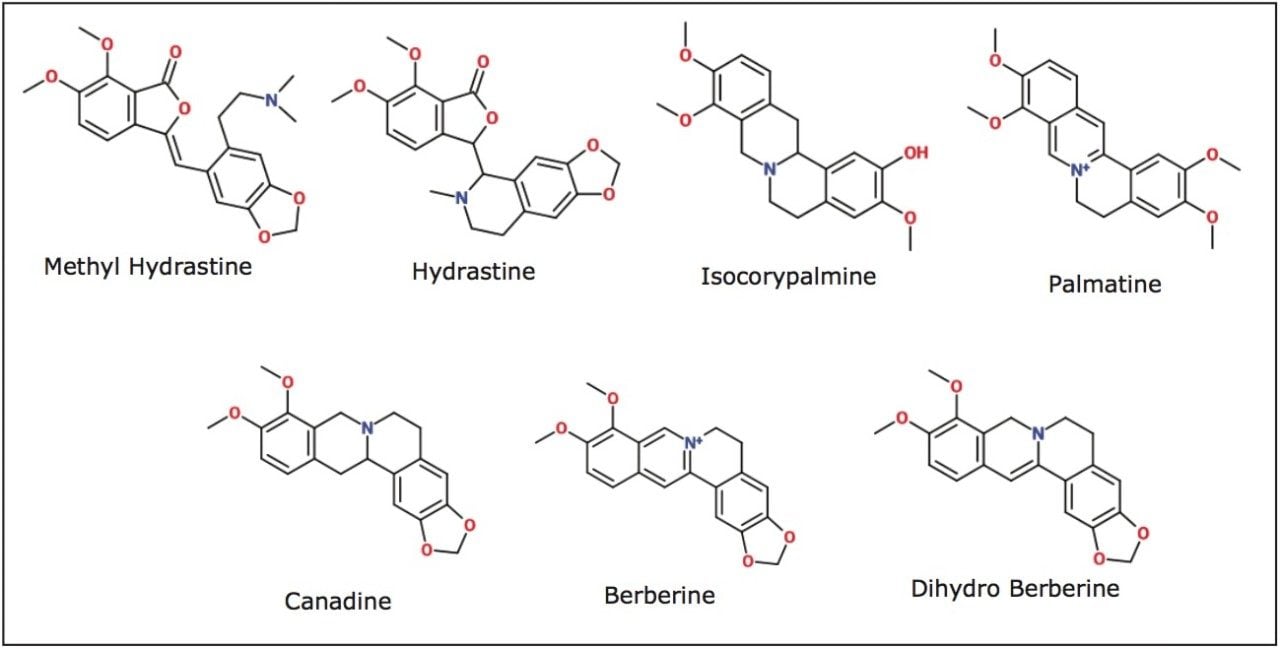
The distinct differences in the UV chromatograms of the five different samples were evident due to the high efficiency of the CORTECS Column. The intensity of peak B varied from 0.04 to 0.02 AU, indicating that different samples had different amounts of that particular compound. Further sample characterization required additional information. To obtain this, ACQUITY QDa mass spectral data was examined and compared with Goldenseal data from the literature.4 A total of seven alkaloids were identified (see Table 2 and Figure 3).
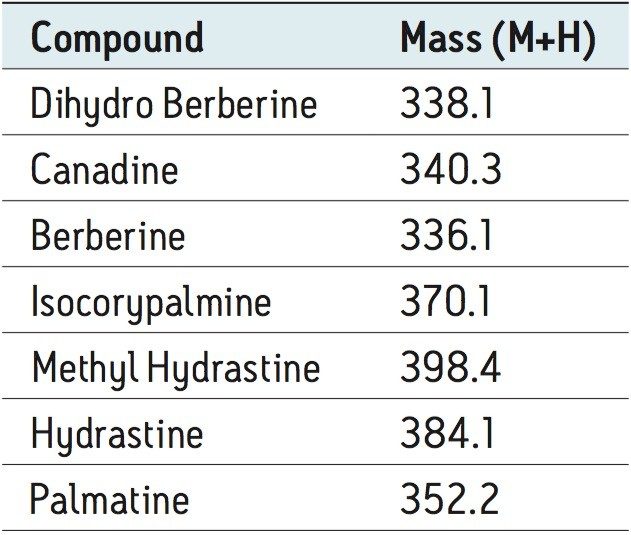
Using the ACQUITY QDa Detector, some of the peaks observed in the UV chromatograms were identified. The liquid sample had the most identifiable peaks, showing the presence of all seven compounds listed in Table 2. The remaining samples each exhibited two or more identifiable peaks. Berberine was the most abundant component and the only compound present in all samples. Figure 4 shows the identification of peaks in the liquid sample using extracted ion chromatograms (EIC).
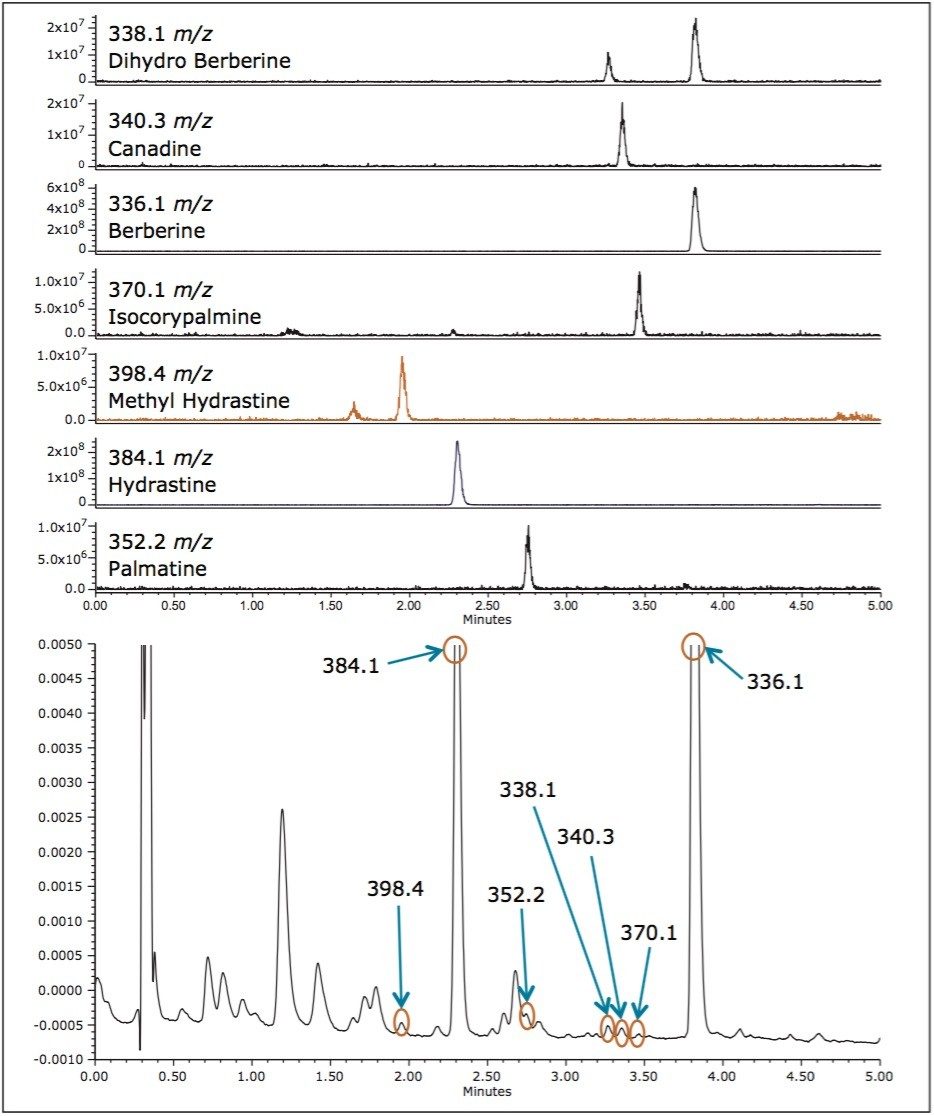
By using the ACQUITY QDa Detector, the two main component peaks (A and B in Figures 1 and 2) were identified as hydrastine and berberine, respectively. An additional five compounds were also identified and linked to peaks in the UV chromatogram. No two samples show exactly the same compounds at the same concentrations. These traces represent a type of “fingerprint” that is characteristic of each Goldenseal sample. Such fingerprints can be useful in comparing Goldenseal from different manufacturers as well as different sources of the plant. The rapid separation of a complex sample such as Goldenseal is possible due to high efficiency of CORTECS 2.7 μm Columns. By combining UV data with the mass data obtained with an ACQUITY QDa Detector, a full characterization of each sample can be made giving an analyst valuable information with minimal effort.
Natural product analysis and characterization can be a difficult process due to the complex nature of the sample. Gathering fast and reliable data is essential for characterization of complex samples such as Goldenseal in this application. Using a CORTECS C18+ 2.7 μm Column, a complex separation can be performed more easily. CORTECS 2.7 μm Columns offer high efficiency while operating within the pressure limits of an HPLC system. By combining the newest column technology with the newest technology in mass detection (ACQUITY QDa), a simple separation of five sources of Goldenseal was performed in five minutes and seven compounds were identified.
720005229, October 2015