This is an Application Brief and does not contain a detailed Experimental section.
This application brief illustrates the effectiveness of convergence chromatography for the separation of compounds with chiral centers. Adding to the complexity of the separation is the addition of three hydroxylated metabolites (structural analogs), which contain chiral centers.
UltraPerformance Convergence Chromatography (UPC2) coupled with MS detection facilitates the enantiomeric separation of parent compounds and three hydroxy metabolites.
Many drug candidates and their metabolites contain one or more chiral centers. During the drug development process, an essential step is to identify and monitor the various enantiomers that may exist. Supercritical fluid chromatography (SFC) is known to be a highly effective technique for separating structurally similar compounds, including structural analogs and enantiomers. Additionally, using SFC provides many benefits such as high efficiency, fast separations, and use of MS-compatible solvents.
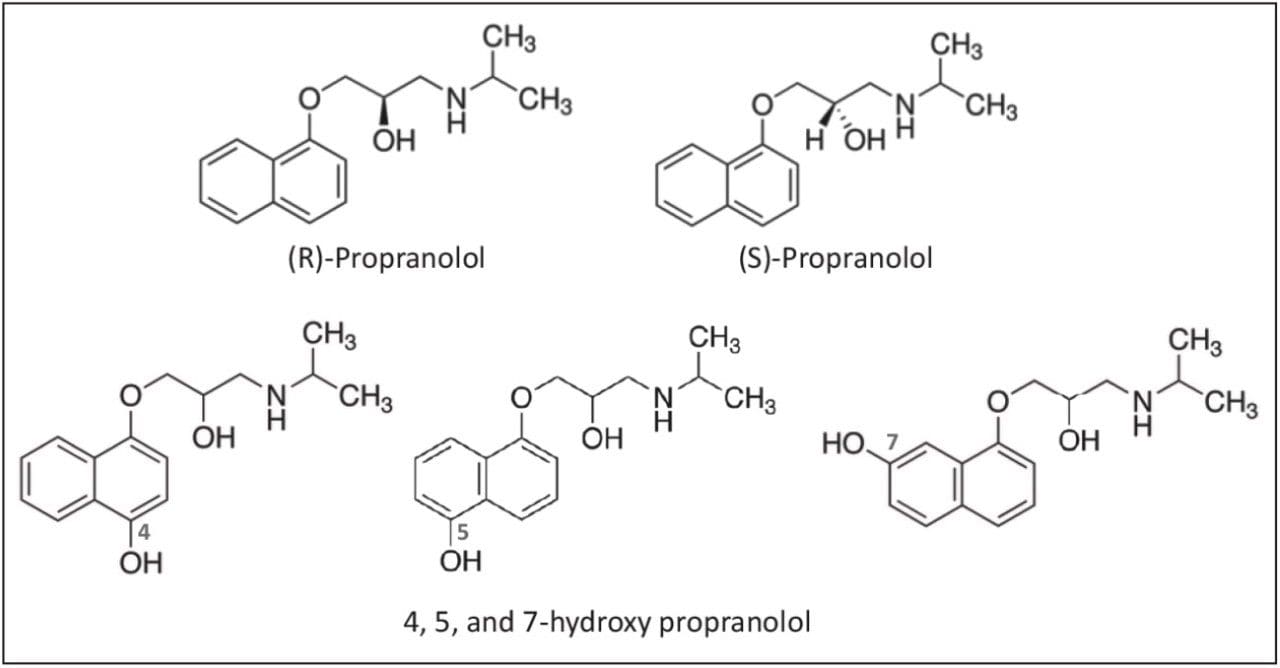
In this technology brief, we demonstrate the ease of enantiomeric separation and detection of warfarin, and three hydroxylated metabolites (Figure 1), as well as propranolol and three hydroxylated metabolites (Figure 2) utilizing UPC2-MS/MS.
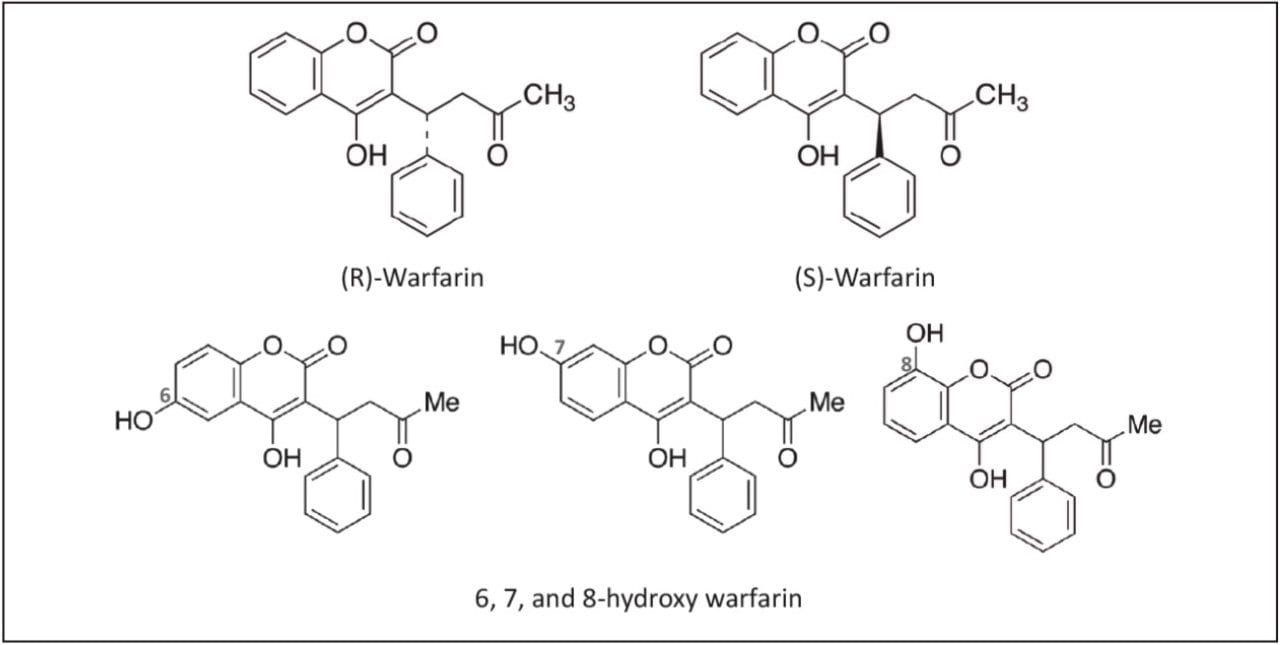
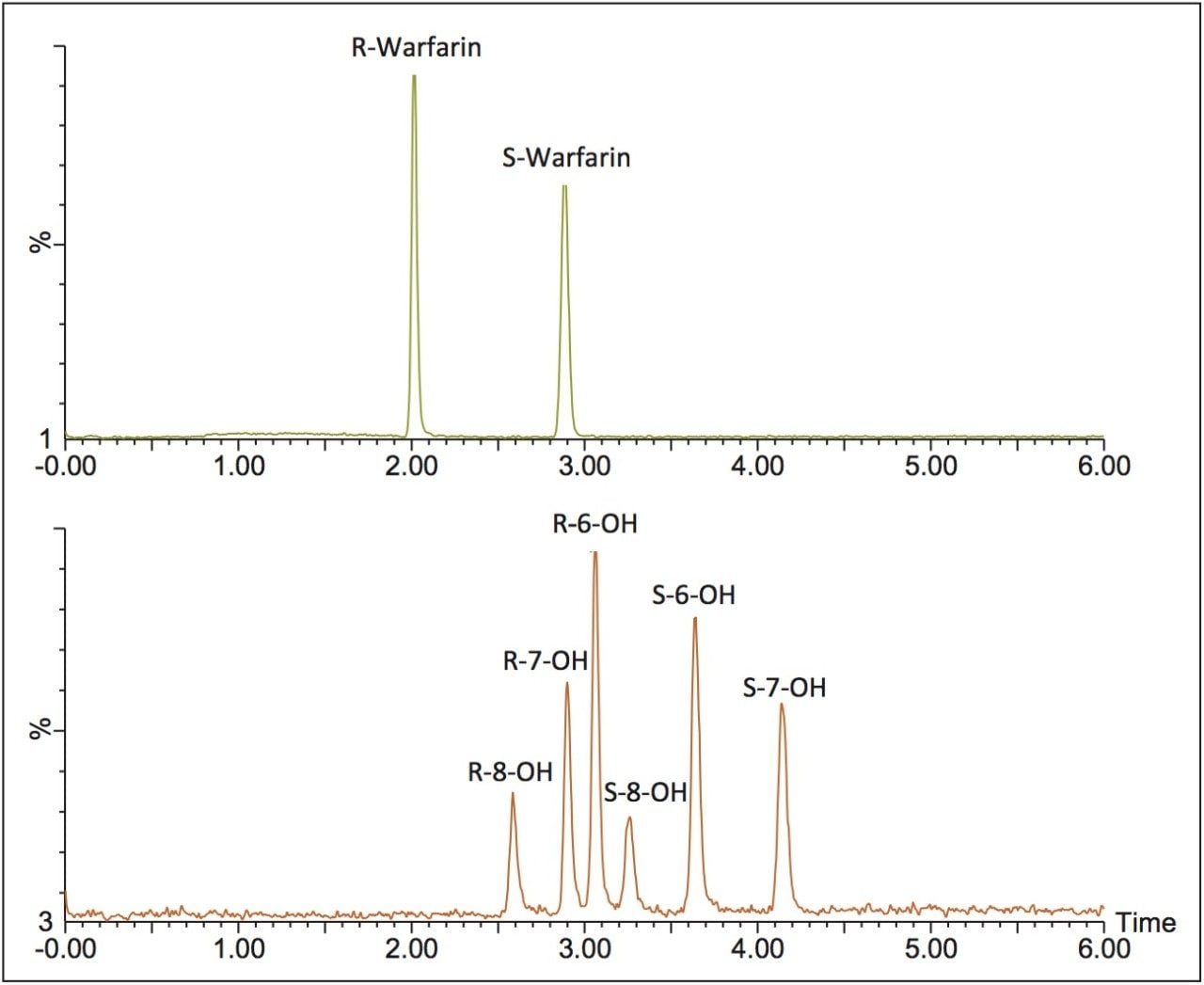
For warfarin, method optimization was achieved using an ACQUITY UPC2 System with an ACQUITY UPC2 Trefoil CEL1, 2.5 μm, 3.0 x 100 mm Column using ammonium formate modified methanol as the co-solvent. Resolution for the six forms of hydroxy warfarin was achieved in under 4.5 minutes using a linear gradient and maintaining the column temperature at 10° C (Figure 3).
For propranolol, the separation was performed on an ACQUITY UPC2 Trefoil CEL1, 2.5 μm, 3.0 x 150 mm Column using ammonium formate modified methanol as the co-solvent. In this example, resolution of the six forms of hydroxy propranolol was achieved in 14 minutes using a linear gradient and maintaining the column temperature at 30° C.
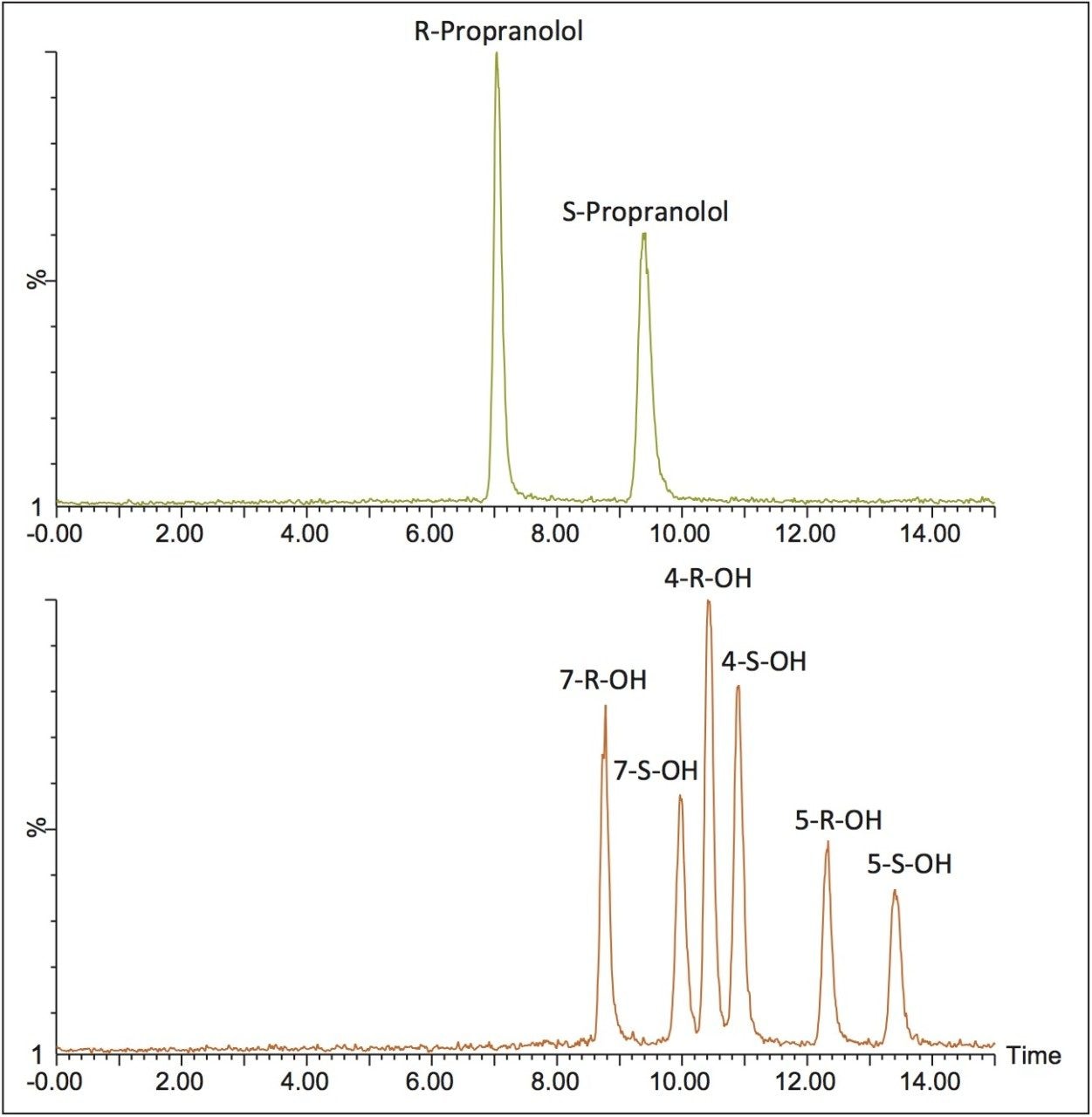
The combination of UPC2 with MS detection facilitated the enantiomeric separation of parent compounds and three hydroxy metabolites. Warfarin, 6-hydroxywarfarin, 7-hydroxywarfarin, and 8-hydroxywarfarin enantiomers were resolved in less than 5 minutes and propranolol, 4-hydroxypropranolol, 5-hydroxypropranolol, and 7-hydroxypropranolol were resolved in under 15 minutes. The ability to identify the various chiral forms of a compound and metabolites is an essential step during the drug development process, and is easily achieved using UltraPerformance Convergence Chromatography.
720005187, October 2014