
In this application note, we describe the analytical profiling of a triazole containing pesticide formulation using UPC2 and a combination of UV and mass detection.
Research associated with the development of new agricultural pesticide formulations centers around the design of products that provide highly effective and specific action towards the target organism with reduced application rates.1 It is estimated that 30% of the pesticides on the market today have optical isomers.2,3 However, the desired activities often result from one single enantiomer in the optical isomer mixtures.4 It is therefore important to assess the enantiomeric purity of the chiral active ingredients in the formulation.1,5 In addition, the detection, characterization, and quantitation of the other components in the formulation are necessary to support product registration.
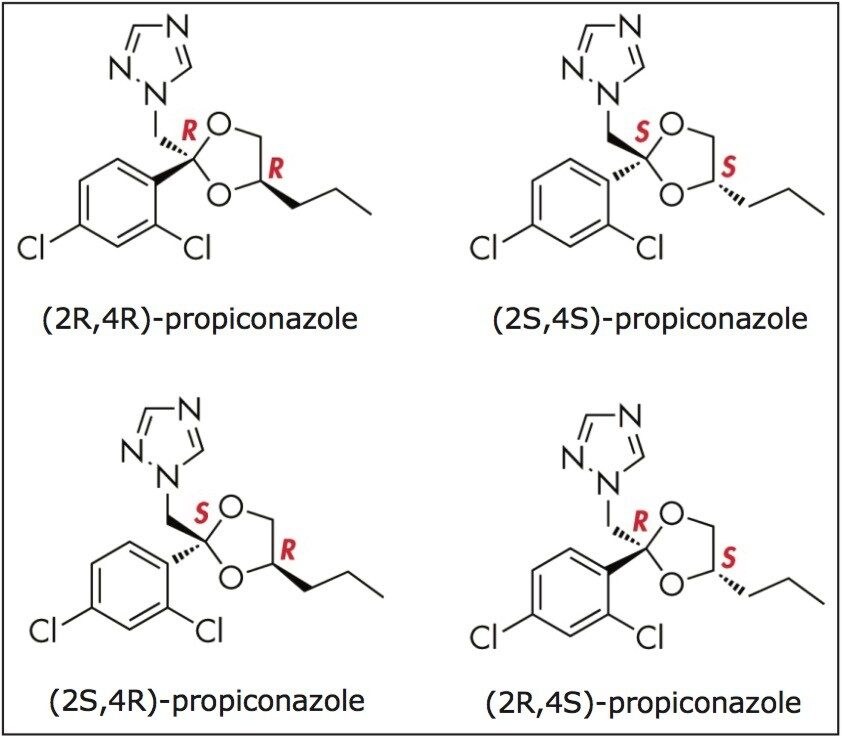
Liquid chromatography (LC) on chiral stationary phases (CSPs), such as polysaccharide stationary phases including amylose and cellulose, has been the most commonly used chiral separation technique.6-9 More recently, there has been an increasing adoption of supercritical fluid chromatography (SFC) on CSPs for chiral separation.10,11 The properties of a supercritical fluid, its high diffusivity and low viscosity in particular, enable high efficiency chiral separations with shorter run times. For example, triazole fungicides, such as propiconazole (structure shown in Figure 1), are a commonly used group of pesticides because of their potent activity against a broad spectrum of crop diseases. Using HPLC, the analysis times for the diastereomeric resolution of propiconazole range from 34 to 50 min.1,6-8 Similar resolutions were achieved for propiconazole using SFC, but the analysis times were reduced to 10 min.11
UltraPerformance Convergence Chromatography (UPC2) applies the performance advantages of UPLC to SFC, combining the use of supercritical CO2 with sub-2-μm particle columns.12,13 UPC2 is an orthogonal analytical technique to reversed-phase LC and can be used to solve complex separations challenges.
In this application note, we describe the analytical profiling of a triazole containing pesticide formulation using UPC2 and a combination of UV and mass detection. Waters ACQUITY QDa Detector is a novel mass detector that can be integrated into existing liquid chromatography configurations in order to increase sensitivity and complement the results obtained when using only UV detectors.
All separations were performed on a Waters ACQUITY UPC2 System equipped an ACQUITY UPC2 Photodiode Array (PDA) and positive ion electrospray mass spectrometry (MS) using an ACQUITY QDa Detector. Empower 3 FR2 Software was used for data acquisition and processing.
The authentic pesticide standards were made up in 50:50 acetonitrile/water. 2 grams (g) of the commercially available pesticide formulation was weighed, and 8 mL of 50:50 (v/v) acetonitrile/water was added. The resulting mixtures were sonicated for 10 minutes and the samples were syringe filtered into an autosampler vial using a 0.2-μm PVDF filter in preparation for sample analysis.
|
Achiral separation |
|
|---|---|
|
Separation mode: |
Gradient |
|
Column: |
ACQUITY UPC2 BEH, 3.0 x 100 mm, 1.7 μm |
|
Co-solvent (B): |
Methanol |
|
ABPR: |
1990 psi/137 bar |
|
Flow rate: |
1.5 mL/min |
|
UV detection: |
220 nm |
|
Column temp.: |
35 °C |
|
Injection volume: |
0.5 μL |
|
Formulation A achiral gradient conditions: |
0 min 3% B, 4 min 30% B, 6 min 30% B, return to initial conditions. |
|
Separation mode: |
Gradient |
|
Column: |
Amylose Chiral, 3.0 x 150 mm, 2.5 μm |
|
Co-solvent (B): |
50:50 2-propanol/ethanol |
|
ABPR: |
1990 psi/137 bar |
|
Flow rate: |
2.0 mL/min |
|
UV detection: |
220 nm |
|
Injection volume: |
1 μL |
|
MS system: |
ACQUITY QDa Detector |
|
Ionization mode: |
ESI + |
|
Capillary voltage: |
0.8 kV |
|
Cone voltage: |
10 V |
|
Desolvation temp.: |
600 °C |
|
Source temp.: |
150 °C |
|
MS scan range: |
100 to 600 m/z |
|
Sampling rate: |
5 Hz |
|
Make up solvent: |
98:2 MeOH/water with 0.1% ammonium hydroxide at 0.3 mL/min |
|
Detector: |
ACQUITY UPC2 PDA |
|
Wavelength range: |
210 to 400 nm |
|
Sampling rate: |
20 Hz |
When MS data is combined with the UV response, it allows the analyst to determine a wider range of analytes in one analytical run with an increased level of confidence.
The formulation sample first underwent achiral separations on a sub-2-μm stationary phase, followed by chiral separations on a 2.5-μm chiral CSP. A minor isomer of the active ingredient (AI) was identified. Further chiral analyses revealed similar chirality between the minor isomer and the AI.
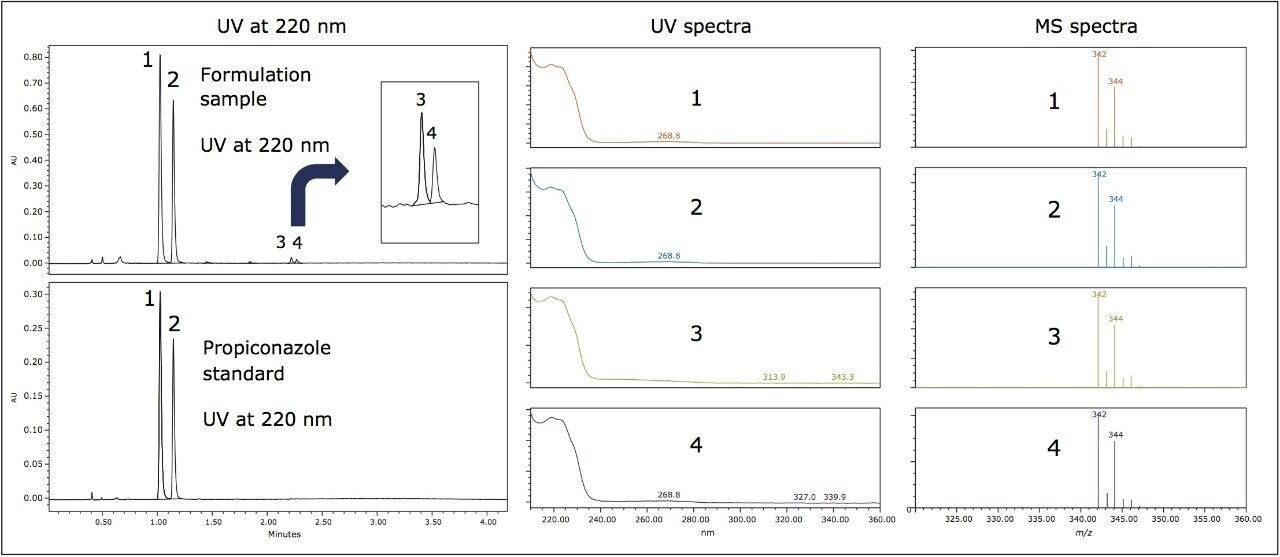
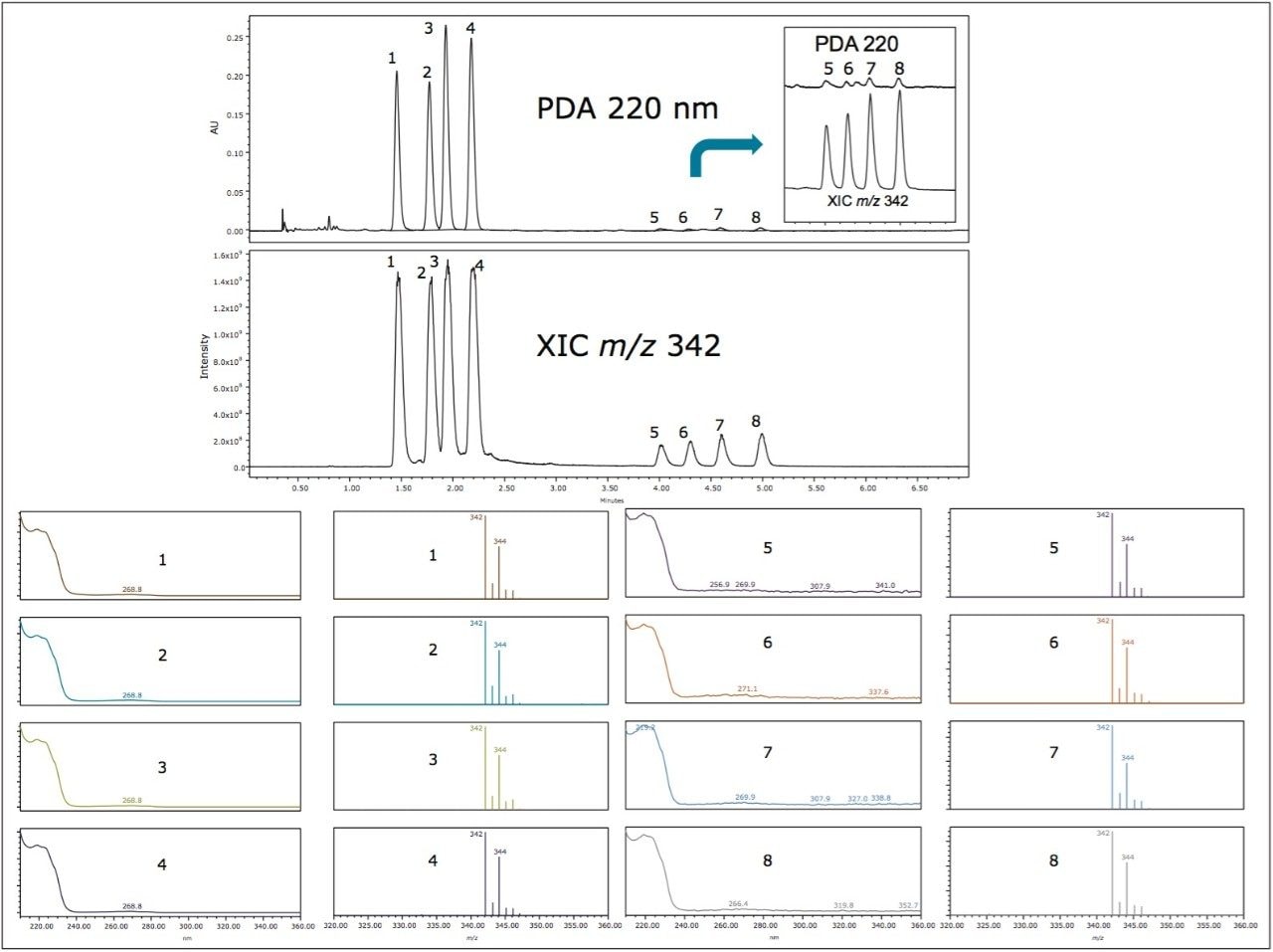
Figure 2 shows the ACQUITY UPC2 chromatograms of the propiconazole standard (lower trace) and the formulation sample (top trace) obtained using an ACQUITY UPC2 BEH Column. The retention times of peaks 1 and 2 in the formulation sample matched those of the propiconazole standard. These two peaks correspond to the propiconazole diastereomers. It is noted, however, that there were two minor peaks (peaks 3 and 4) observed in the formulation with retention times (tR) of 2.22 min and tR 2.26 min, respectively.
The UV spectra of peaks 3 and 4 resemble those for peaks 1 and 2 (Figure 2), indicating their structural similarity. In addition, the four peaks resulted in identical mass spectra with base peaks at m/z 342 and an isotopic pattern characteristic of dichlorinated compounds. The m/z matched the protonated propiconazole.
The ACQUITY UPC2 System has multi-column switching capabilities and a choice of up to four co-solvents which conveniently allows both achiral and chiral method development and sample analysis to be performed on the same system. The method development process can be completed rapidly due to the shorter analysis times that are possible using this technique.
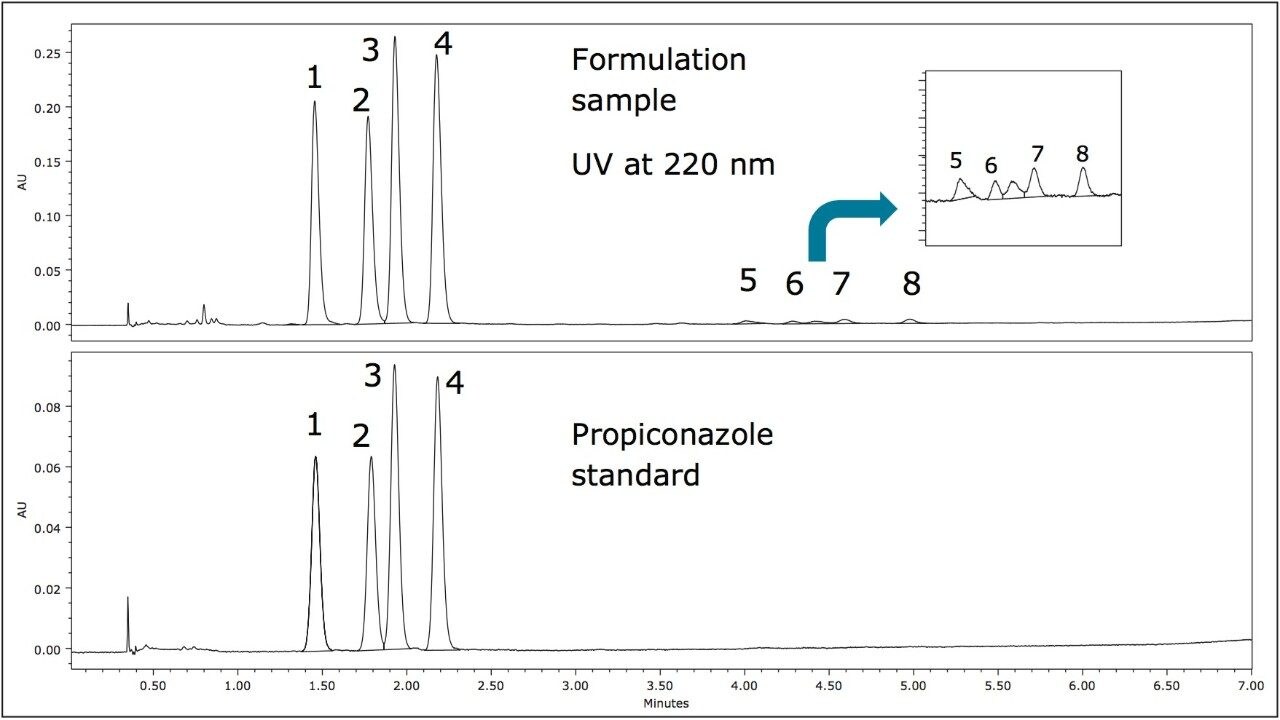
Figure 3 shows the ACQUITY UPC2 chromatograms of propiconazole standard and the formulation sample using gradient separation on an Amylose Chiral Column. The two diastereomer peaks of propiconazole observed in Figure 2 were resolved into four individual peaks (1-4). Interestingly, the two minor isomer peaks in Figure 2 were also resolved into four peaks (5-8) in a comparable manner, indicating a similar chirality for propiconazole and the minor components.
Interrogation of the formulation sample data in Empower Software’s mass analysis window showed similar UV and identical MS spectra for all eight peaks (Figures 4 and 5). Empower 3 Software’s Mass Analysis window provides a single location to associate chromatographic peaks from all detectors used in the analysis with their corresponding spectra. The spectra from the detected peaks are time-aligned and displayed in a window above the chromatograms.
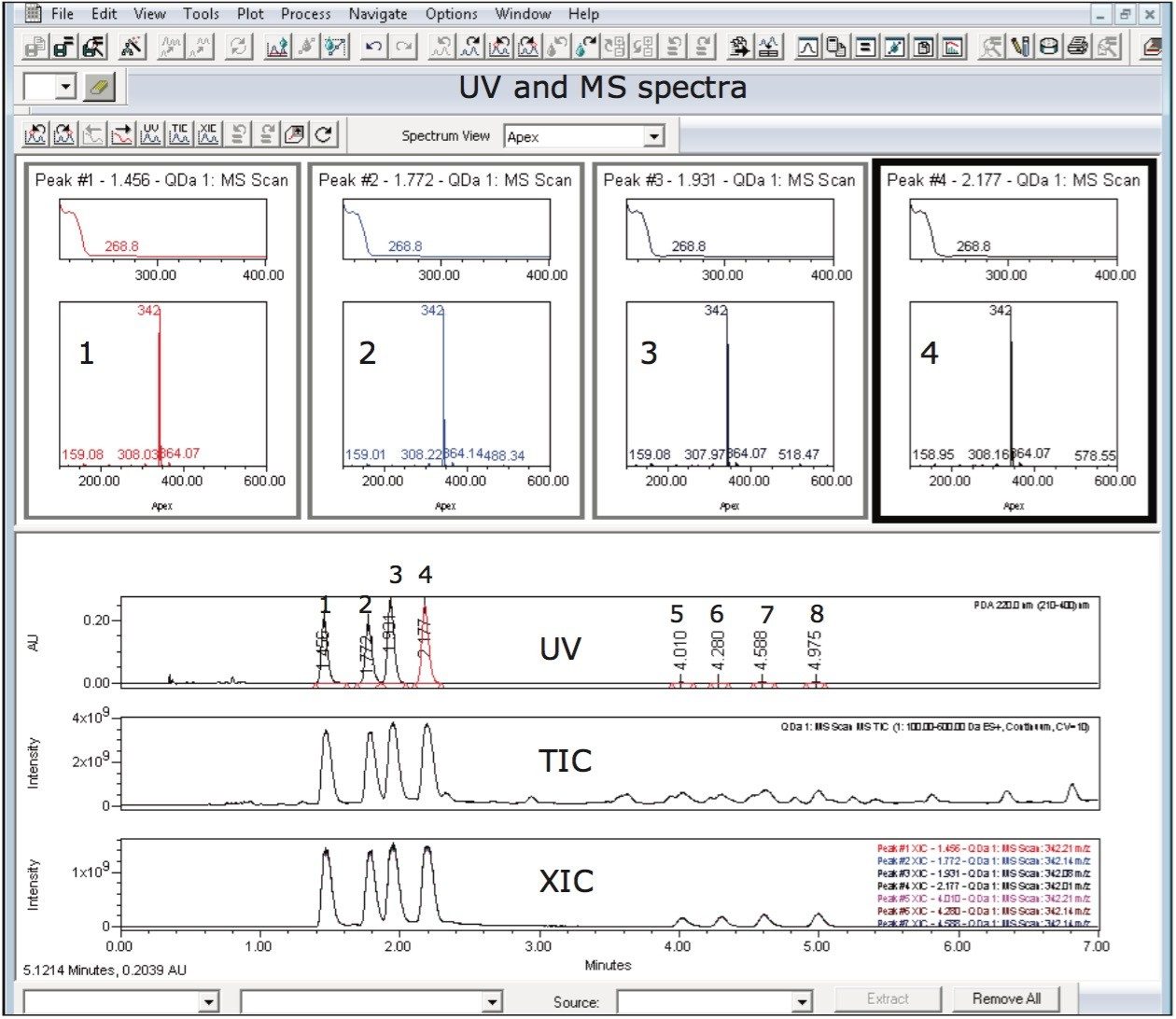
Matrix components visible in the UV chromatogram and the MS total ion chromatogram (TIC) of the formulation sample are clearly differentiated from the isomeric peaks of interest using an extracted ion chromatogram (Figure 5). The detection sensitivity and selectivity of the method are improved when using mass detection in combination with UV detection which is used in order to measure the enantiomeric purity of the chiral pesticide.
Based on the observations, it is postulated that the minor component is a regioisomer of propiconazole. A regioisomer of propiconazole originating from one of the nitrogens on the triazole ring was characterized by Glaser et al.14 Further experiments to isolate this compound for positive identification are currently underway.
In this study the achiral screen of the formulated pesticide products showed that the minor components detected using UV and mass detection had similar structural characteristics to the AI, propiconazole. The minor components had the same m/z and shared the same isotopic pattern as the triazole fungicide, AI. Subsequent chiral resolution of the propiconazole in the formulation in combination with simultaneous mass and UV detection provided valuable spectral information which allowed the minor components to be characterized as probable stereoisomers.
The addition of mass detection as a complementary analytical detection technique enhances confidence in compound detection and identification. The ACQUITY QDa Detector provides a cost-effective means to make mass detection part of the routine analysis in laboratories that have previously relied on less selective detectors.
The ACQUITY UPC2 System has column switching capabilities so that both chiral and achiral columns can be used with a choice of four co-solvents that are compatible with MS analysis. The chiral and achiral method development and analysis can be performed on the same system. These methods use supercritical CO2 as the primary mobile phase. The need to use large volumes of potentially hazardous solvents is reduced compared to normal phase separations. Consequently the cost associated with solvent waste disposal can also be reduced.
The ACQUITY UPC2 System allows high efficiency separations that can increase sample throughput compared to traditional normal-phase separations.1,6-8 The diasteromeric resolution of propiconazole using UPC2 took place in less than 3 minutes, which is at least 10 times faster than normal phase methods reviewed in the literature.
The study of enantioselective properties has previously been a challenge due to the difficulty in resolving chiral compounds. The benefit of having faster analytical methods to resolve chiral compounds is that critical information pertaining to their stereoselective behavior can be obtained more rapidly leading to increased laboratory productivity.
720004977, March 2014