
This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates the performance of the new ACQUITY UPC2 Torus 2-PIC Column for analyzing low level pharmaceutical degradation products.
Analyzing low level pharmaceutical degradation products using the new ACQUITY UPC2 Torus 2-PIC Column.
During the manufacture of pharmaceutical compounds, it is important to fully understand any possible degradation products. This is especially true in the case of pharmaceutical compounds administered orally. These analyses are challenging due to the nature of the sample. Degradation samples typically have numerous peaks making it difficult to get separation between the active pharmaceutical ingredient (API) and the degradation products. Additionally, degradation of an API can generate unexpected products depending on the parameters. Traditionally, liquid chromatography would be used to perform these separations. However, an additional technique, UltraPerformance Convergence Chromatography (UPC2), exists to help an analyst perform these difficult separations.
An ACQUITY UPC2 System harnesses the power of liquid CO2 as the mobile phase for the separation of complex samples such as pharmaceutical degradation samples. UPC2 Technology offers orthogonal separation compared to reversed phase liquid chromatography. Using the newest column technology available on the UPC2 platform, ACQUITY UPC2 Torus Columns, an analyst can rapidly perform the analysis of these types of samples. The degradation of Omeprazole tablets, used in the treatment of acid reflux and heartburn, was analyzed on an ACQUTIY UPC2 System using the new ACQUTIY UPC2 Torus 2-PIC Column. This column, created using a two-stage bonding technique, has a 2-Picolylamine ligand (2-PIC). This stationary phase provides retention stability and a unique selectivity compared to the other Torus Columns. The ACQUITY UPC2 Torus 2-PIC Column is a good starting point when developing methods for supercritical fluid chromatography (SFC).
As Figure 1 shows, the separation of Omeprazole from the degradation products is achieved. By using the ACQUITY UPC2 Torus 2-PIC Column numerous degradation products including Omeprazole Sulfide can be identified by using standard injections or mass spectrometry. The new ACQUITY QDa Detector was used to identify the Omeprazole and Omeprazole Sulfide peaks in this analysis. By using the UPC2 along with the new Torus column chemistries, an analyst can perform separations of complicated matrixes while using minimal amounts of organic solvent per analysis, reducing analysis cost.
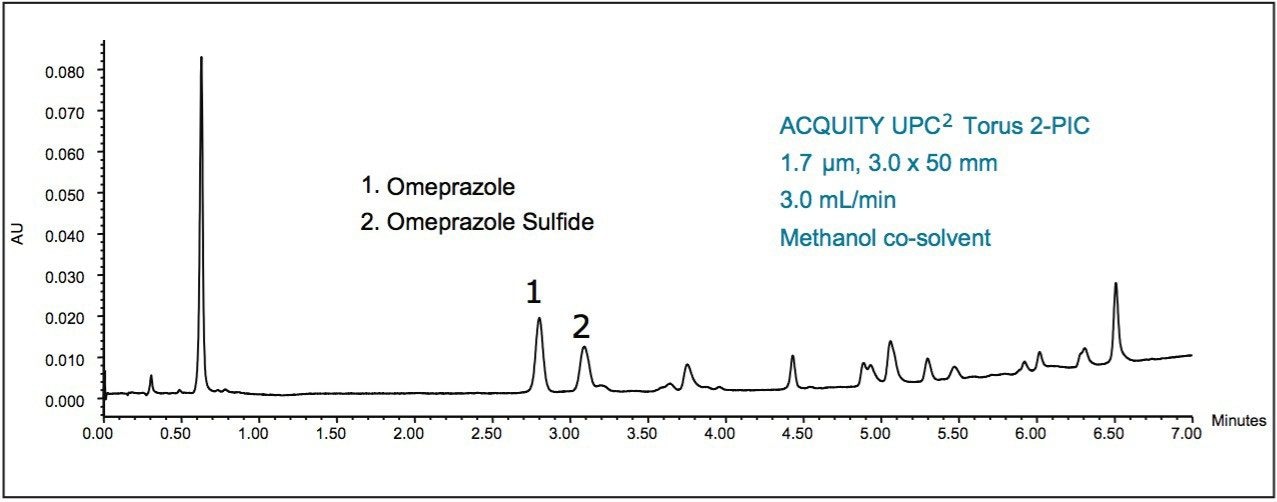
The new ACQUITY UPC2 Torus 2-PIC Column was used to analyze the acid degradation products of formulated Omeprazole. The active pharmaceutical ingredient was successfully separated from all of the degradation products. By using the ACQUITY UPC2 Torus 2-PIC Column, the degradation peaks are well separated and can be identified by either standard injections or by using a mass spectrometer. The two stage bonding procedure of the ACQUITY UPC2 Torus Columns ensures retention stability and a wide range of selectivity, making this column family a useful tool in sample analysis.
720005196, October 2014