
In this application note, a workflow to systematically develop isocratic prep LC methods for target compound isolation from complex natural product mixtures is demonstrated. The workflow starts with a generic LC analysis from which the elution %B of the target compound is calculated. The target compound is then isolated from the sample using an isocratic preparative LC method run at the calculated %B, leading to the collection of target compound with purities ranging from 73% to 100%. The isocratic prep LC methods were also proven rugged and reproducible. Compared to the commonly used gradient methods, the isocratic prep chromatography dramatically reduced the total run time and solvent usage, resulting in improved overall productivity and cost-effectiveness of the purification process. The proposed workflow is ideally suited as a quick, first step in a multi-step process for target compound isolation from natural products.
Preparative chromatography is the technique of choice for isolation and purification of different types of compounds, including pharmaceutical drug candidates, target compounds from synthetic mixtures, and bioactive compounds from natural products. As a result, there has been continuous research interest in improving the fundamental knowledge and instrumentation of preparative chromatography, with an ultimate goal of obtaining pure target compounds efficiently and cost-effectively.1-6
Most purification strategies start with chromatographic profiling of the crude mixture at the analytical scale using either gradient HPLC or UPLC, followed by a scale-up to the semi-prep or preparative level. The most straightforward method transfer from analytical to prep chromatography is a direct geometric scale-up of a linear gradient. Alternatively, a focused gradient approach, where a shallow gradient encompasses the elution %B of the target compound, is used for target compound isolation. While generally applicable, these two approaches often lead to long cycle time and increased solvent usage. Furthermore, depending on the sample complexity and desired purity, an iterative process might still be required to attain the target compounds. This is particularly true for natural product isolation, where a multi-step purification procedure is generally required due to the complexity of the sample.
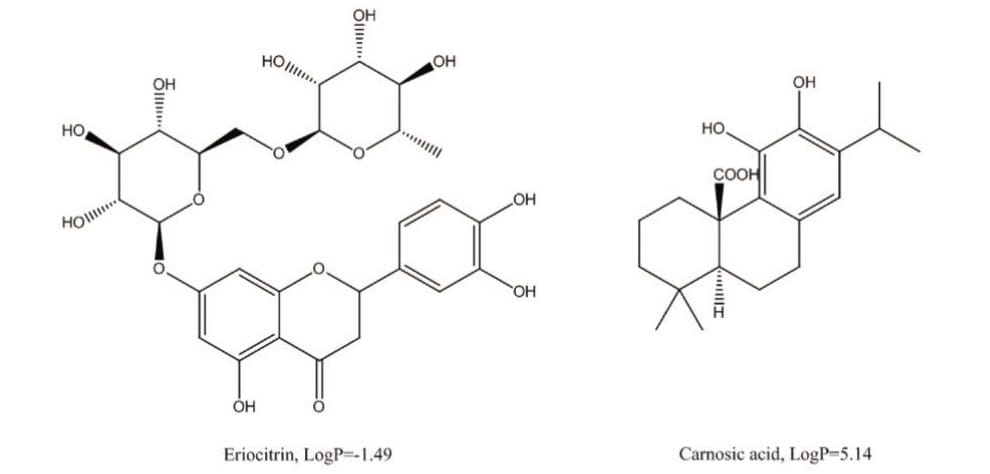
This application note describes the use of isocratic chromatography as a first purification step to quickly and efficiently simplify the mixture and enrich the target compound prior to more stringent purification steps. Eriocitrin,7 a polar compound found in peppermint extract (Figure 1), was used as a model to demonstrate the isolation and purification workflow. The purity and solvent usage for each of the different purification approaches were compared. To ensure the general applicability of the proposed workflow, the methodology was further validated by isolating carnosic acid8 (Figure 1), a relatively non-polar compound from rosemary extract.
|
Eriocitrin: |
A total of 4.9 g dried peppermint was extracted with 40 mL of 80:20 methanol/water solution for 72 hours at room temperature, with stirring. The supernatant was filtered with an Acrodisc Syringe Filter with GHP Membrane, 25 mm, 0.45 μm. The final extract volume was 29 mL. |
|
Columns: |
XSelect CSH C18 5 μm, 4.6 x 100 mm XSelect C18, OBD Prep 5 μm, 19 x 100 mm |
|
Mobile phase A: |
0.1% trifluoroacetic acid in water |
|
Mobile phase B: |
0.1% trifluoroacetic acid in acetonitrile |
|
Detection: |
220 nm |
|
Flow rate: |
Analytical, 1.46 mL/min; Prep, 25 mL/min |
|
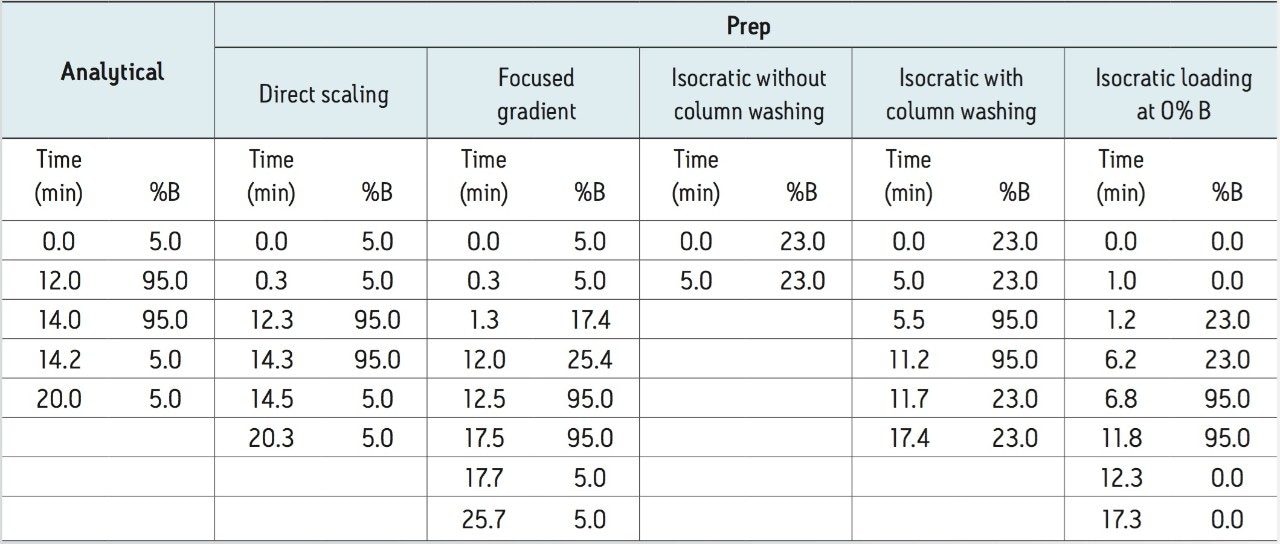
Gradient: |
See Table 1 |
|
Injection volume: |
See figures |
|
Carnosic acid: |
The extraction of carnosic acid was performed using supercritical fluid extraction (SFE) on a Waters MV-10 ASFE System controlled by ChromScope Software. A total of 1.9 g of ground rosemary was loaded into a 5-mL extraction vessel. The extraction was performed for 90 minutes with 6 mL/min CO2 and 0.2 mL/min methanol at 80 °C and 300 bar extraction pressure. The effluent was collected into a 20-mL scintillation vial with a makeup flow of 0.25 mL/min of isopropanol. The 5.5 mL of cloudy yellow extract was filtered through an Acrodisc Syringe Filter with GHP Membrane, 13 mm, 0.45 μm. The final volume of the filtered extract was brought to 6.5 mL by adding 1 mL methanol. |
|
Columns: |
XBridge C18 5 μm, 4.6 x 150 mm XBridge C18 OBD Prep 5 μm, 19 x 150 mm |
|
Mobile phase A: |
0.1% formic acid in water |
|
Mobile phase B: |
0.1% formic acid in acetonitrile |
|
Detection: |
220 nm |
|
Flow rate: |
Analytical, 1.46 mL/min; Prep, 25 mL/min |
|
Gradient: |
See Table 2 |
|
Injection volume: |
See figures |
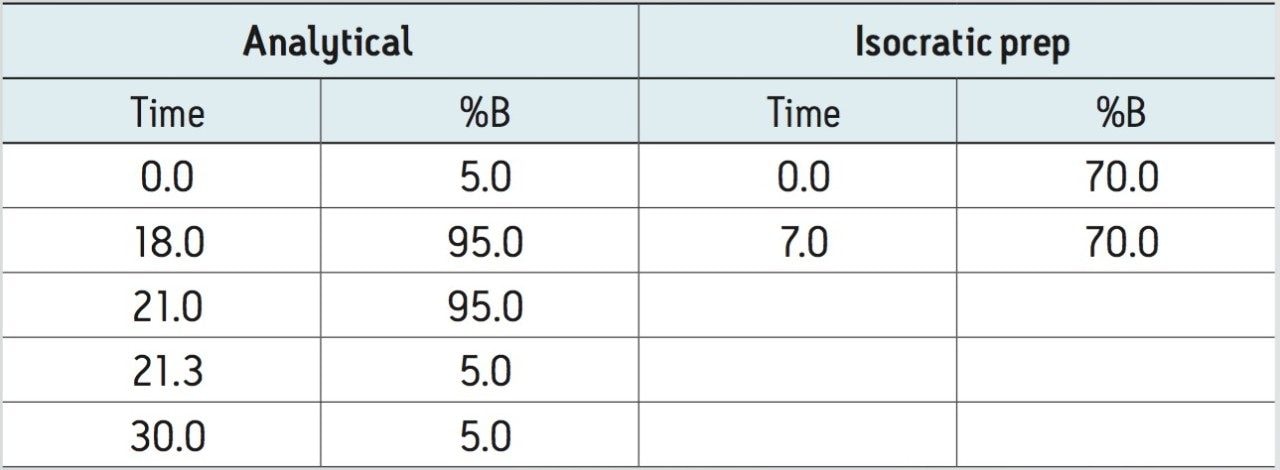
Table 2. LC methods used for the analysis and isolation of carnosic acid from rosemary.
The analytical flow rate was 1.46 mL/min, and the preparative flow rate was 25.00 mL/min.
The SFE experiment was performed on a Waters MV-10 ASFE System controlled by ChromScope Software. All LC experiments were performed on a Waters AutoPurification System controlled by MassLynx Software.
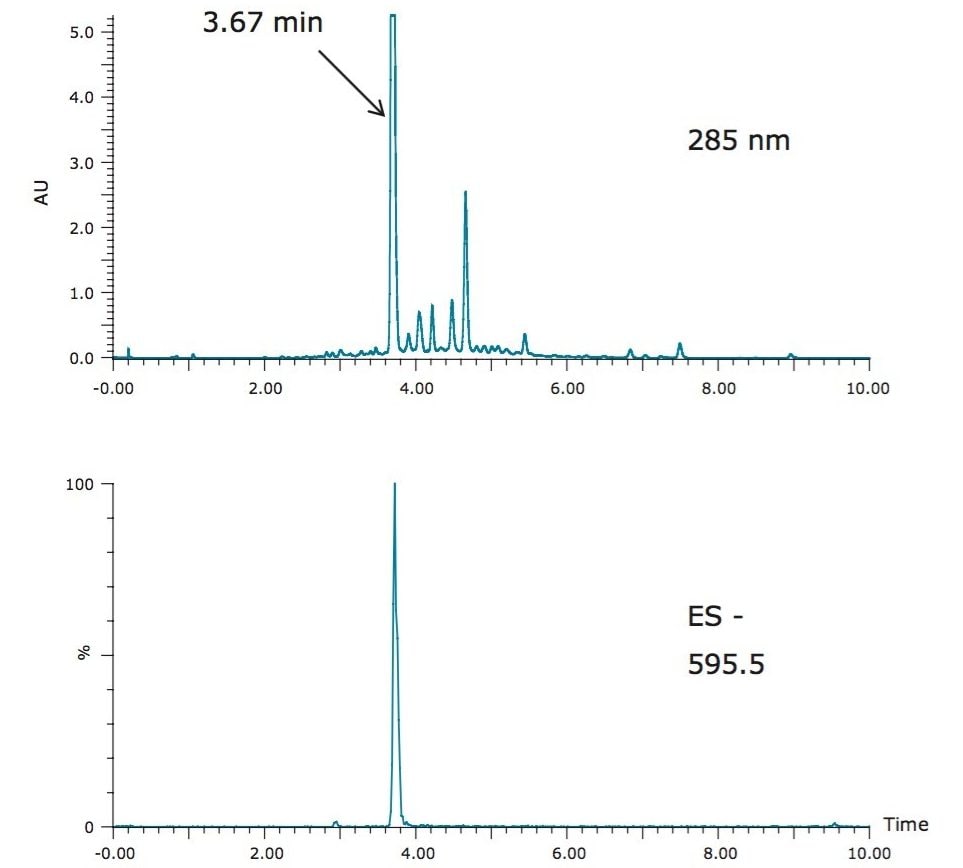
Eriocitrin, a flavonone compound rich in peppermint, has shown therapeutic effects against inflammation, allergies, and hypertension.7 Figure 2 shows the HPLC/UV chromatogram of eriocitrin using a generic gradient. Due to its polarity (LogP=-1.49), the target compound eluted from the column at 3.67 minutes.
The most common approach for target compound isolation is direct geometric scaling of the analytical gradient to the preparative column. A representative prep chromatogram using the direct geometrical scale-up is shown in Figure 3A. As noted in Table 1, the total run time for the preparative run was only slightly longer than the analytical one, due to the difference in system volume (primarily from the sample loop size difference) between the analytical and preparative systems.
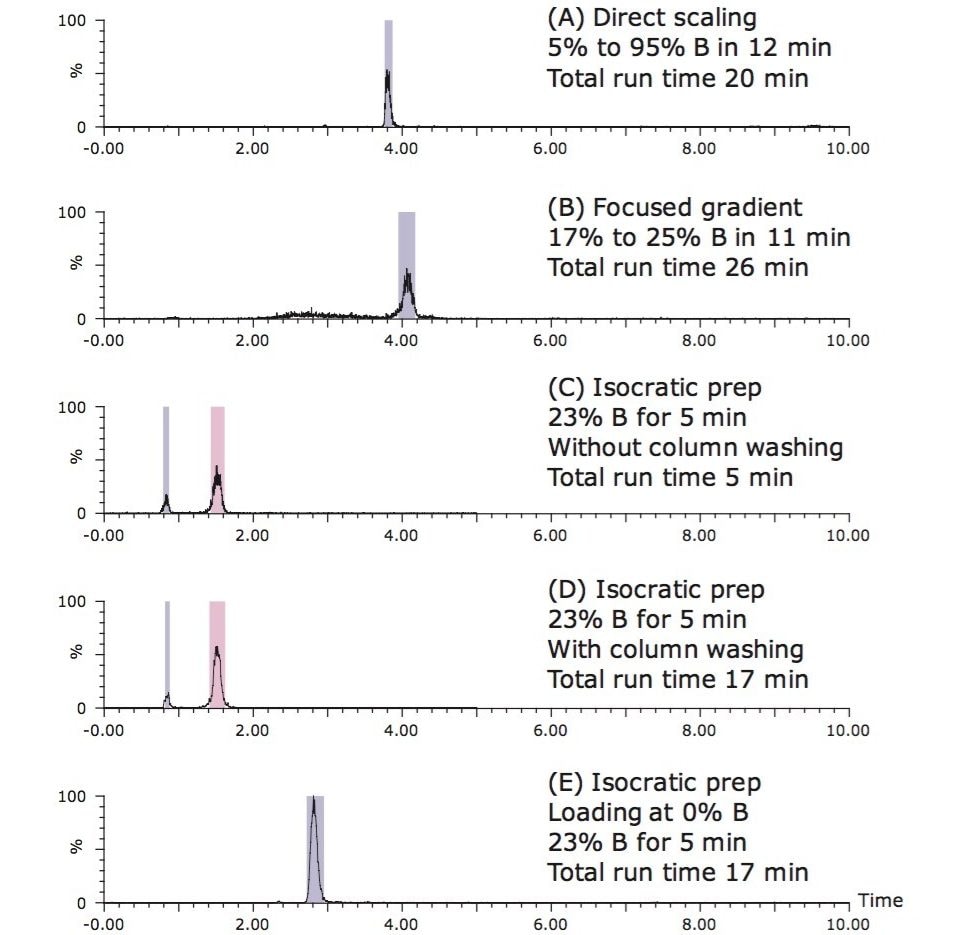
Alternatively, a focused-gradient approach9 can be used to isolate the target compound. Focused gradients are created with a shallow gradient segment centered around the %B where the target compound elutes from the column. A method for calculating the target compound elution percentage is as follows:
Based on the calculation, a focused gradient ranging from 17% B to 25% B in 10.7 minutes was developed. The slope of the focused gradient was 0.7% B per column volume, one-tenth of the original slope of 7.2% B per column volume. The shallow slope improved the resolution between the target compound and its adjacent peaks in the extract. In the preparative run using the focused gradient (Figure 3B), the target compound eluted off the column at ~4 minutes. The total run time for this method was 26 minutes, including column washing and equilibration.
The actual %B (23%) when the peak eluted in the analytical run was used for the isocratic preparative methods. Figures 3C through 3E show three different isocratic runs at 23% B. In addition to the eriocitrin peak at ~1.5 minutes, there was a small peak with a retention time of 0.8 minute, as shown in Figures 3C and 3D. This small peak contained a small amount of eriocitrin or its isomer, as well as other compounds. The presence of the small peak likely resulted from the breakthrough of the less retentive compounds, due to the loading at 23% B. To address the target compound loss due to the early eluting peak, the method was modified so that the initial loading condition was 0% B. After an initial one-minute hold at the loading condition, the %B was rapidly increased to 23% B. The resulting chromatogram is shown in Figure 3E. Clearly, the early eluting peak was eliminated. Injecting the sample at a lower %B in the method effectively mitigated the breakthrough of those less retentive compounds.
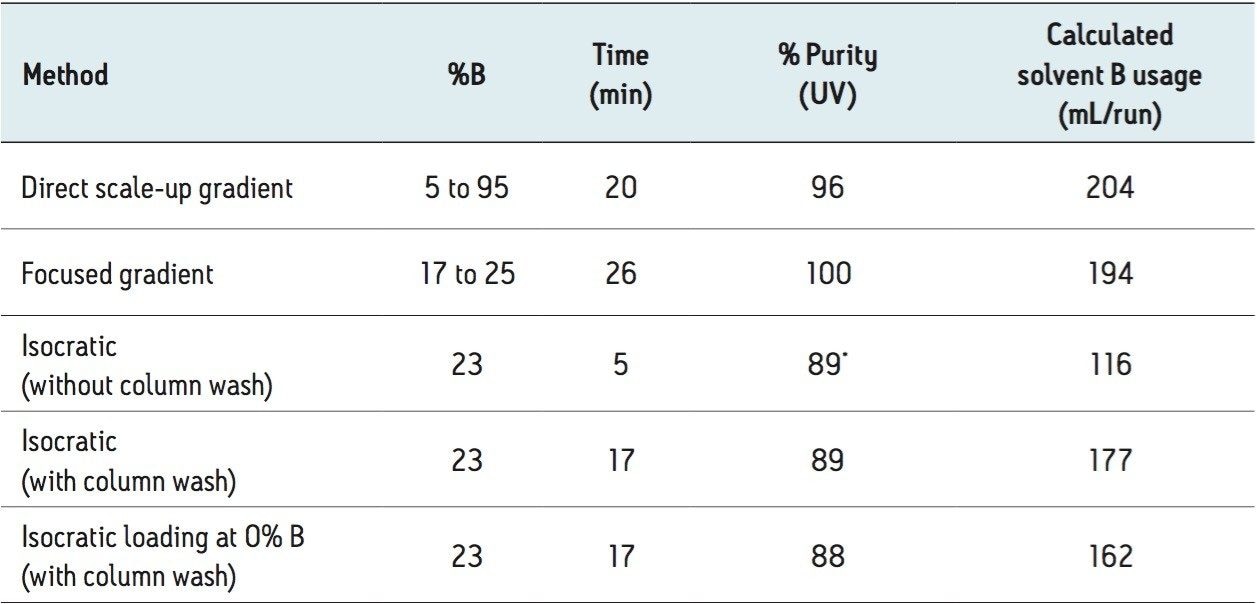
Table 3 summarizes the comparison of five different preparative methods for the eriocitrin purification. In terms of purity, both gradient methods resulted in >95% purity of the target compound, while the three isocratic methods led to a similar purity of 89%. Incorporating a column washing and an equilibration step to the isocratic prep method increased the total run time to 17 minutes (Figures 3D and 3E), but did not lead to a significant improvement in target compound purity. With respect to solvent usage, all isocratic methods consumed less solvent than the gradient methods. Compared to the directly scaled gradient and focused gradient methods, the five-minute isocratic method reduced the solvent use by 88 and 78 mL/run, respectively.
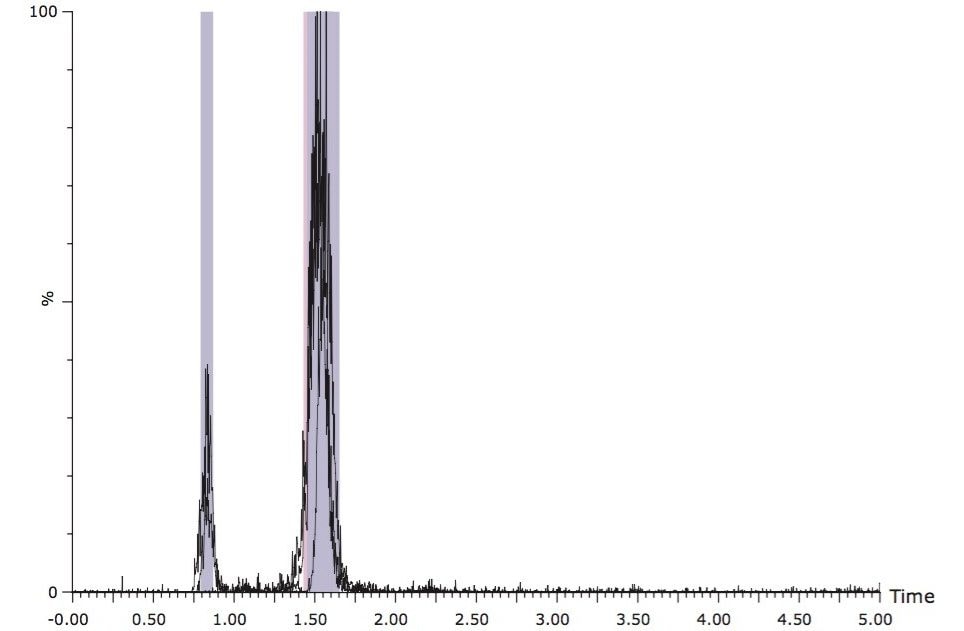
To gauge the ruggedness of the isocratic method, three replicates of the five-minute isocratic runs were performed. Figure 4 shows an overlay of three consecutive runs without column washing between injections. The results indicated that the isocratic methods were rugged and reproducible. Depending upon the assumed usage of the collected product, the described isocratic methods can be used as a high-throughput, first-step clean-up prior to more stringent purification, or as a final purification step. The isocratic methods improve productivity and cost-effectiveness due to the shortened run time and reduced solvent usage.
To ensure the general applicability of the described isocratic methodology, the same workflow was used for the isolation of carnosic acid, an anti-oxidant compound with a LogP of 5.14, found in the perennial herb, rosemary. Figure 5 shows the LC chromatograms of the rosemary SFE extract on an XBridge C18 5 µm, 4.6 x 150 mm Column, with a column volume of 2.093 mL. Note that a 30-minute screening gradient was used due to the column length (Table 2). The carnosic acid eluted at 14.9 minutes (Figure 5). Using the same procedure described in the previous section, the acetonitrile percentage at the elution of carnosic acid was determined to be 70%.
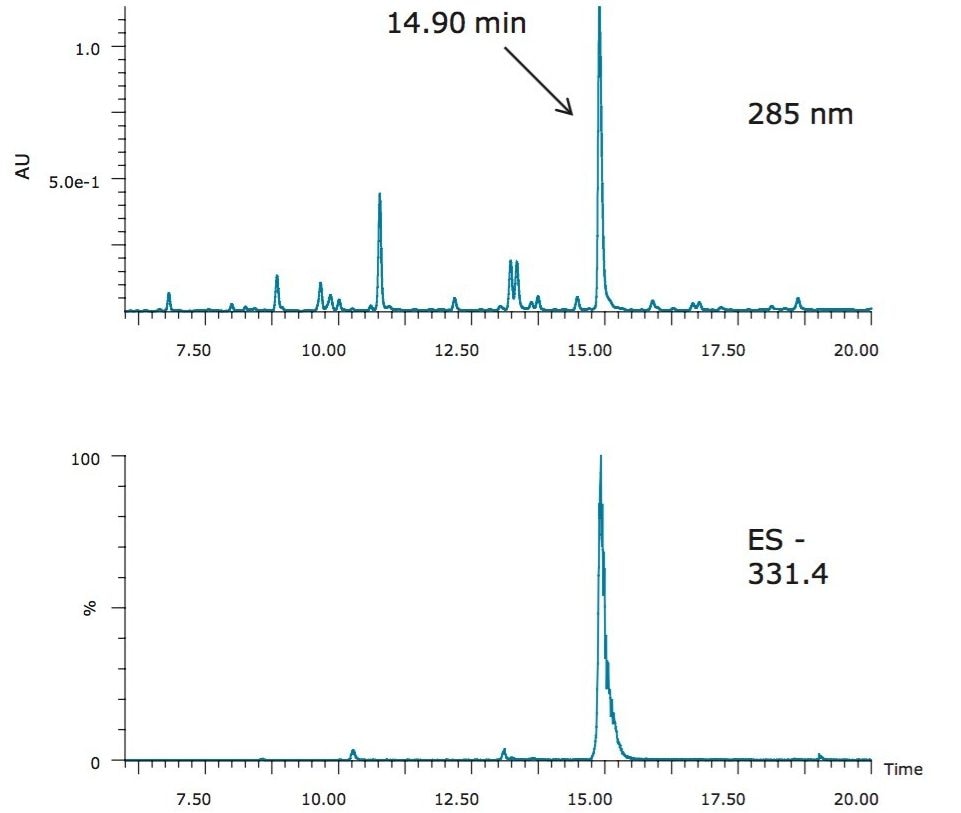
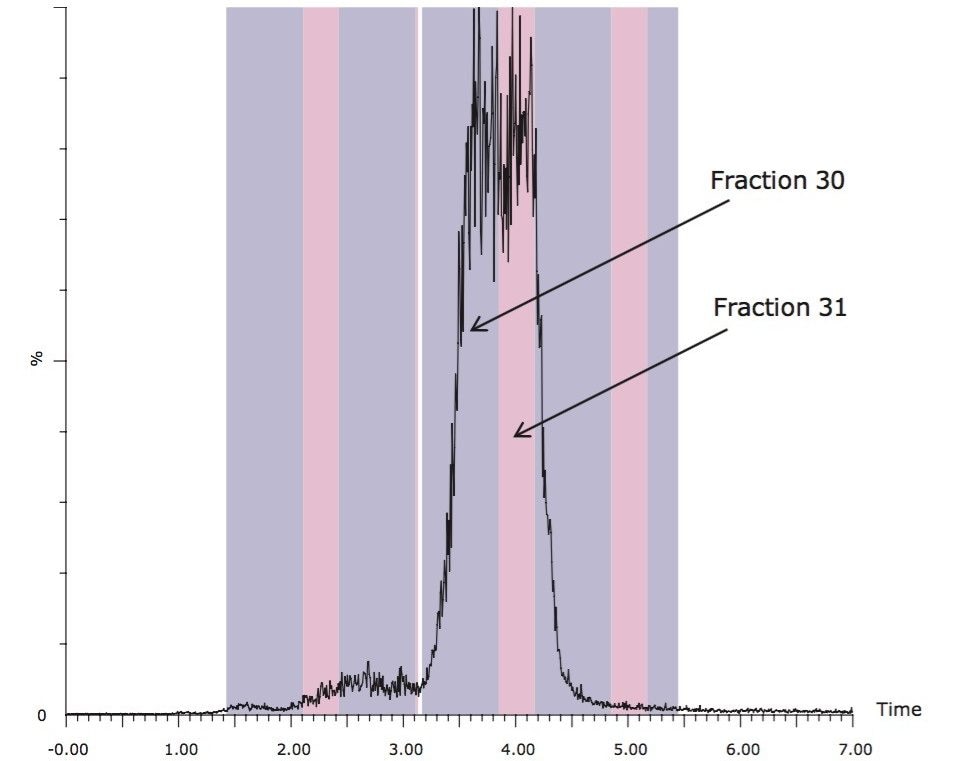
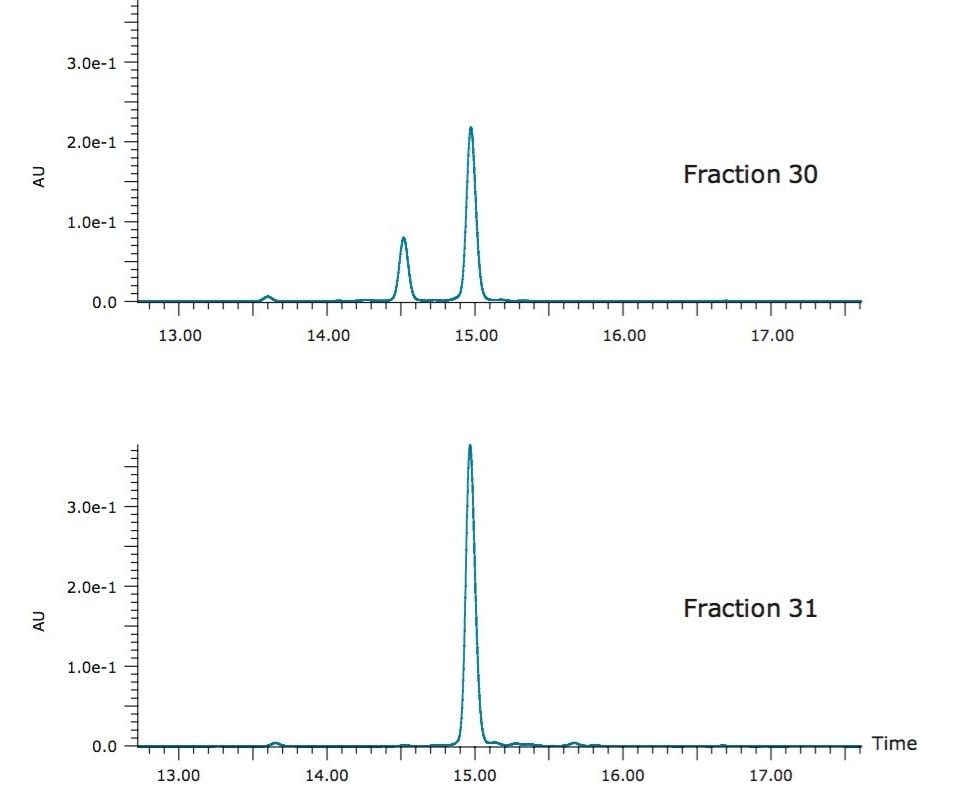
Figure 6 shows the isocratic preparative chromatography of the rosemary SFE extract on an XBridge C18 OBD Prep 5 µm, 19 x 150 mm Column run at 70% B. Because of the volume of the fraction, the target compound was collected into two tubes. Fraction analysis (Figure 7) revealed that fraction 30 was 73% pure, and fraction 31 was 100% pure by UV. For fraction 30, the impurity peaks at 13.6 minutes and 14.5 minutes account for 2% and 25% of the total area, respectively. However, these two impurity peaks were well resolved from the carnosic acid. A second purification step would remove these impurities with relative ease.
In this application note, a workflow to systematically develop isocratic prep LC methods for target compound isolation from complex natural product mixtures is demonstrated. The workflow starts with a generic LC analysis from which the elution %B of the target compound is calculated. The target compound is then isolated from the sample using an isocratic preparative LC method run at the calculated %B, leading to the collection of target compound with purities ranging from 73% to 100%. The isocratic prep LC methods were also proven rugged and reproducible. Compared to the commonly used gradient methods, the isocratic prep chromatography dramatically reduced the total run time and solvent usage, resulting in improved overall productivity and cost-effectiveness of the purification process. The proposed workflow is ideally suited as a quick, first step in a multi-step process for target compound isolation from natural products.
720004693, May 2013