In this application note we report a novel analytical technique of analyzing petroleum biomarkers in source rocks and crude oils using APGC-MS/MS.
Oil exploration and field development rely heavily on the recognition and use of ‘biomarkers’ in oils and source rocks for resolving questions regarding the source, preservation, and migrational history of oils and gases. These ‘biomarkers’ are molecular fossils that are stable under geological conditions. They are the ‘DNA’ of oils providing information not only on the biological source of the oil, but also on the environment in which the parent organic matter was deposited and on the thermal history of the buried organic matter. Knowledge of these properties is useful in a number of ways especially in upstream applications, and is ultimately linked to the economic productivity from a particular geological location. Providing the hydrocarbons have not been exposed to the atmosphere (and degraded) or to elevated temperatures, the biomarkers are stable throughout geological time. They provide a powerful tool for fingerprinting oils and source rocks and are invaluable in correlating oils to oils and to their parent source rocks.
Oils may derive from a number of sources. Principally oils can derive from organic matter deposited in terrestrial or aquatic environments. Terrestrial oils come from land plant derived material such as coals and resins. The biomarkers will reflect this with bicadinanes, indicating a tree resin input, whilst other biomarkers, oleanane and lupane for example, are associated with flowering plants. Oils from aquatic environments include those derived from marine organic matter contained within shales, carbonates and evaporates, or from lakes (lacustrine environments). Here, again, biomarkers will reflect the source organic matter input and the mineralogy of the source rock. 24-n-propyl cholestanes (C30 sterane), for example, are ubiquitous to marine environments but are absent from lacustrine or terrestrially derived oils. Similarly, significant concentrations of gammacerane indicate that the source rock of the oil was deposited in an environment of enhanced salinity.
Biomarkers are present in oil at low concentrations and are often masked by the much more abundant n-alkanes. Although routinely evaluated by gas chromatography-mass spectrometry (GC-MS) with electron ionization (EI), operated in selected ion recording (SIR) mode, the oil usually has to be separated into saturate and aromatic fractions prior to analysis. Even then, if n-alkanes are particularly prominent in the C25 to C35 range (the region in which the biomarkers elute), it may be necessary to remove the n-alkanes to pre-concentrate the biomarkers. The mass chromatograms from these analyses, m/z 217 for the steranes and m/z 191 for the hopanes, are then used in a geochemical appraisal of the oil. Whilst these analyses are invaluable, some information is lost due to either the very low abundance of the biomarker of interest (C26 and C30 steranes) or of co-elution of different markers (identification of the steranes can be difficult if the hopanes are many more times abundant than the steranes). The great complexity of these samples and the relatively low abundance of the biomarkers mean that gas chromatography-tandem mass spectrometry (GC-MS/MS) analyses are often required to answer specific geochemical questions. C30 steranes are ubiquitous to marine source oils. However, their low abundance and the fact that they co-elute with 4-methyl steranes mean that they can only be detected and quantified accurately with MS/MS.
Just as MS/MS has been shown to improve the determination of these compounds for mass analysis, use of atmospheric pressure (AP) ionization demonstrates specific advantages over EI. Atmospheric Pressure Gas Chromatography (APGC) generates molecular ions, M•+, but of much higher relative abundance than EI. This improves the selectivity of the first stage of MS and delivers a highly abundant molecular ion to the collision cell for controllable fragmentation in the MS/MS process. This is in contrast to the fixed fragmentation and poor molecular ion abundance in EI. Since the same form of the molecular ion is used in both EI and APGC under charge exchange conditions, the MRM transitions used for APGC-MS/MS and GC-MS/MS are exactly the same facilitating correlation of historical data to this new approach.
In this application note we report a novel analytical technique of analyzing petroleum biomarkers in source rocks and crude oils using APGC-MS/MS.
Asphaltenes were removed from the oil samples by precipitation with excess n-heptane.
|
System: |
Agilent 7890 and 7683B autosampler |
|
Column: |
Restek Rxi-5Sil MS 30 m x 0.25 mm, 0.25 μm |
|
Carrier gas: |
Helium at 1 mL/min |
|
Temperature program: |
50 °C for 2 min 10 °C/min to 180 °C 2.5 °C/min to 315 °C, hold 11 min |
|
Injection port: |
300 °C |
|
Injection type: |
1 μL splitless, 1.5 min purge |
|
Makeup gas: |
Nitrogen at 400 mL/min |
|
Transfer line: |
350 °C |
|
System: |
Xevo TQ-S |
|
Source: |
APGC |
|
Corona current: |
2 μA |
|
Source offset: |
50 V |
|
Source temp.: |
150 °C |
|
Cone gas: |
Nitrogen at 150 L/hr |
|
Auxillary gas: |
Nitrogen at 300 L/hr |
|
Acquisition: |
Multiple reaction monitoring (MRM) |
Ionization using APGC is analogous to atmospheric pressure chemical ionization (APCI) insomuch as molecular ions are produced. APGC is a ‘soft’ ionization technique which results in lower fragmentation. The presence of strong molecular ions gives ideal conditions for MS/MS analysis.
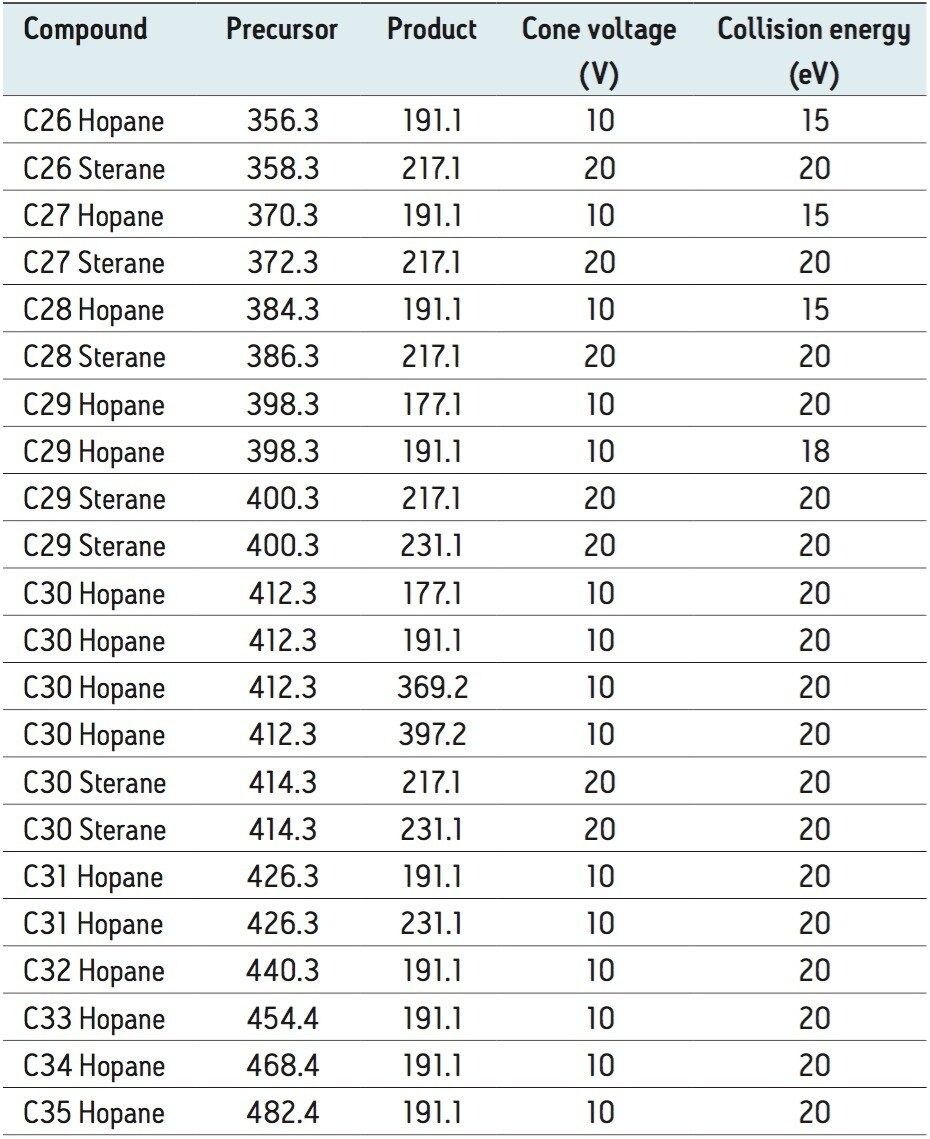
Figure 1 shows the comparison between the APGC charge transfer spectrum for a C30 pentacyclic triterpane from a marine source crude oil and an EI spectrum. Extensive fragmentation is observed in EI mode resulting in high abundance, low mass fragments. These make poor precursor ions for a MRM transition and are less selective. The molecular ion is present but a loss of sensitivity would have to be accepted if this was chosen for the precursor ion. Less fragmentation is observed in APGC mode and the molecular ion is the base peak. For MRM analysis the ability to obtain an intense and specific precursor ion is critical in obtaining low detection limits.
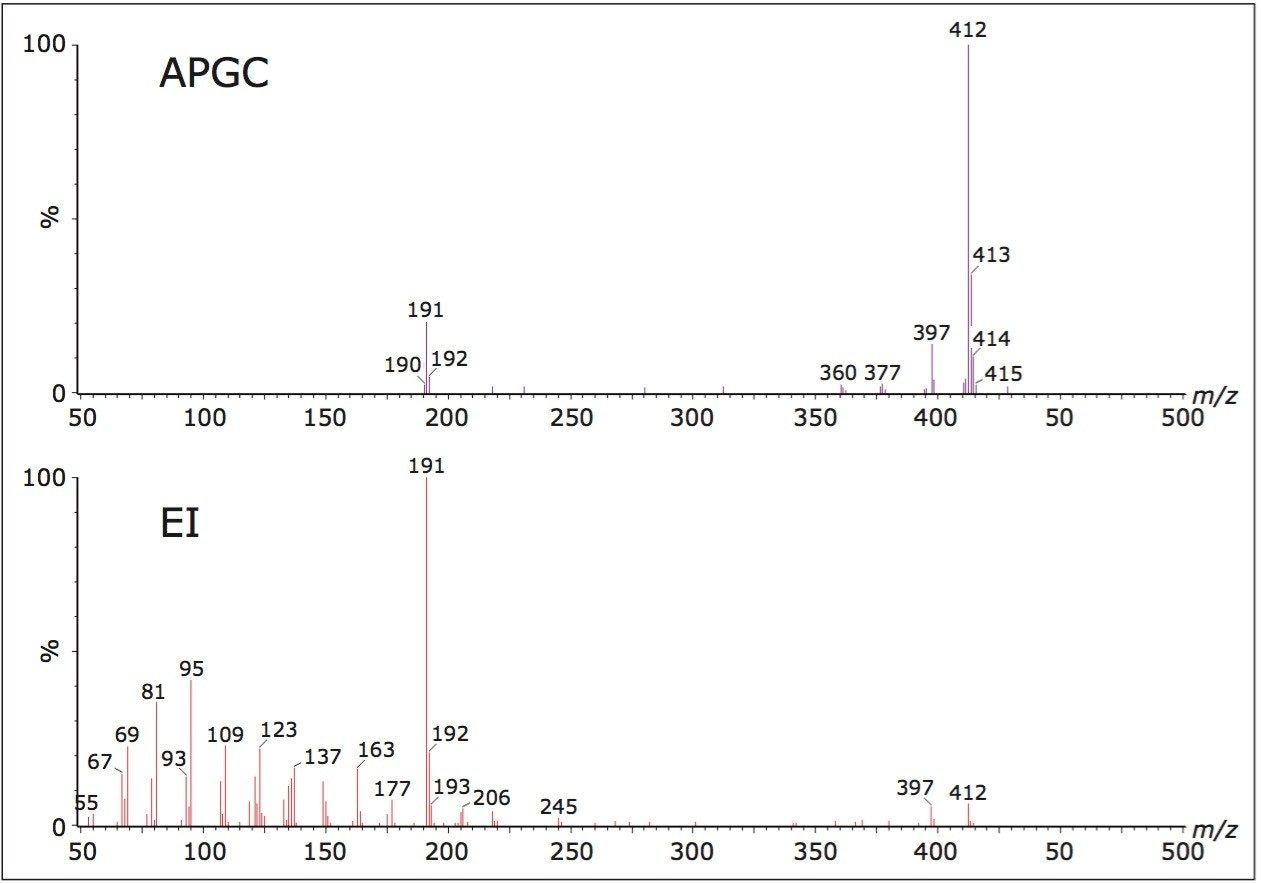
The low concentration of petroleum biomarkers in high maturity oils mean that their identification and quantification often require the use of highly sensitive and selective mass analysers. Figure 2 illustrates the typical MS fragments observed for hopanes and steranes. MS/MS reduces matrix interference and allows the geochemist to monitor specific precursor-product transitions providing information that is unavailable from GC-MS in SIR mode. Figure 3 shows the regions where C26 and C30 steranes should elute and the problems associated with low abundance of these components along with the co-elutions of the C27-C29 steranes using GC-MS in SIR mode.
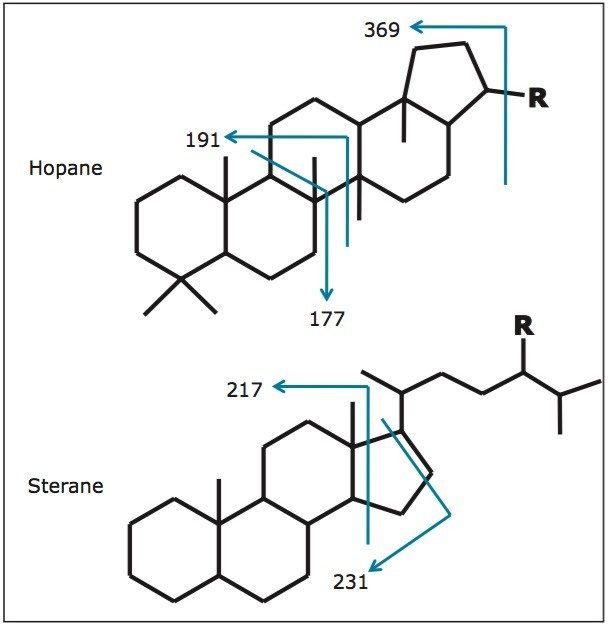
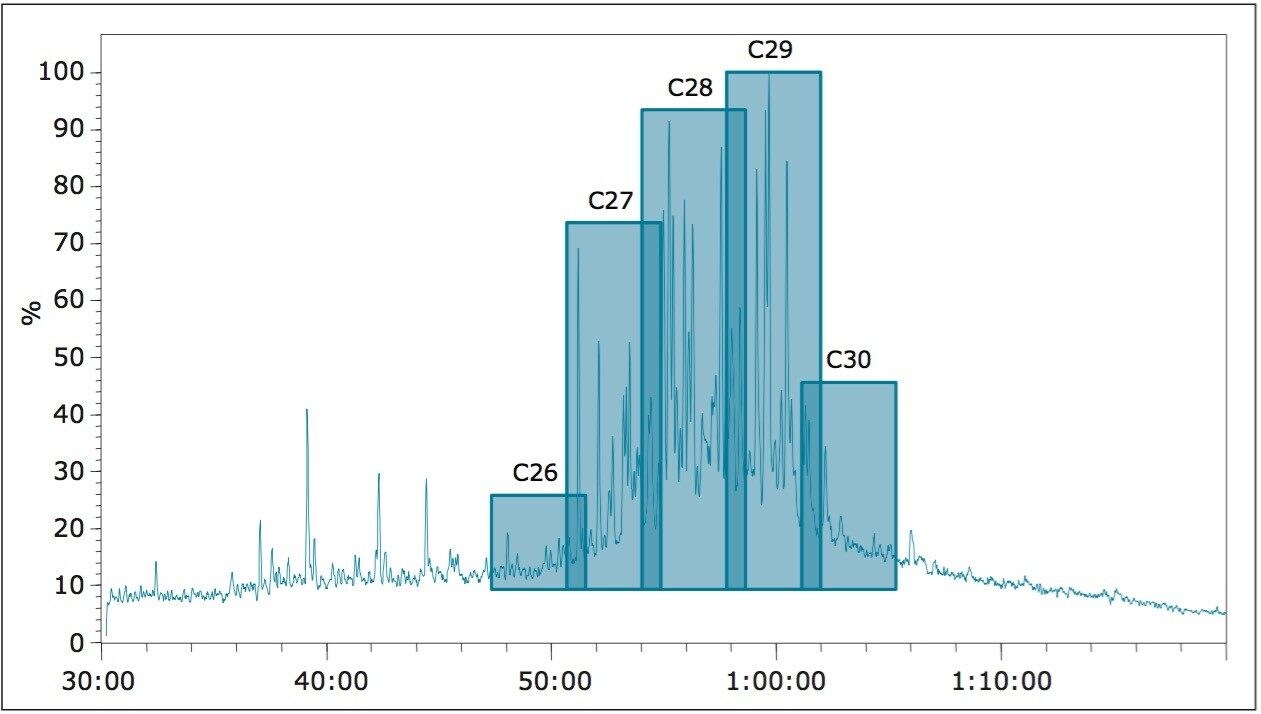
One of the most common uses of GC-MS/MS in biomarker analysis is for the detection and quantification of the steranes. These are used to define:
1. If the oil is from a marine or non-marine source by assessing the presence or absence of the C30 steranes
2. The age of the source rock that generated the oil by assessing the C28/C29 sterane ratio along with the C26 steranes.
The example illustrated in Figure 4 is from a UK North Sea oil which has been sourced from marine source rocks of Jurassic age. By analysing in MS/MS mode the geochemist can not only confirm a marine origin (presence of C30 steranes) but can use both the C28/C29 sterane ratio (0.41) and the 24-nordiacholestane or NDR ratio (Peaks 1+2/1+2+3+4 = 0.2) to confirm that the oil has a Jurassic source. The selectivity of MS/MS makes quantification easy as no co-elution occurs between the components.
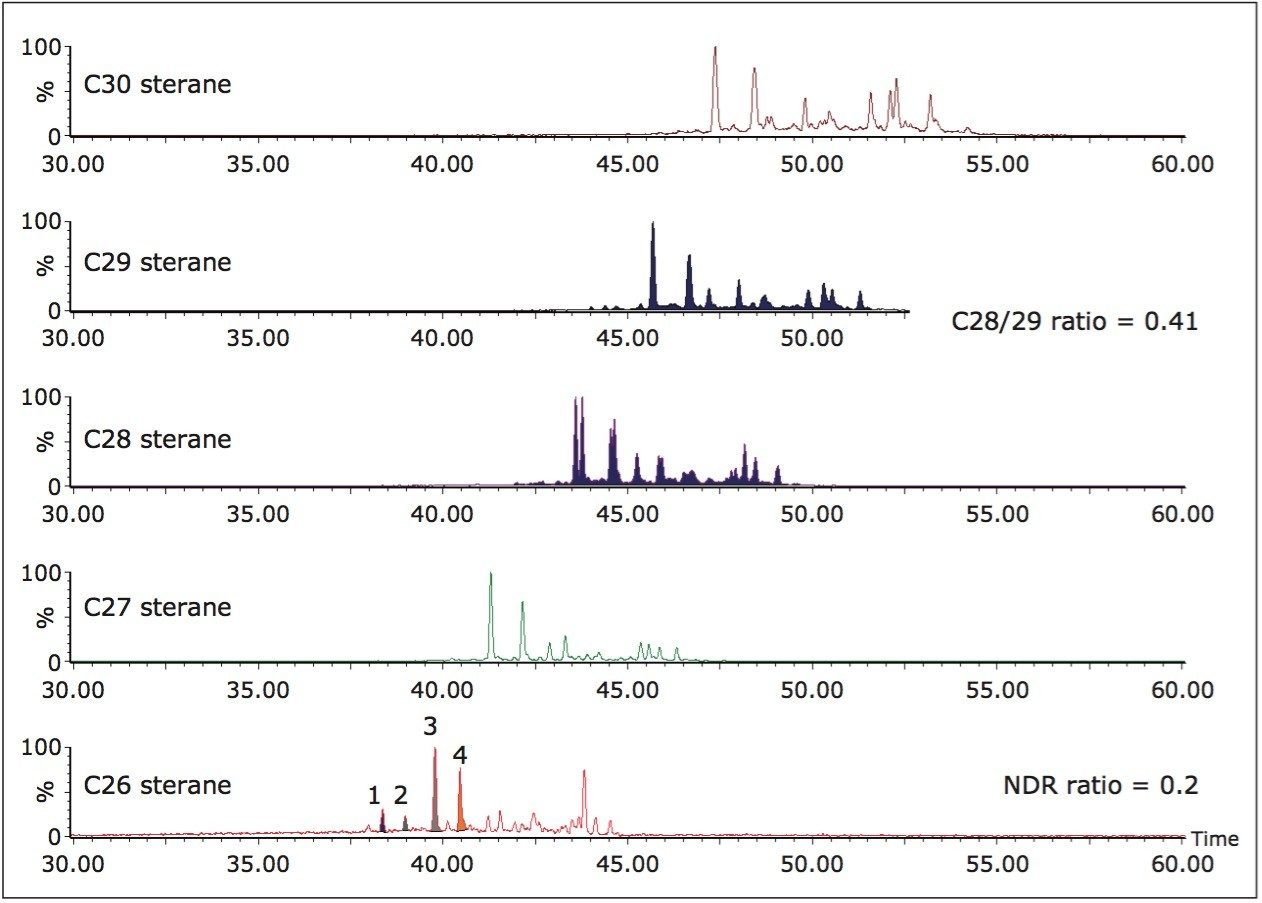
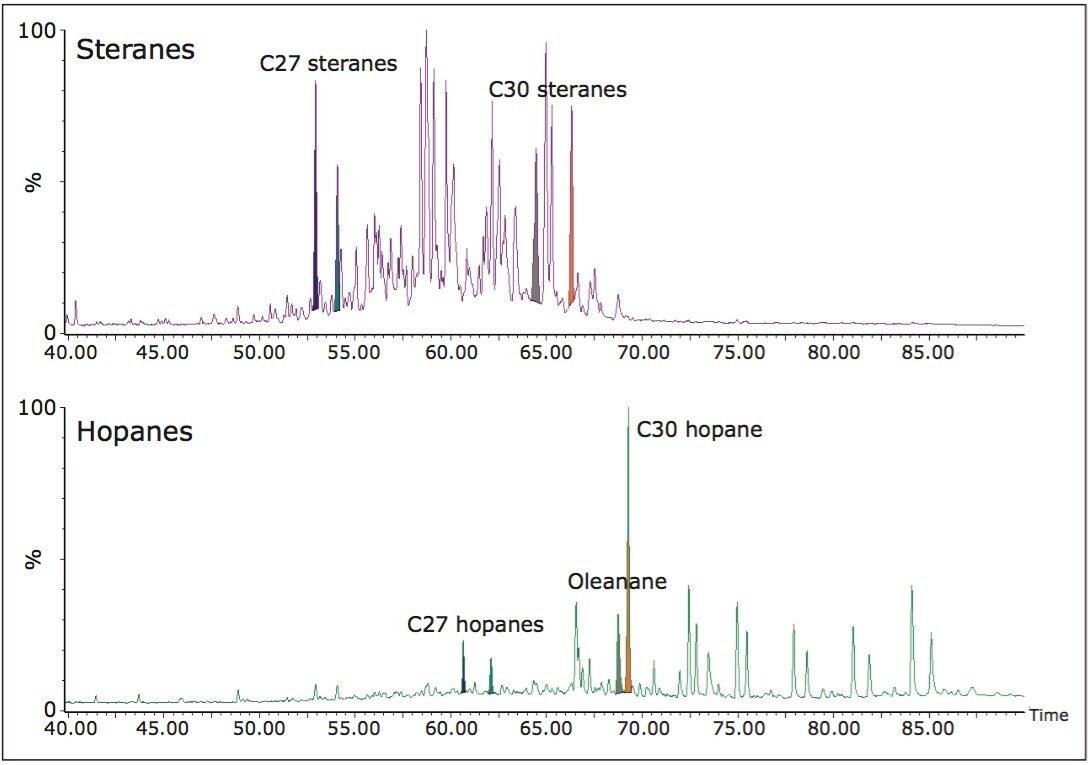
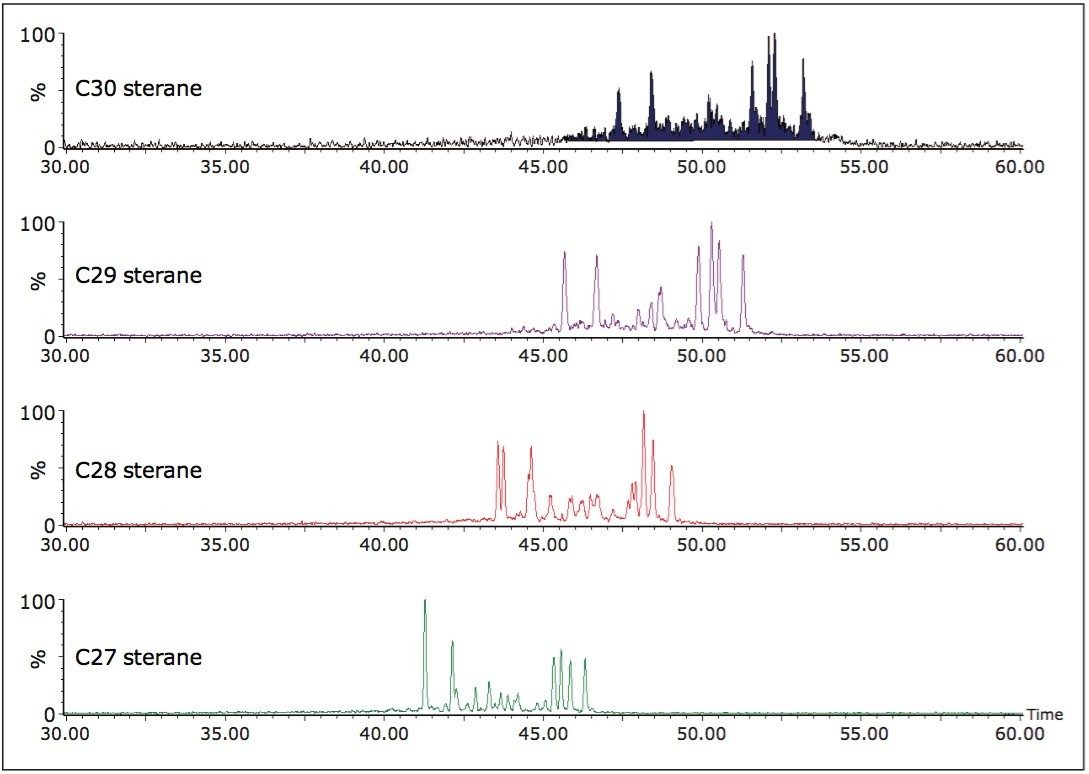
Figure 5 shows the sterane and hopane GC-MS SIR data for a mixed input oil from the Cote D’Ivoire. Here, stacked reservoirs contain oils that derive from source rocks deposited under marine conditions but with a significant contribution from terrestrially derived organic matter. An examination of the biomarkers from these data initially provided an interpretation of terrestrially derived oil because the oils were characterized by abundant markers indicating a fluvio-delatic depositional environment for the parent source rock. These markers included oleanane (derived from higher plants) and abundant C29 steranes (significant contribution from land plants). The identification of C30 steranes, illustrated in Figure 6, is only possible by APGC-MS/MS and confirmed the marine contribution to the oils parent source rock
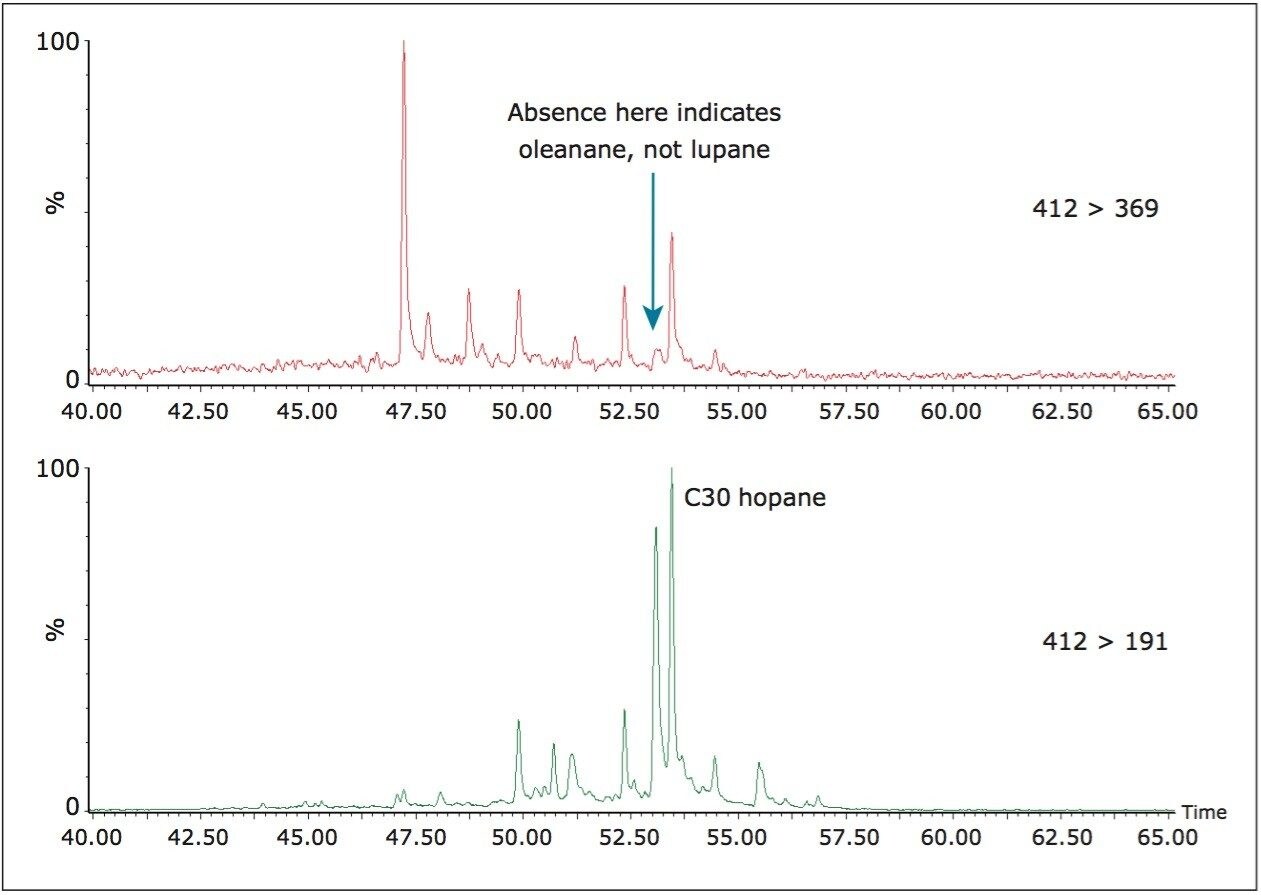
Non-polar GC columns are most commonly used for GC-MS analysis of petroleum biomarkers, and one feature of this is the peak eluting just prior to the C30 hopane, that is usually identified as oleanane. Oleanane has the same precursor ion (m/z 412) as the C30 hopane, but a slightly shorter retention time. However, lupane has the same precursor mass and very similar mass spectrum to oleanane, except for the presence of a product ion of m/z 369. The importance of lupane as a geochemical marker is poorly understood at present but this is largely because it is being identified as oleanane. These compounds can be adequately separated by reverse-phase HPLC but if a single analytical system is preferred, APGC-MS/MS can assess whether the peak eluting just before the C30 hopane is oleanane or lupane. Figure 7 shows an example of a saturated fraction with a peak eluting just before the C30 hopane. By monitoring the second transition we can immediately see that there is no peak eluting at the same retention time. This shows that oleanane has been correctly identified and that the sample analyzed does not contain lupane.
APGC is a soft ionization technique which produces lower levels of fragmentation compared to traditional EI. Working in dry conditions, APGC typically produces spectra with strong radical cation molecular ions that facilitates the direct comparison of historic, archival EI data from many different sources. The abundant nature of these ions makes it possible to generate specific and sensitive MRM transitions for petroleum biomarkers which provide information on the biological source of the oil, the environment in which the parent organic matter was deposited, and the thermal history of the buried organic matter. Knowledge of these properties is especially useful in upstream applications and is ultimately linked to the economic productivity from a particular geological location.
The universal ionization source offered on all Waters instrumentation facilitates the quick and simple coupling of APGC, UPLC or UPC2 on a single MS platform. Coupled with Xevo TQ-S, APGC enables low-level quantification of petroleum biomarkers that have proved challenging to analyze using GC-MS systems, allowing unambiguous confirmation of the deposition environment from which the oil originates.
720004645, March 2013