
For research use only. Not for use in diagnostic procedures.
This method demonstrates the separation power of convergence chromatography utilizing either UV or MS detection. While the limits of detection and quantification presented here are not compatible with the low levels of steroid concentrations typically found in biological samples (e.g. plasma), additional optimization of MS parameters, with the possibility of additional sample derivitization to improve ionization, may help to reach higher sensitivity levels. This could further enable the application of this method to the analysis of steroids for clinical research.
Steroid biosynthesis is a complex metabolic pathway utilizing simple precursors to synthesize multiple steroidal forms. This biosynthetic pathway is unique to animals and provides a common target for antibiotics and other anti-infective drugs. Precise and accurate steroid analysis is critical for the development of steroid-based therapeutics. For mass spectrometric analysis of steroids, due to their structural similarity, chromatographic separation of the steroids is essential prior to analysis. Typical research analyses utilize either gas (GC-MS) or liquid (LC-MS) chromatographic methods. GC-MS methods require sample derivitization prior to analysis resulting in analysis times of approximately 40 minutes.1 For LC-MS methods, typical analysis times are approximately 12 minutes for HPLC, or four to five minutes with the use of more recent UHPLC methods.2,3
This study focuses on the application of convergence chromatography (CC), utilizing liquid CO2 as the primary mobile phase, for the rapid chromatographic analysis of endogenous steroids (structures shown in Figure 1).
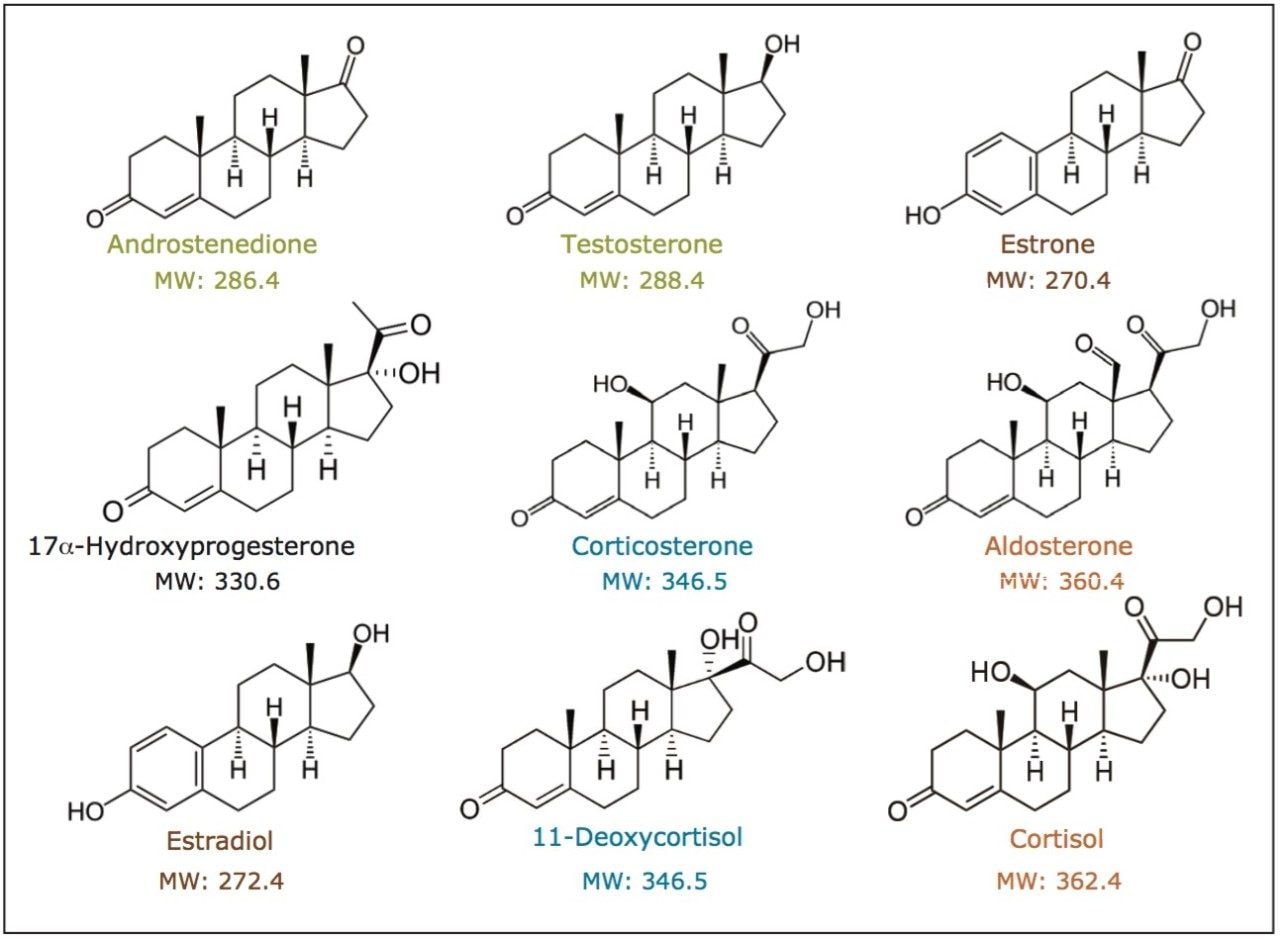
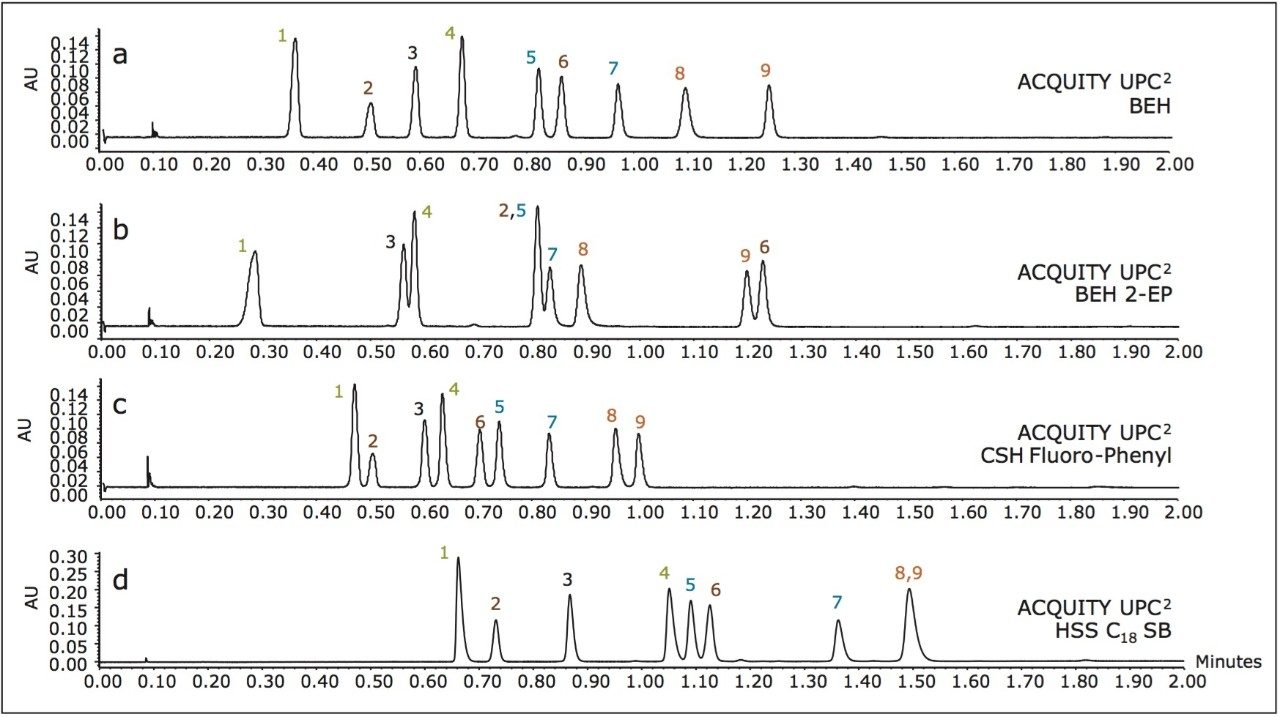
Column screening (UV detection): A mixture of nine steroid standards was prepared at 0.2 mg/mL each, using a diluent of 88:12 methanol/ethanol.
Mass spectrometer evaluations: Often times, matrix interferences can limit the applicability of a technique. For this reason, standards were evaluated in a human plasma matrix. However, to insure these evaluations were indicative of technique sensitivity and not affected by recovery issues during sample preparation, the plasma samples were post-spiked after a 3:1 acetonitrile protein crash of the plasma. After centrifugation, the supernatant was collected and spiked with a mixture of the nine steroid standards. Spiking of steroids to various levels was achieved by serial dilution of the sample with additional crashed plasma.
|
Screening columns: |
ACQUITY UPC2 BEH 1.7 μm, 3.0 x 50 mm (p/n 186006562) ACQUITY UPC2 BEH 2-Ethylpyridine 1.7 μm, 3.0 x 50 mm (p/n 186006580) ACQUITY UPC2 CSH Fluoro-Phenyl 1.7 μm, 3.0 x 50 mm (p/n 186006571) ACQUITY UPC2 HSS C18 SB 1.8 μm, 3.0 x 50 mm (p/n 186006621) |
|
UPC2 conditions |
|
|---|---|
|
System: |
ACQUITY UPC2 with PDA detector |
|
Mobile phase A: |
CO2 (tank, medical grade) |
|
Modifier B: |
Methanol (Fisher Optima grade) |
|
Column temp.: |
40 °C |
|
ABPR: |
1800 psi |
|
Gradient: |
2% to 17% modifier B in two minutes, re-equilibration at 2% modifier B for one minute |
|
Flow rate: |
3.65 mL/min |
|
UV detection: |
220 nm (compensated 380 to 480 nm) [40 pts/s] |
|
Injection volume: |
1 μL |
|
Needle wash: |
50:50 methanol/2-propanol |
|
Seal wash: |
Methanol |
Empower 3 Software
|
Solvent: |
Methanol with 2.5% water and 0.1% ammonium hydroxide |
|
Flow rate: |
0.4 mL/min |
|
Mass spectrometer: |
Xevo TQ |
|
Capillary voltage: |
1 kV |
|
Desolvation temp.: |
500 °C |
|
Desolvation gas flow: |
750 L/h |
|
Data management: |
MassLynx Software |
Here, we present data collected with the ACQUITY UltraPerformance Convergence Chromatography (UPC2) System. In combination with stationary phases designed specifically for UPC2, based on the bridged ethylene hybrid, BEH Technology, this technique results in the analysis of steroids in approximately two minutes. After initial method development using UV detection, the system was coupled to a tandem quadrupole mass spectrometer for analysis of steroid-spiked plasma samples. In addition to the significant reduction in analysis time relative to other techniques, convergence chromatography minimizes the consumption of mobile-phase solvents (e.g., methanol), thereby generating less waste for disposal and significantly reducing the cost of analysis per sample.
A generic two-minute screening gradient was used to evaluate the separation of the nine-steroid mixture on four different stationary phases to determine which would provide the best separation. The chromatograms in Figure 2 demonstrate the selectivity differences of the ACQUITY UPC2 stationary phases, as well as the inherent speed of this chromatographic technique. Based on these results, the ACQUITY UPC2 BEH stationary phase was chosen for additional application development with mass spectrometric detection.
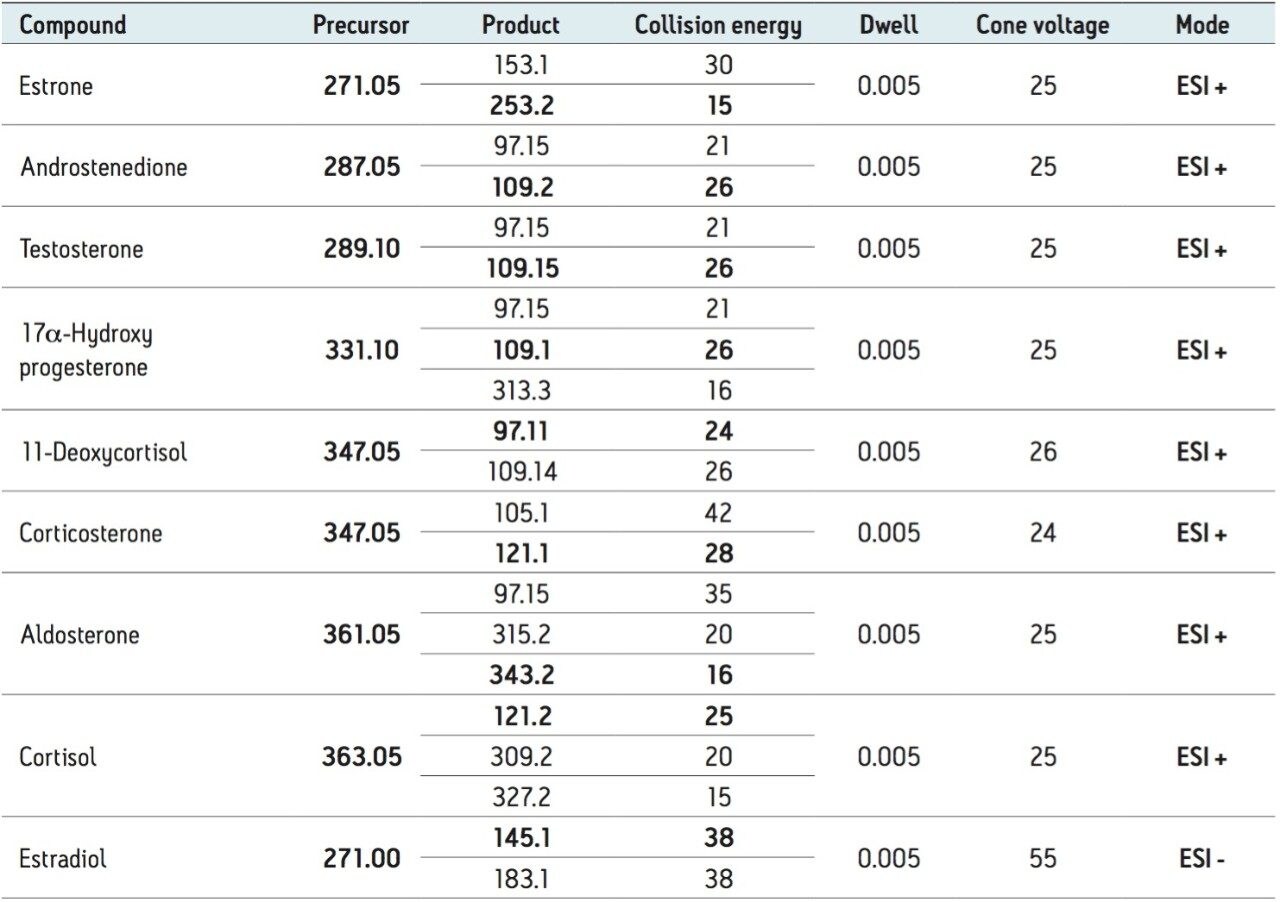
Individual mass spectrometer (MRM) transitions were optimized by direct infusion of standards into the Xevo TQ MS using on-board fluidics, without the connectivity of the ACQUITY UPC2 System (Table 1). After optimization of transitions, the mass spectrometer was coupled to the UPC2 system using a mass spectrometer splitter, incorporating the addition of a make-up flow pump, to facilitate sample flow into the MS and subsequent ionization (Figure 3).
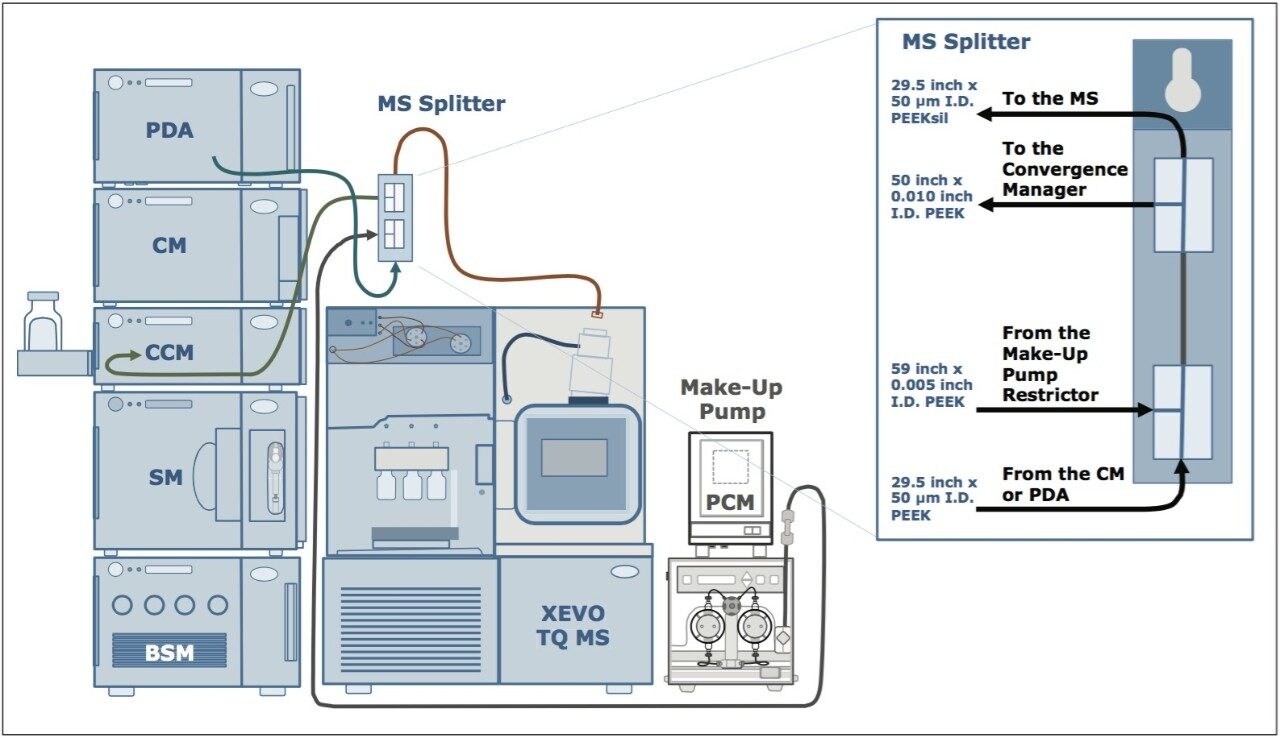
The make-up flow introduced through the mass spectrometer splitter has a dual purpose. It facilitates the post-mixer transfer of the sample through the tubing, as the CO2 in the mobile phase starts to decompress as it reaches the mass spectrometer. This is especially important at low concentrations of the organic modifier in the mobile phase, as seen in the early stages of the current gradient profile. In addition, the use of additives in the make-up flow (such as water, ammonium hydroxide (NH4OH), or formic acid (FA)) can assist in ionization of the analytes within the mass spectrometer source, thereby improving sensitivity. To optimize the make-up flow and additional MS conditions, a plasma sample spiked with the nine steroids (at 50 ng/mL) was used to evaluate various conditions including: additive used in make-up flow solvent, capillary voltage, desolvation temperature, and gas flow. The results of those evaluations are shown in Figure 4.
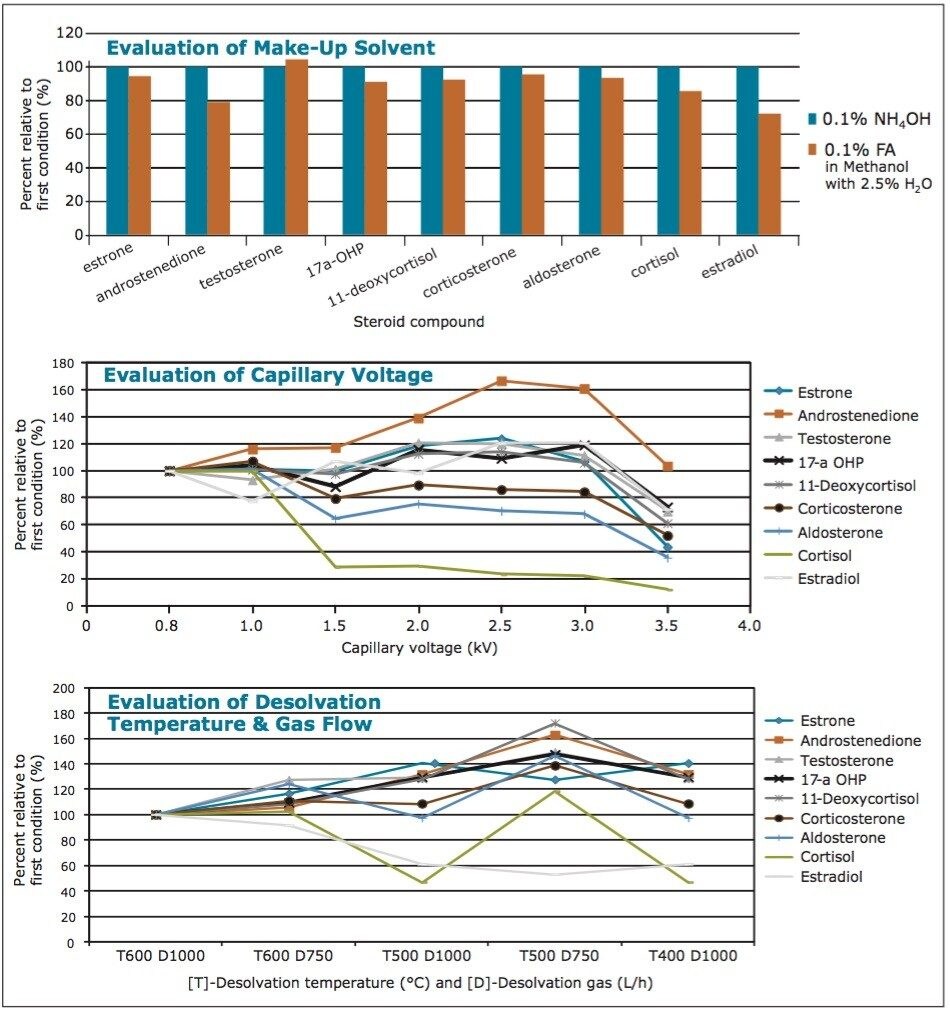
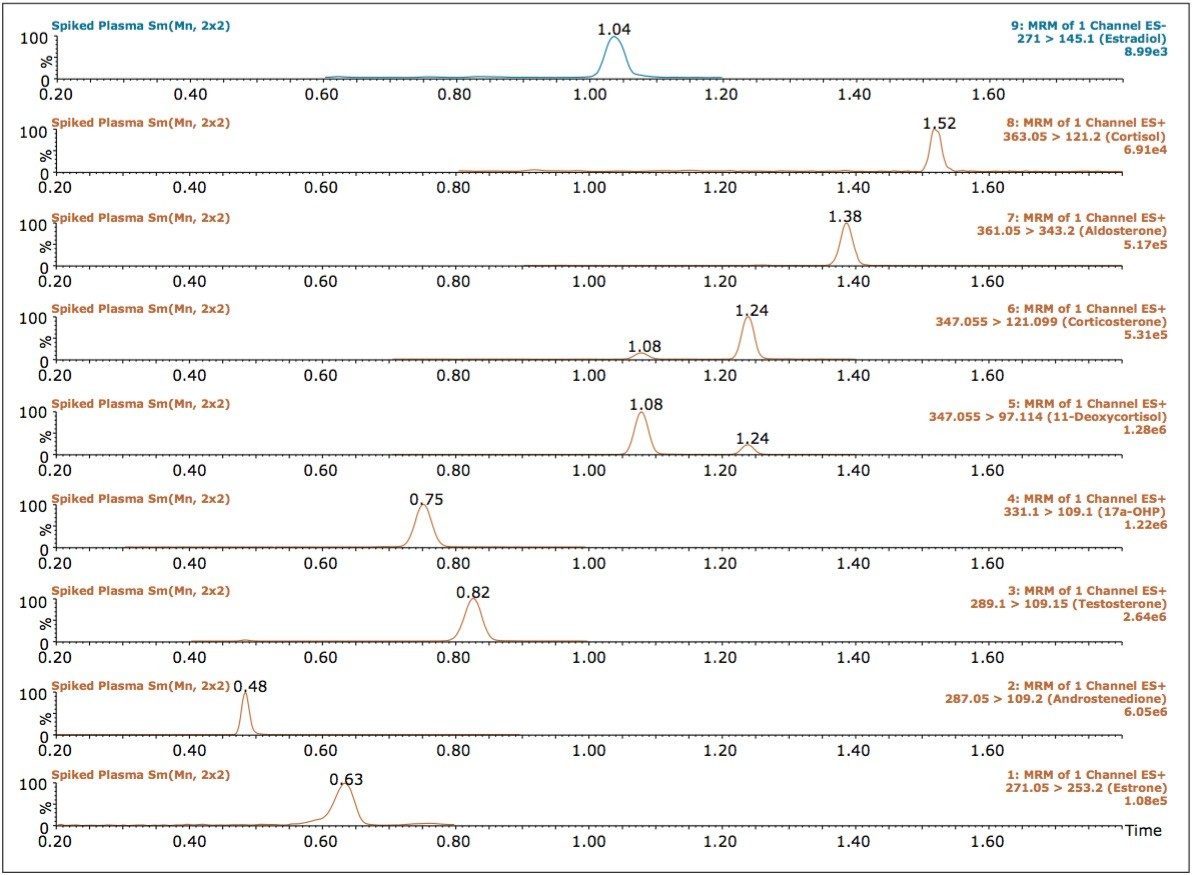
The top panel of Figure 4 shows eight of the nine steroids yielded higher MS signals (better ionization) when using ammonium hydroxide as an additive in the make-up flow. In addition, most of the steroid signals were ambivalent to capillary voltages between 0.8 and 3.0 kV, as shown in the middle panel of Figure 4. However, at voltages higher than 1.0 kV, the signal for cortisol diminished dramatically. Based on these evaluations, the optimum conditions were determined, with the best overall signal obtained for all steroids using a make-up solvent composed of methanol with 2.5% water and 0.1% ammonium hydroxide, and a flow rate of 0.4 mL/min. The optimum results were obtained by MS when using a capillary voltage of 1.0 kV, with a desolvation temperature of 500 °C, and a gas flow of 750 L/h (bottom panel of Figure 4). The resulting chromatography for the nine steroids post-spiked into the human plasma after protein crash is shown in Figure 5.
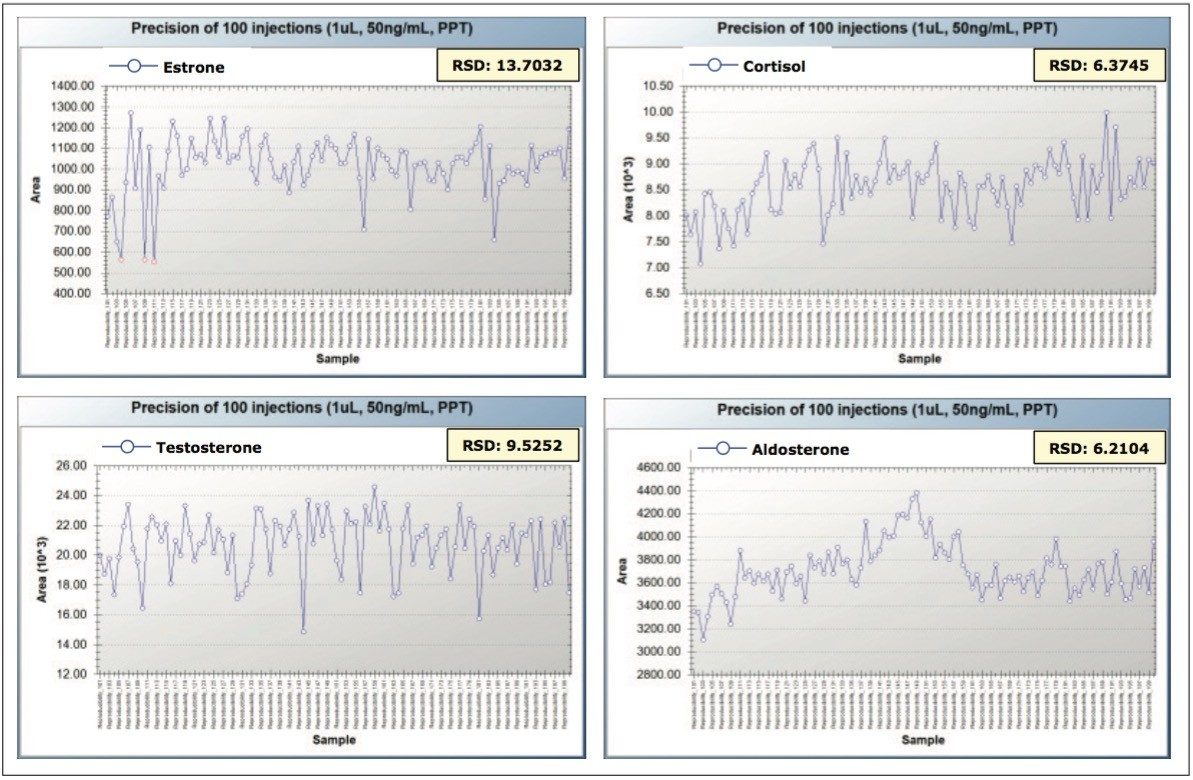
As in any method development, the accuracy and reproducibility of the method is critical for success. To evaluate reproducibility of the method, the peak areas for the individual steroids were monitored over the course of 100 injections (using 1-μL injection volumes of 50-ng/mL steroid spiked in plasma). The RSD values for the peak areas ranged from 5.6% to approximately 13.7%. A representative example of the reproducibility results are shown in Figure 6. Similar results were obtained for the other steroids evaluated.
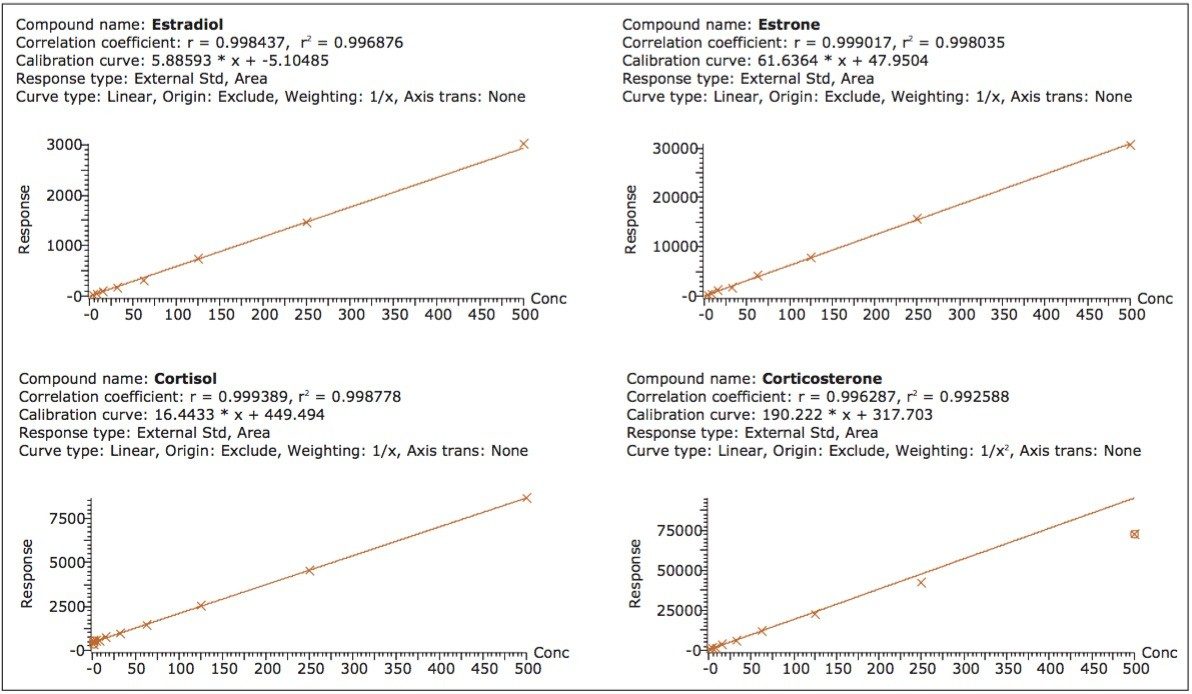
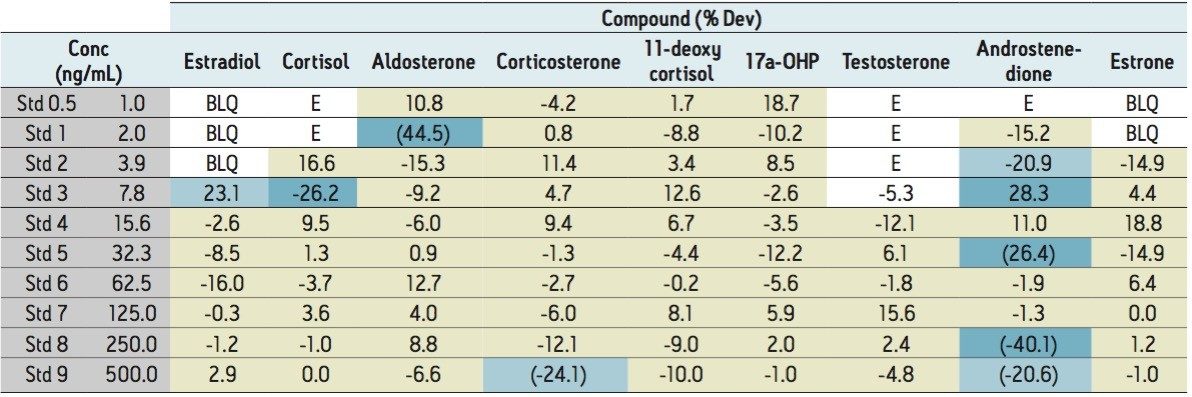
To evaluate the linearity of response, calibration curves were generated using 5-μL injections of the spiked steroid plasma samples (after 3:1 acetonitrile protein crash). Concentrations of the steroids ranged from 0.98 to 500 ng/mL. Representative calibration curves are shown in Figure 7, with more complete data for each of the steroids shown in Table 2.
Convergence chromatography enables fast, accurate analysis of steroids with reduced analysis times relative to current LC and GC methodologies. UPC2 offers scientists a unique workflow, application, and environmental impact benefits, compared to LC and GC platforms, with simplified sample preparation. Samples extracted in organic solvents can be injected without additional steps to exchange solvents for RP-compatible diluents. In addition, with CO2 as the primary mobile phase, the cost of analysis per sample is reduced, using a more environmentally friendly solvent relative to conventional RP methods. This method demonstrates the separation power of convergence chromatography utilizing either UV or MS detection. While the limits of detection and quantification presented here are not compatible with the low levels of steroid concentrations typically found in biological samples (e.g. plasma), additional optimization of MS parameters, with the possibility of additional sample derivitization to improve ionization, may help to reach higher sensitivity levels. This could further enable the application of this method to the analysis of steroids for clinical research.
720004692, May 2013