
For research use only. Not for use in diagnostic procedures.
In today’s world of regulated and non-regulated bioanalysis, one of the biggest challenges is addressing upcoming analytical demands, such as the ability to perform LC-MS analysis of large molecules (peptide, protein, oligonucleotides, etc.) with accuracy, reproducibility, and desired sensitivity. This application note demonstrates the benefits of Waters Regulated Bioanalysis System Solution for quantification of large molecules, such as peptides. Oasis WCX Ion-Exchange Micro-Elution Plates, the ACQUITY UPLC System, and the Xevo TQ-S Mass Spectrometer were used in this study for the development of an LC-MS method to analyze pg/mL concentration of goserelin in plasma.
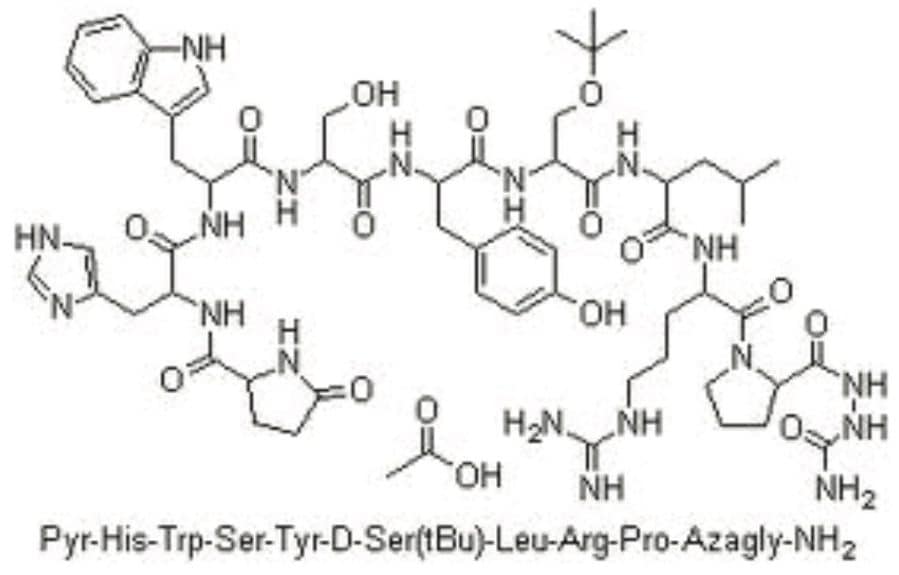
Goserelin is a synthetic hormone analogue.1 Structurally it is a decapeptide, as shown in Figure 1, which is used to suppress the production of the sex hormones (testosterone and estrogen). Goserelin is particularly used in the treatment of breast and prostate cancer. For identification and quantification of goserelin in plasma, a method has been reported in rabbit plasma2 with an LLOQ of 100 pg/mL with an overall runtime of 10 min. Other methods include radio-immunoassay,3 capillary zone electrophoresis (CZE)-UV/MS,4 multiple-injection CZE (MICZE),5 CE hydrogen deuterium exchange-MS (CE-H/D-MS),6 and fast atom bombardment-MS (FAB-MS)7 for crude peptide mixtures only. However, none of these methods address the challenge of analyzing and quantifying goserelin at LLOQ concentrations. In this application note, we report an LC-MS method to determine and quantify goserelin at an LLOQ of 2.5 pg/mL with an overall runtime of 3.5 min in plasma. These results demonstrate the capability of Waters ACQUITY UPLC, Xevo TQ-S, sample preparation and column chemistries to address several bioanalytical challenges, such as achieving the desired sensitivity, addressing upcoming analytical challenges, and regulatory concerns while maintaining high throughput and desired robustness.
Samples were extracted using solid phase extraction (SPE) employing Oasis WCX Micro-Elution 96-Well Plate. An aliquot of plasma was diluted with acidic water, and loaded onto the plate previously conditioned with organic solvent and water. The plasma solution was then washed with water in basic conditions followed by an organic wash, and then eluted using acidified organic elution solvent. The eluted samples were then mixed with Milli-Q water, and injected on to the system. Triptorelin, a decapeptide (M.W. 1311.5), which is a gonadotropin-releasing hormone agonist, was used as an internal standard (IS).
Goserelin and triptorelin (IS) eluted at 1.43 min with a peak width of about 5 s, as shown in Figure 2. The data shown below illustrates the blank signal, shown in Figure 3. The signal obtained from the lower limit of quantification (LLOQ) of goserelin in human plasma is also, shown in Figure 3.
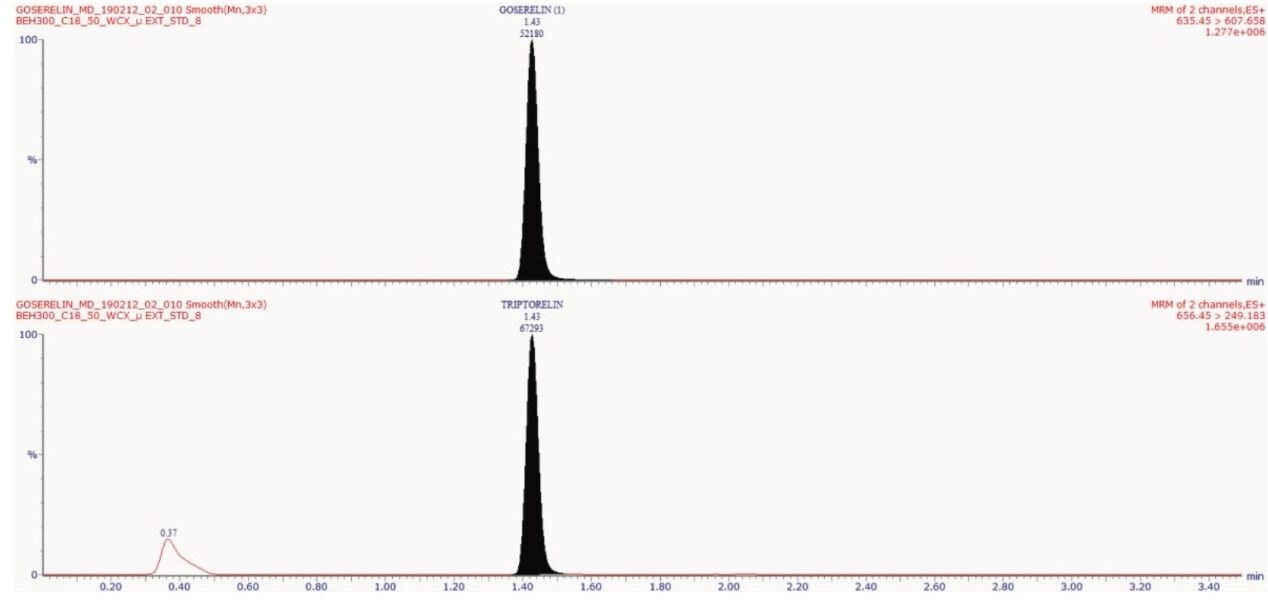
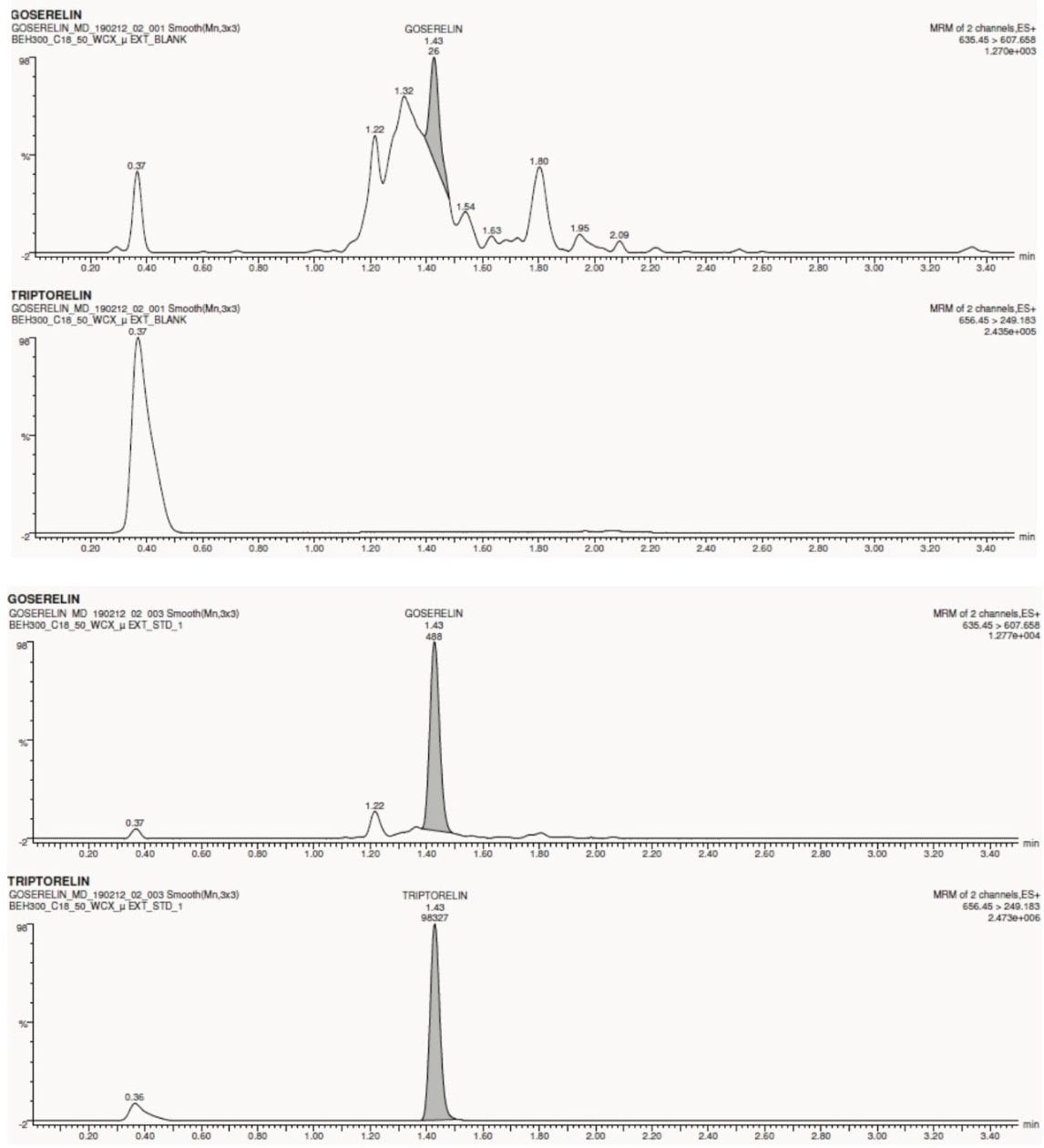
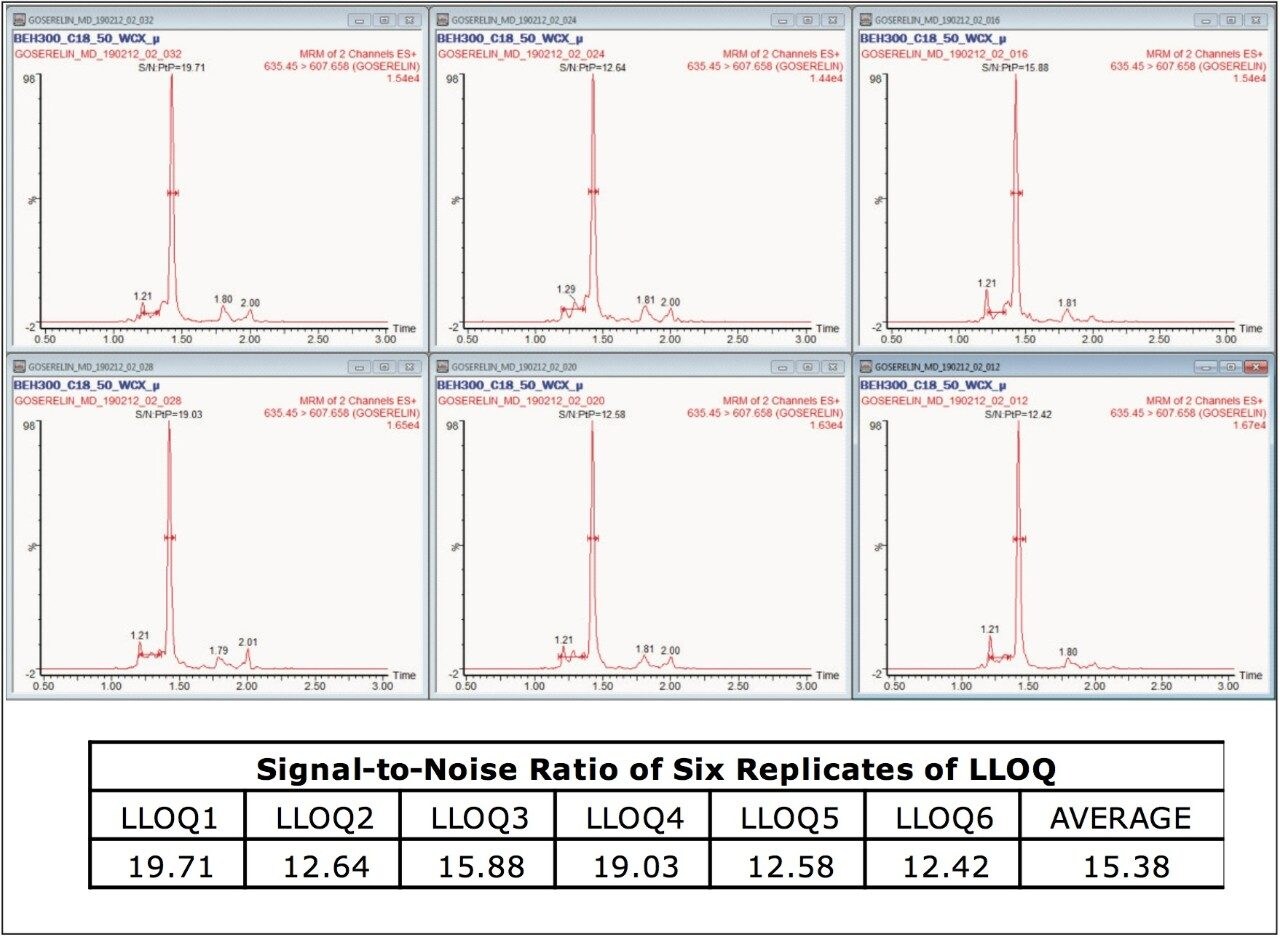
No significant interference(s) were observed in the retention time (1.43 min) where goserelin elutes, shown in Figure 3. In addition, the signal-to-noise (S/N) ratio of ~16:1 was observed at the LLOQ concentration of 2.5 pg/mL for an average of six replicates of LLOQ samples, as shown in Figure 4.
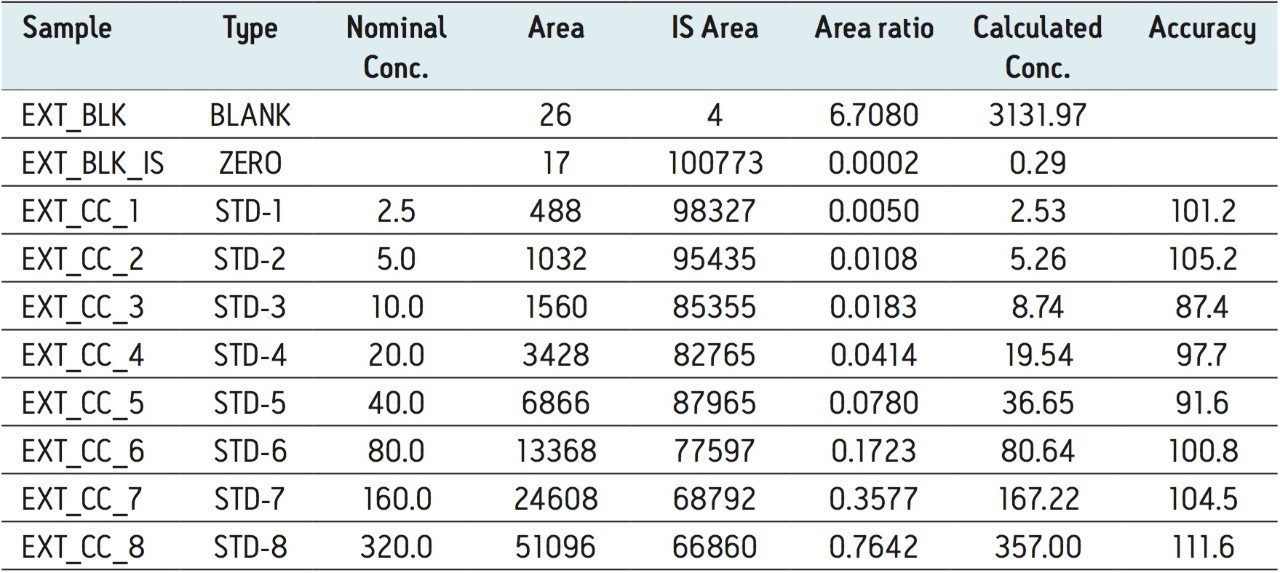
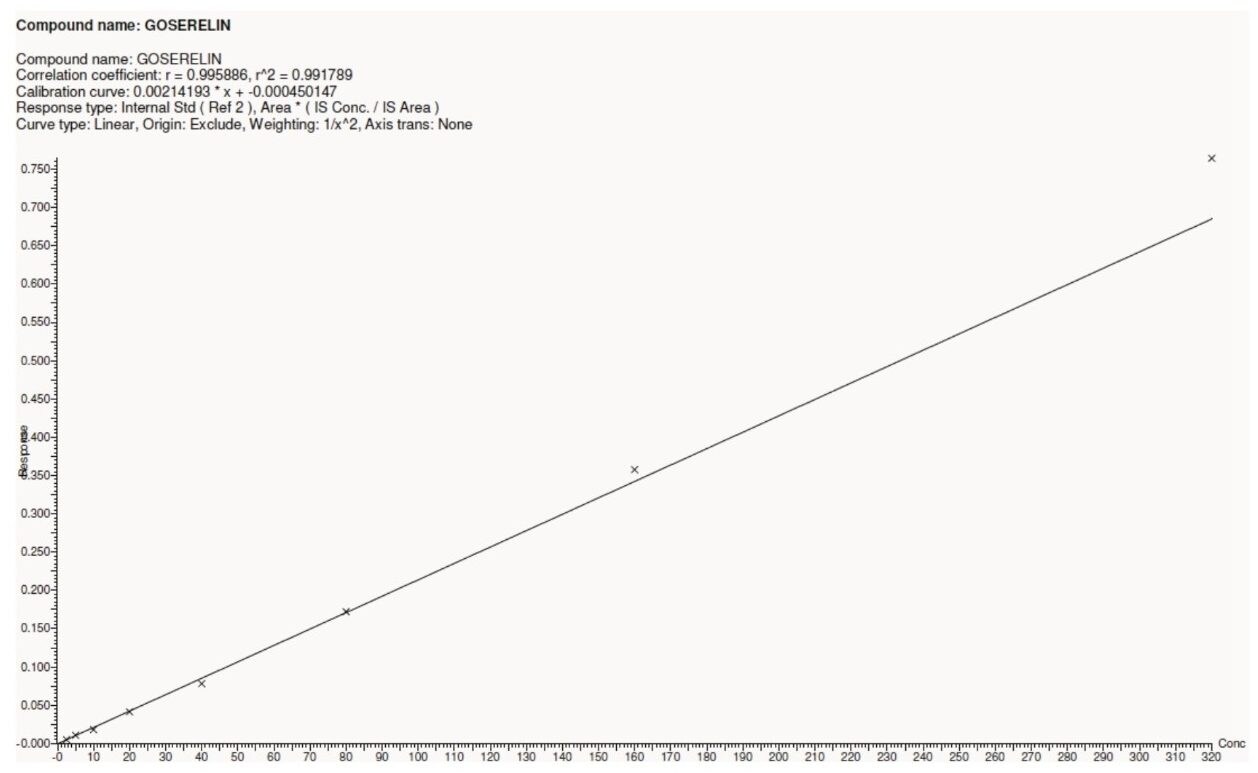
The assay in this report showed linear calibration over the range of 2.5 to 320.0 pg/mL with an excellent r2 value of 0.99, shown in Table 1 and Figure 5. This assay was performed with a 3.5 min injection-to-injection time scale highlighting the capability of ACQUITY UPLC in delivering fast gradients with desired sensitivity, high throughput value, and precision.
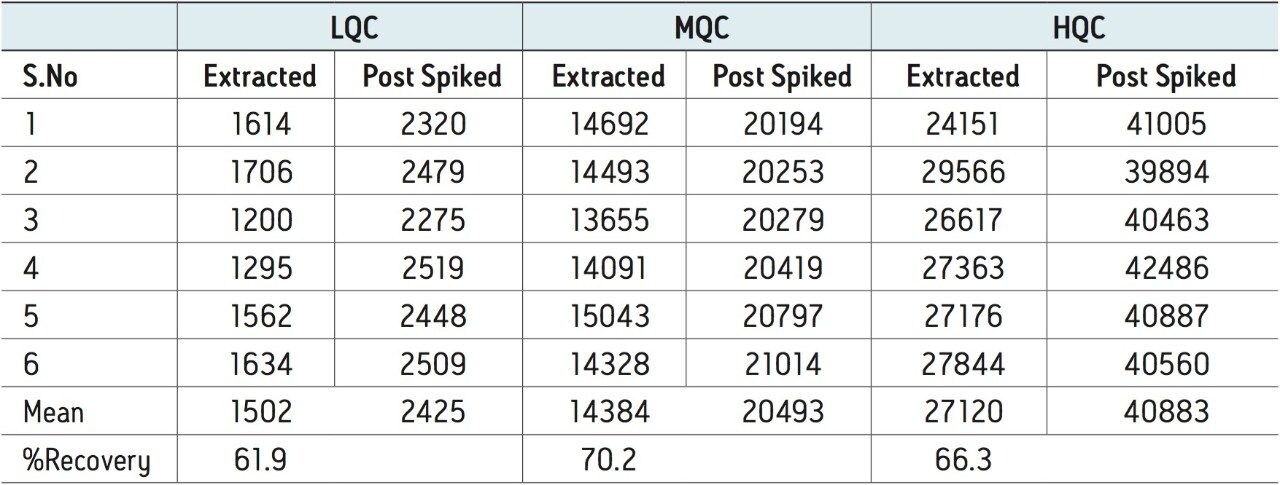
Recovery of the analyte and internal standard (IS) was calculated by comparing the extracted QC samples against six post-extracted samples and was found to be approximately 66% for the analyte, shown in Tables 2 and 3.

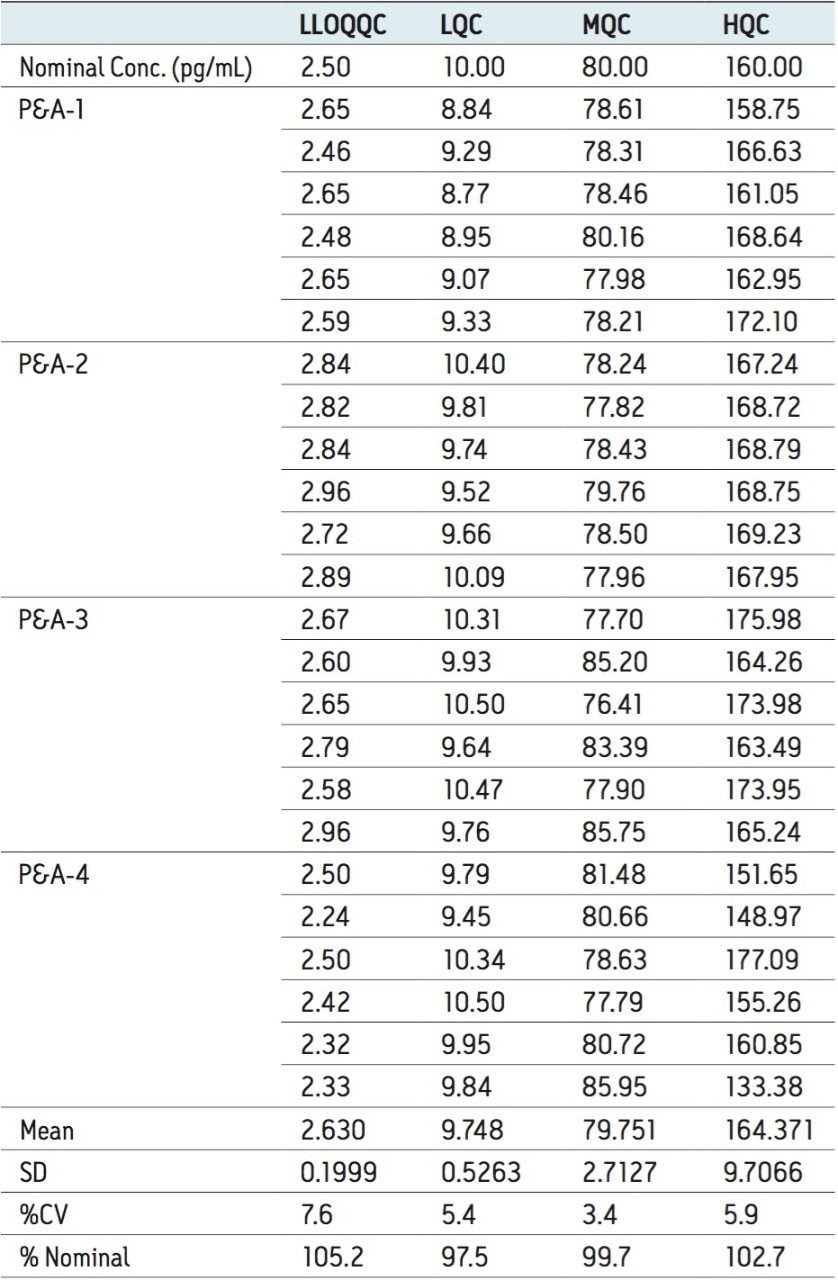
For a comparison of samples within the global batches, four separate batches were analyzed with six samples in each batch at the LLOQQC, LQC, MQC, and HQC concentration levels. The data showed excellent agreement between the six samples in all the four batches, as shown in Table 4. The mean accuracy obtained for all the sample levels was found to be >97% for every concentration, as shown in Table 4. The %CV for repeat batches was found to be within 8% of LLOQQC and varied between 3% and 6% for all QC levels. This variability is well within the acceptable limits in the regulated bioanalysis world.
Goserelin is a synthetic hormone analogue and it is specifically used in the treatment of breast and prostate cancer. An LC-MS method to determine and quantify goserelin in low concentrations in plasma has not previously been developed. In this application note, we report a rapid, robust, and sensitive method that was developed for the determination of decapeptide, goserelin from human plasma by using the ACQUITY UPLC System and Xevo TQ-S Mass Spectrometer. Goserelin and triptorelin (IS) were well separated within 3.5 min having good peak shapes, under the optimized conditions of UPLC and MS/MS. Finally, the established method is suitable for the determination of goserelin from human plasma. This application note successfully addresses the bioanalytical challenge of upcoming analytical demands while addressing several other challenges such as sensitivity, robustness, and regulatory concerns.
720004409, June 2012