
This application note demonstrates the use of charged surface hybrid (CSH) column technology which greatly improves loadability of alkaloids found in Goldenseal extract, when compared to a high purity, fully-porous C18 and a superficially porous C18 column. Improved sample loading for basic compounds such as isoquinoline alkaloids in Goldenseal extract were observed using CSH technology.
Goldenseal (Hydrastis canadensis) is a plant native to Northeastern US and Canada that has long been used as a traditional medicine across many cultures. Goldenseal root extract contains several isoquinoline alkaloids including berberine and hydrastine, which have been studied extensively for their medicinal properties.1 These alkaloids typically show poor peak shape on traditional reversed-phase columns in low-ionic strength mobile phases due to their basic nature and limited sample loadability.
This application note demonstrates the use of charged surface hybrid (CSH) column technology which greatly improves loadability of alkaloids found in Goldenseal extract, when compared to a high purity, fully-porous C18 and a superficially porous C18 column. By improving sample loadability, the peak shape, resolution and sensitivity of detection of the alkaloids by UV is enhanced, facilitating identification of these components using a time-of-flight mass spectrometer (ToF-MS). The Goldenseal separation is then seamlessly transferred from UPLC to preparative HPLC using scalable CSH chemistries, alleviating the need to fully re-develop a preparative HPLC method for target compound purification. Using the same CSH stationary phase on the preparative scale, the selectivity of the Goldenseal separation and improved peak shape of the alkaloids is maintained.
|
Column: |
ACQUITY UPLC CSH C18 2.1 x 100 mm, 1.7 μm, part number: 186005297 |
|
Flow Rate: |
0.4 mL/min |
|
Inj. Volume: |
1 μL |
|
Gradient: |
0 to 40% B in 6 minutes, hold for 2 minutes, re-equilibrate at 0% B to 10 min. |
|
All instruments (unless otherwise noted) |
|
|
Mobile Phase: |
A: 0.1% formic acid in water B: 0.1% formic acid in acetonitrile |
|
Needle Wash: |
10:90 water:acetonitrile |
|
Sample Purge: |
90:10 water:acetonitrile |
|
Seal Wash: |
90:10 water:acetonitrile |
|
Column Temp.: |
30 °C |
|
Detection: |
UV at 254nm |
|
Column: |
XSelect CSH C18 OBD 19 x 100 mm, 5 μm, part number 186005421 |
|
Flow Rate: |
17 mL/min |
|
Inj. Volume: |
164 μL |
|
Gradient: |
0 to 40% B in 11.6 minutes, hold to 15.40 minutes, re-equilibrate to 0% B |
|
Mass Scan Range: |
150-500 amu |
|
Mode: |
ESI, V+ mode |
|
Capillary Voltage: |
3000 V |
|
Cone Voltage: |
10 V |
|
Desolvation Temp.: |
350 L/hr |
|
Desolvation Gas: |
700 L/hr |
|
Reference Standard: |
200 pg/mL leucine enkephalin in 50:50 water:acetonitrile |
|
Data Management: |
Empower 2 CDS (UPLC and HPLC) MassLynx (UPLC-ToF-MS and Preparative HPLC) |
|
Goldenseal Extract: |
1605 mg lyophilized goldenseal root powder was extracted at room temperature in 10 mL of ethyl acetate. The sample was stirred for one hour and filtered through a 0.2 μm nylon filter. |
Major secondary metabolites of Goldenseal include isoquinoline alkaloids such as berberine and hydrastine. These basic compounds typically have poor peak shape and limited loading capacity on reversed-phase columns using low ionic strength mobile phases.2 When analyzing complex mixtures, poor peak tailing and resolution due to overloading can easily mask minor components that may be of interest. Charged Surface Hybrid (CSH) column technology applies a low-level charge to the surface of a hybrid particle, which provides superior peak shape and loading of basic compounds in mobile phases containing additives such as formic acid.
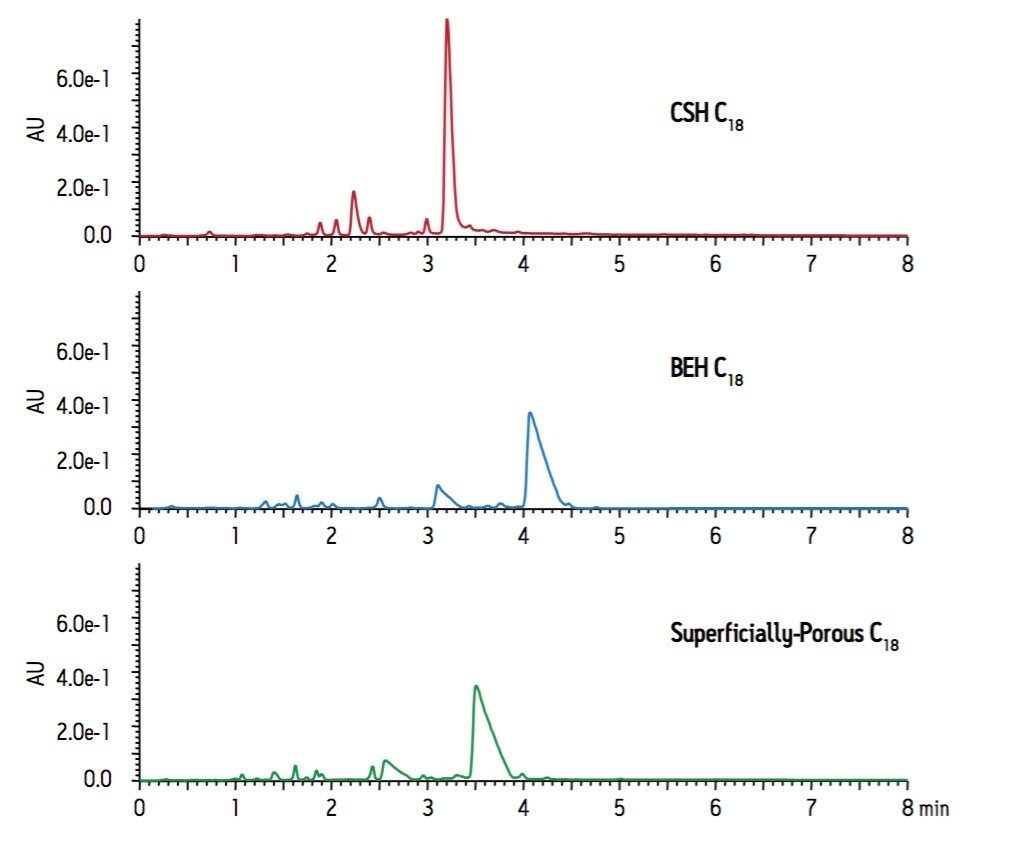
The Goldenseal extract separation on a CSH C18 column was compared to a BEH C18 column as well as a superficially-porous C18 column on an ACQUITY UPLC system. The resulting chromatograms are shown in Figure 1. Both the BEH C18 column and the superficially-porous C18 column display peak shape that is indicative of mass overloading for the two major peaks. By comparison, the CSH C18 column shows much sharper peak shape and increased sensitivity by UV for metabolites in the Goldenseal extract.
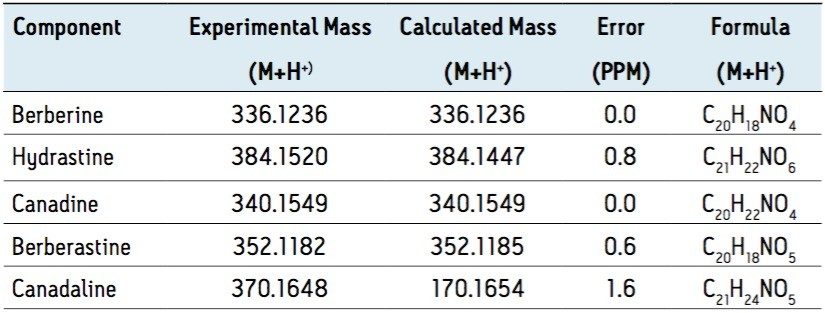
In complex separations, peak tailing and poor resolution can interfere with the identification of components of interest. Using CSH technology in combination with UPLC-ToF MS, a rapid one-step identification of target compounds from complex mixtures can be performed. In this case, Goldenseal extract was analyzed using the CSH C18 column on an ACQUITY UPLC coupled to an LCT Premier ToF mass spectrometer to identify secondary metabolites. The masses of unknown major and minor peaks in the total ion chromatogram were evaluated and compared to known secondary metabolites of Goldenseal. The identity of each component was confirmed by accurate mass analysis (Figure 2) using the Elemental Composition Calculator in MassLynx software, which provides a formula that best fits the experimental mass based on isotope fit and mass error, compared to the theoretical calculated mass.
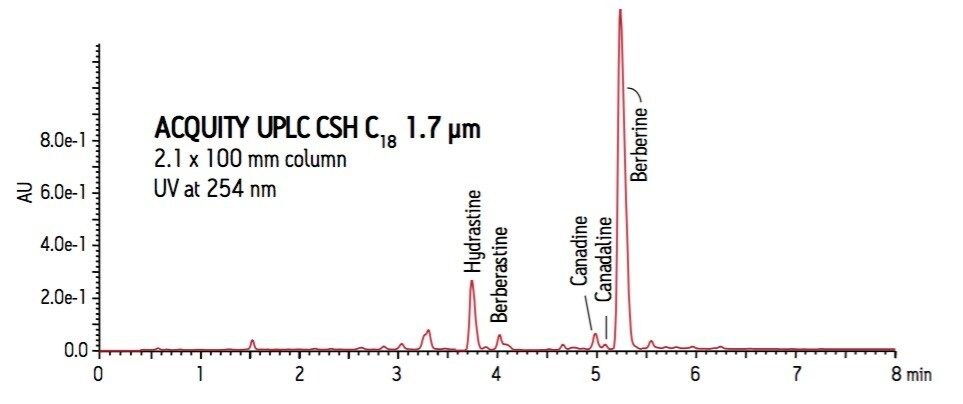
Based on the LC-MS analysis, the two main peaks in the UV chromatogram were identified as hydrastine and berberine (Figure 3). The improved loadability and peak shape using CSH technology allowed for closely eluting minor components canadine, canadaline and berberastine to be resolved and identified.
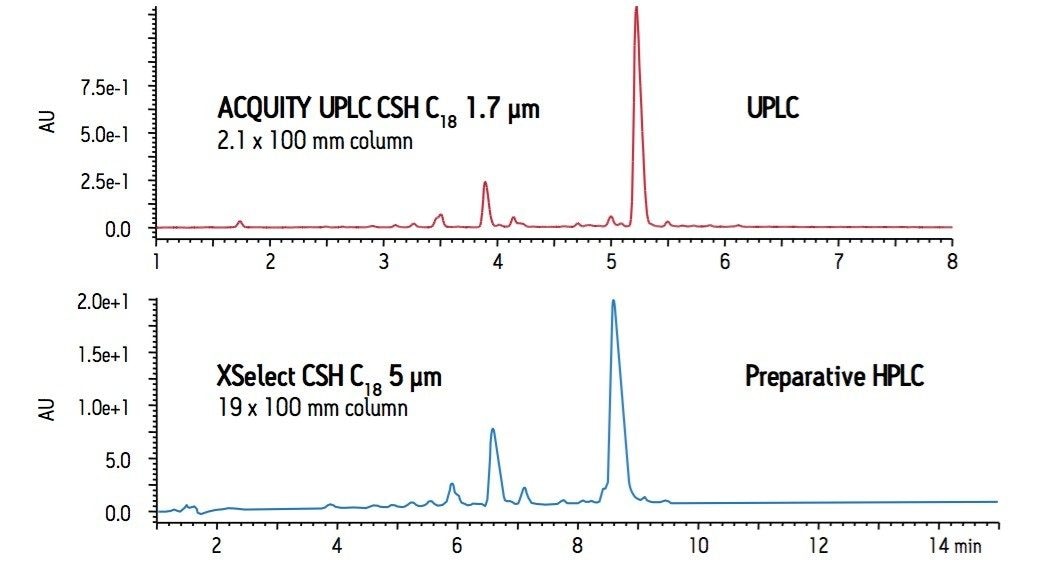
Analytical methods (i.e., UPLC or HPLC) are often used in screening natural product extracts for target compounds. Method with compounds of interest are often further scaled to preparative HPLC for isolation of desired components. The use of different column stationary phases for the analytical and preparative separations can affect selectivity and peak identification, necessitating time-consuming re-development of the method on the preparative scale. The availability of CSH stationary phases in analytical and preparative sizes allows for seamless scaling of separations, maintaining peak elution order. In addition, the increased sample loading capacity of CSH columns compared to BEH or superficially-porous columns is highly advantageous for preparative-scale separations, where improved peak tailing can result in cleaner fractions and fewer iterative purification steps.
The Goldenseal separation was transferred from the ACQUITY UPLC CSH C18 column on a UPLC system to the equivalent chemistry (XSelect CSH C18 19 x 100 mm, 5 μm) on a preparative HPLC system. The method was first transferred to HPLC using the ACQUITY UPLC Columns Calculator3 and subsequently scaled to preparative HPLC using the Waters Preparative Columns Calculator. Properly scaling the 10-minute UPLC method to HPLC, while maintaining resolution by keeping the column length to particle size (L/dp) ratio constant, would require a 4.6 x 250 mm 5 μm column and result in a method that is approximately 75-minutes long. Rather than run this long HPLC method, the calculated HPLC method was transferred directly to preparative HPLC, disregarding particle size and scaling for constant linear velocity. Scaling for constant linear velocity by adjusting flow rate, injection volume and gradient time resulted in a very short preparative run time, sacrificing only a small amount of resolution. Using the same column chemistry from analytical to preparative scale, the increased loading capacity, improved peak shape and separation using CSH technology that was observed by UPLC is maintained on the preparative HPLC system (Figure 4).
720004098, September 2011