To provide a complete system solution for determining low parts per trillion levels (ng l-1) of EPA method 5211 stated N-nitrosamines in drinking water.
The rapid and accurate analysis of drinking water is essential in protecting human health and well-being. The assurance of clean, safe drinking water has become more critical given the potential of accidental or intentional contamination, which has increased in recent years.
The detection of potentially carcinogenic N-nitrosamines in public water supplies has raised regulatory concerns and led to the requirement for surveillance of these unregulated contaminants. One source of concern is the probability of carcinogenicity of these compounds at low parts per trillion (ng l-1) concentrations.2
Water companies currently monitoring their supply have an opportunity to take the necessary steps to ensure the delivery of high quality water to their customers and prepare for the possibility of regulatory limits being applied. This will also help to demonstrate the supplier’s commitment to public health.
N-nitrosamines are thought to occur in drinking water as a by-product of water treatment process operations.3 They take the generic structure as shown in Figure 1 with variants arising from the substitution of the two R groups as shown in Table 1.
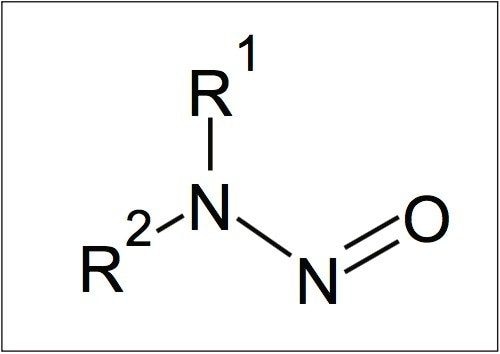
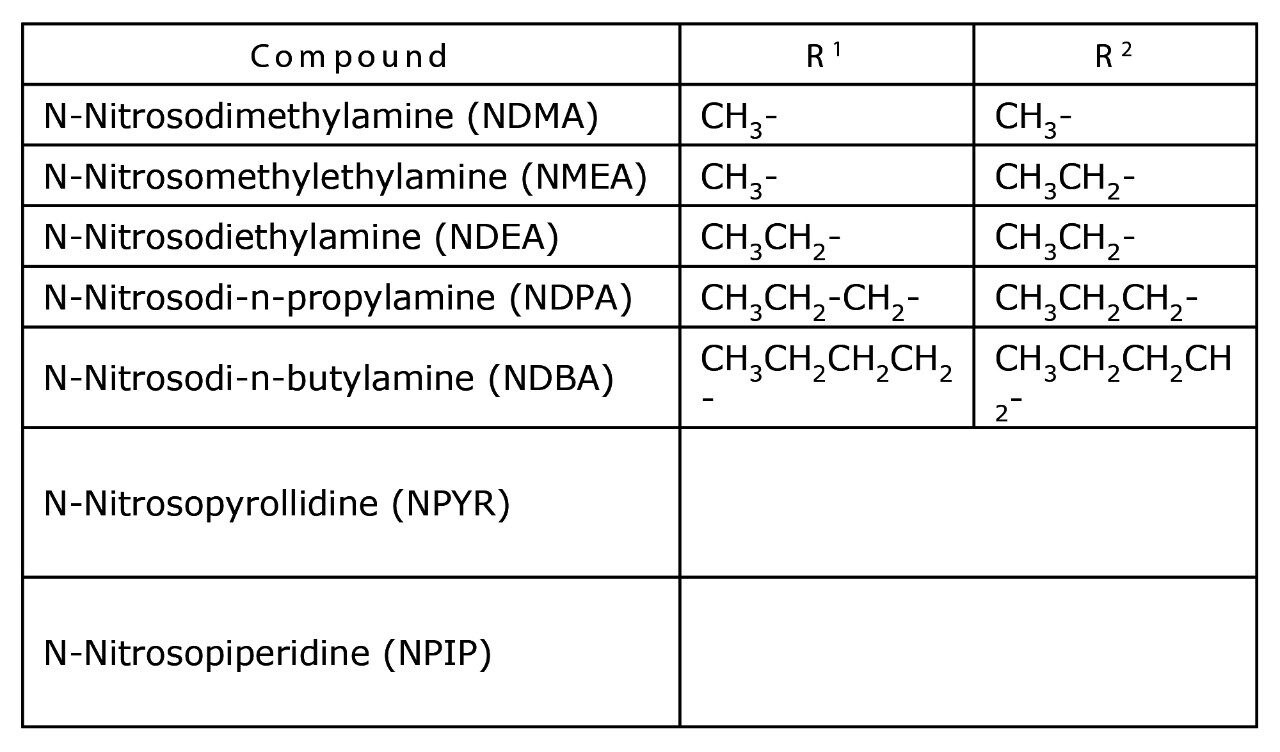
When relatively low molecular weight groups are present for R1 and R2, N-nitrosamines are volatile and hydrophilic compounds. This adds difficulty to the analytical measurement in aqueous matrices. Addressing this challenge requires the use of an integrated approach of specialist chemistries and innovative instrumental technology.
The following method describes a complete system solution that allows the determination of N-nitrosamines in drinking water at low ng l-1 concentrations.

Tap water samples were dechlorinated, fortified with varying levels of N-nitrosamines, spiked with 100 μl d14-NPDA (10 ng ml-1 ) and extracted using a Waters extraction manifold under vacuum in conjunction with Sep-Pak AC2 cartridges. Sep-Pak AC2 is a highly hydrophobic, low ash content, activated carbon used to remove or enrich highly polar organic molecules from water. Prior to extract elution, Sep-Pak AC2 cartridges were fitted directly to pre-conditioned Waters Sep-Pak Dry cartridges. Sep-Pak Dry is a high capacity desiccant used to remove residual water from water immiscible solvents. The procedure employed was a variation of EPA 5211 methodology.
|
Cartridge: |
Sep-Pak AC2, (Waters part no. JJAN20229) |
|
Condition: |
6 ml dichloromethane 6 ml methanol 10 ml H2O (analyte free) |
|
Load: |
500 ml sample (<10 ml min-1) |
|
Dry: |
Under vacuum (35 mins) |
|
Condition: |
Sep-Pak Dry cartridges (Waters part no. WAT054265) 4 ml dichloromethane |
|
Elute: |
Connect Sep-Pak AC2 Cartridges directly to conditioned Sep-Pak Dry cartridges Elute 3 x 2 ml dichloromethane |
|
Concentrate: |
To 500 μl under N2 at 20 °C |
|
GC conditions |
|
|
Instrument: |
Quattro micro GC |
|
GC column: |
BPX-VOL 30 m x 250 μm x 1.4 μm df |
|
Injector : |
PTV operated in solvent vent mode |
|
Liner: |
Tenax TA packed |
|
Injector temp: |
20 °C (0.65 min), 200 °C min-1 to 300 °C (3 min), 300 °C min-1 to 325 °C (5 min) |
|
Vent pressure: |
0 KPa |
|
Vent flow : |
70 ml min-1 |
|
Vent end : |
0.65 min |
|
Purge time: |
4.05 min |
|
Injection: |
20 μl @ 0.5 μl s-1 |
|
Oven temp: |
35 °C (4 min), 25 °C min-1 to 280 °C, 30 °C min-1 to 300 °C (4 min) |
|
Column flow: |
1.3 ml min-1 constant flow |
|
Ion mode: |
EI+ |
|
Electron energy: |
70 eV |
|
Source temp: |
180 °C |
|
Acquisition: |
Multiple Reaction Monitoring (MRM) |
|
Collision gas: |
Argon (3.0 x 10-3 mBar) |
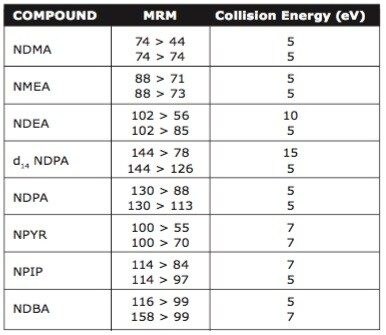
Data was acquired using Waters MassLynx v. 4.1 Software. Data processing was carried out using Waters TargetLynx Application Manager.
The volatile nature of these N-nitrosamines favors measurement by gas chromatography with a sensitive detector such as MS. However, the low molecular weights of these compounds make them difficult to detect against background in real samples when using single stage nominal mass spectrometry. Quattro micro GC allows a greater opportunity to detect low levels in real samples without suffering from matrix saturation problems associated with other MS/MS instruments. This can also be achieved without the need for source reagent gas, which has the added advantage of further improving longer term measurement stability.
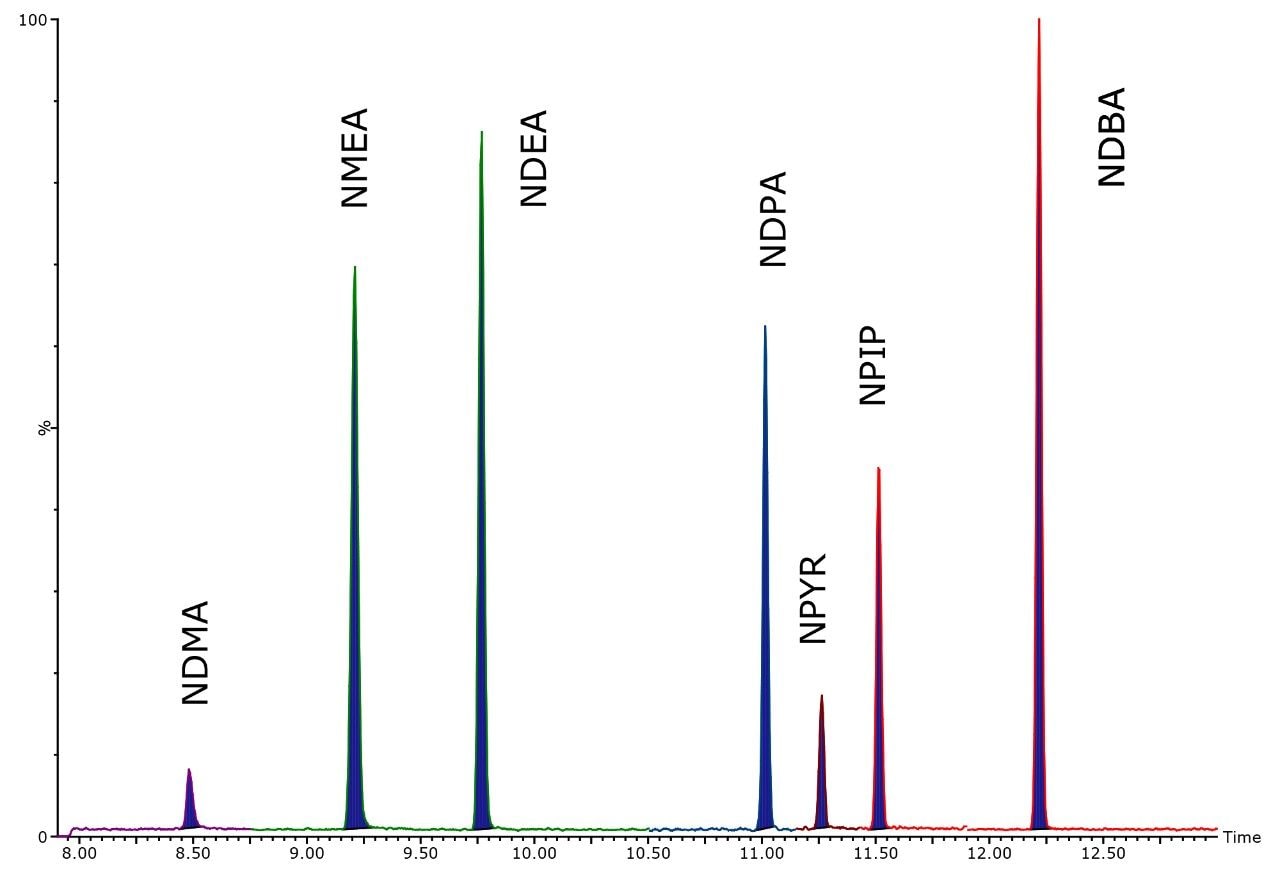
N-nitrosamine analysis requires pre-concentration and large volume injection to achieve the required ppt detection levels. Large volume injection presents its own problems due to the relative vapor pressures of the lower molecular weights species when compared to common GC injection solvents. Use of Tenax TA packed GC inlet liners aids retention of the N-nitrosamines during solvent venting and allows large injection volumes to achieve low detection levels. Figure 2 shows Quattro micro GC MRM chromatogram of N-nitrosamines at 1 ng ml-1 using 20 μl solvent vent injection.
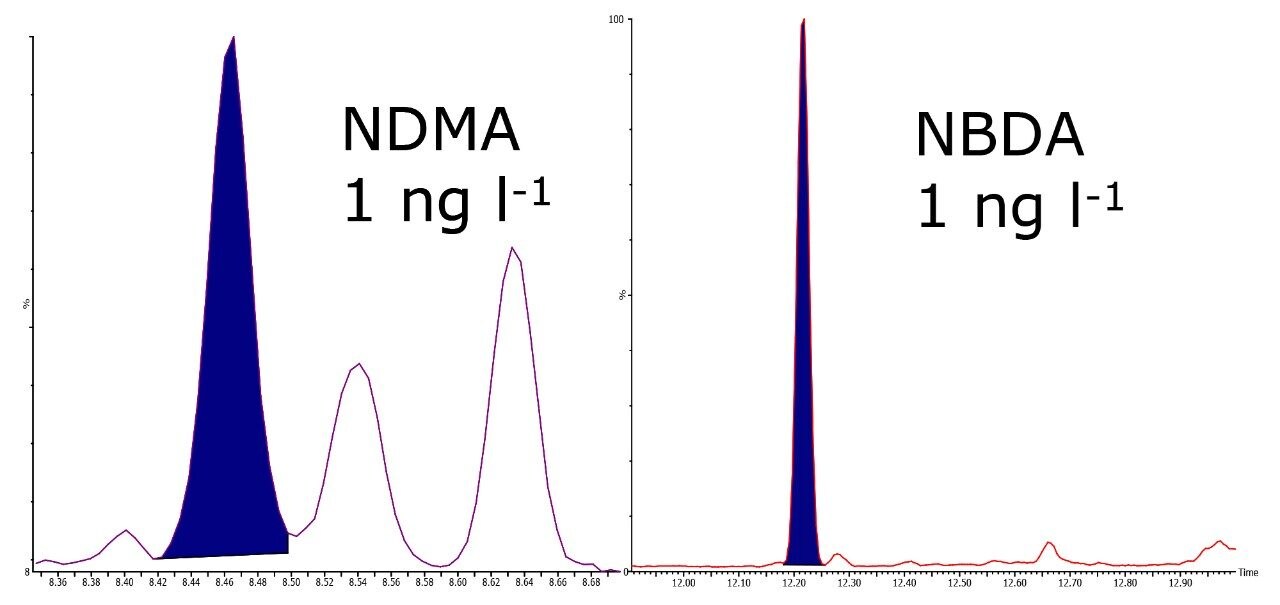
Sample pre-concentration is achievable using solid phase extraction (SPE) but due to the hydrophilicity of N-nitrosamines, it is critical to select the correct chemistry for extraction. When large volume injection is used along with Sep-Pak chemistries and MRM on the Quattro micro GC, it is possible to achieve highly reproducible results at low concentrations with high recoveries. Figure 3 shows NDMA and NDBA in spiked drinking water at 1 ng l-1. Table 3 shows reproducibility and recovery data of N-nitrosamines spiked at 1 ng l-1 in tap water.
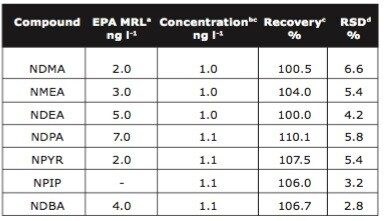
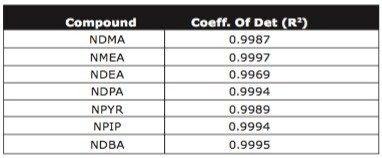
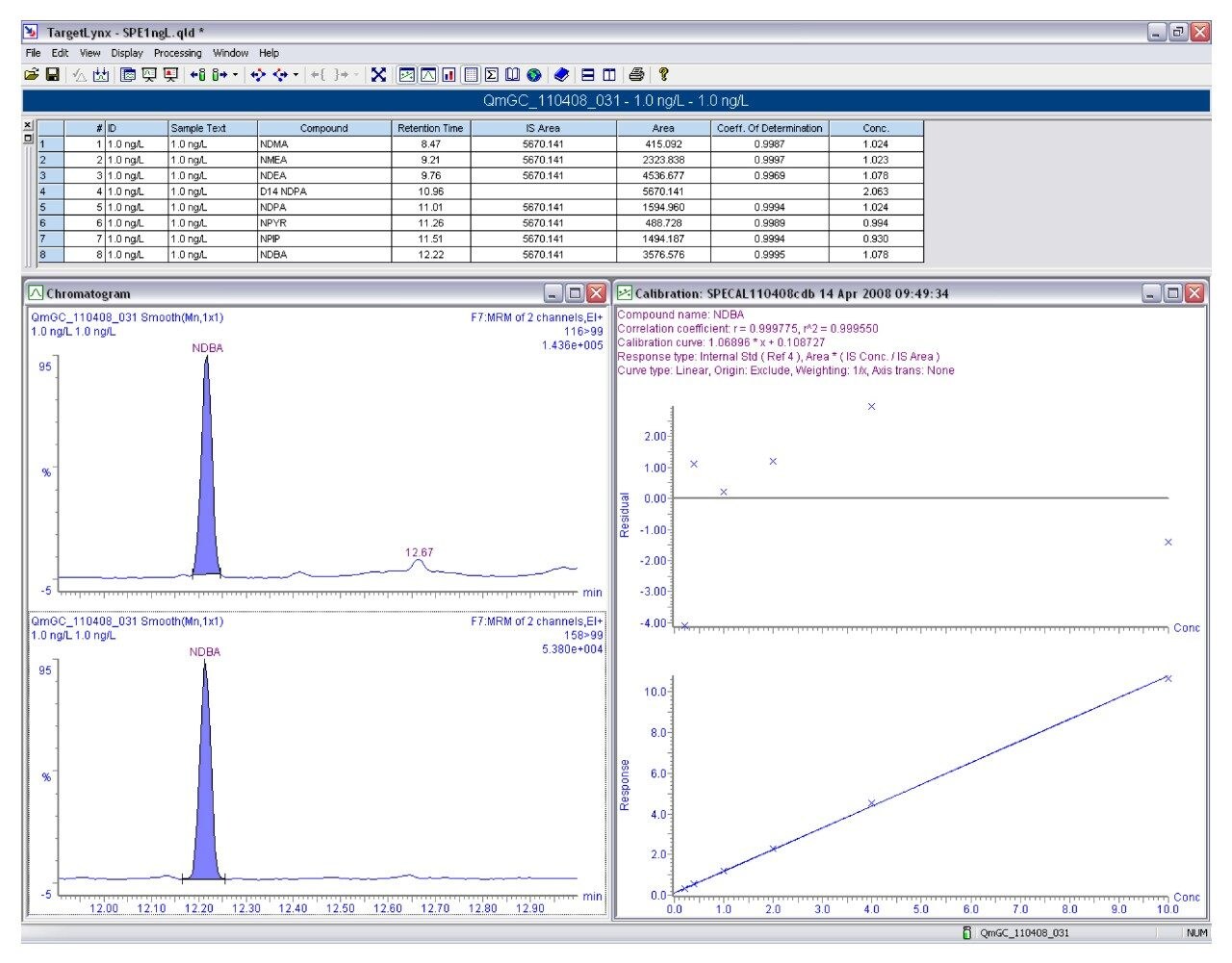
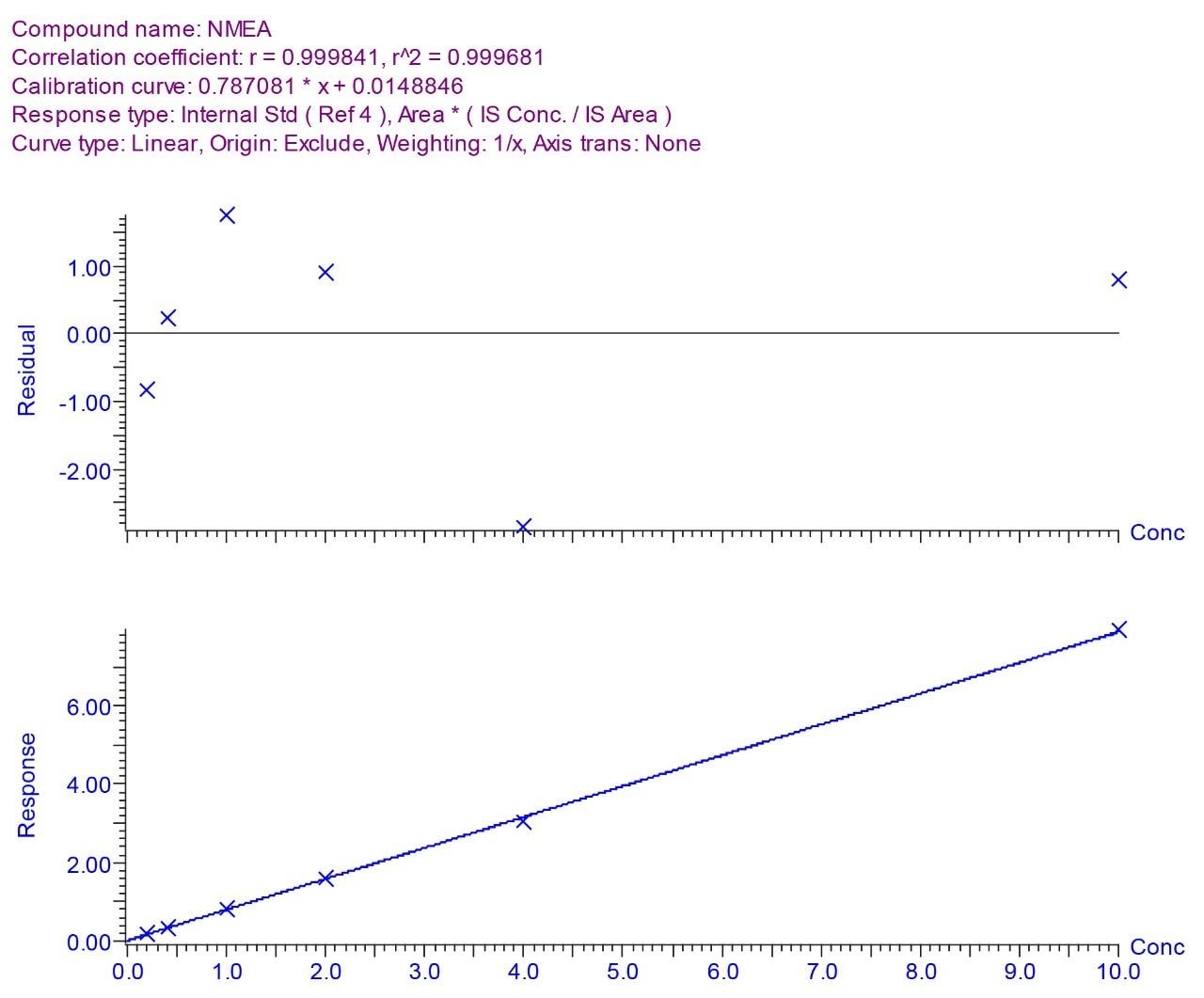
To assess method linearity, calibration curves (0.2 - 10 ng l-1) were prepared by extracting spiked tap water. High recoveries and reproducibility allowed excellent linearity for each N-nitrosamine. Table 4 gives calibration curve coefficient of determination values for each N-nitrosamine spiked. Figure 4 shows calibration curve and residual plot for NMEA (0.2 - 10 ng l-1). Calibration curves were produced using the TargetLynx Application Manager. Figure 5 shows the TargetLynx browser window.
Cristiana Leandro - University of Dundee; for her contribution regarding the initial work on this project.
Mike Young - Waters Corporation; for information regarding SPE.
720002627, May 2008