Application Brief
This is an Application Brief and does not contain a detailed Experimental section.
Emery Domain, Stephan M. Koza, Steve Shiner
Waters Corporation, United States
Published on March 12, 2026
This is an Application Brief and does not contain a detailed Experimental section.
This application brief describes the expected performance of BioResolve Protein A Affinity Columns and the benefits of minimized sample carryover across titer analyses. Lower carryover can enable greater sensitivity through lower detection limits and bolsters confidence in titer measurement accuracy. The data presented herein describe the carryover observed on Waters BioResolve Protein A 3.5 µm Columns, which is demonstrated to be on average 65% lower than the carryover observed on a comparable commercially available column.
Owing to the unique “on/off” retention mechanism harnessed in affinity chromatography, a major pain point of affinity resins is sample carryover across injections. For Protein A (ProA) Affinity Columns, carryover occurs when monoclonal antibodies (mAbs) of interest and/or host cell-related impurities remain bound to the column following an elution cycle. These sample remnants are then released during subsequent elutions, resulting in poor reproducibility and introducing variability during titer quantitation. Alternatively, the small quantity of remaining proteins can begin to aggregate and affect chromatographic performance, sample recovery, and column lifetime.1,2 Carryover can result from several factors, both system and column dependent, though only those related to the ProA resin and column hardware are within the scope of this application brief. These include incomplete elution from the Protein A ligand under suboptimal conditions, non-specific interactions with column hardware or resin packing, and incomplete or ineffective cleaning protocols.1,2,3
Sample carryover can be particularly problematic during bioprocess development, where analysts may require reuse of one ProA Affinity Column to monitor titer output from multiple reactors or for differing mAbs. If the eluate is intended for further downstream analyses, carryover can lead to cross-contamination by not only errant mAbs but also process-specific impurities.4,5 ProA Affinity chromatography is largely considered the first purification step in mAb bioprocessing, and impurities introduced at this step can confound further downstream analyses. To bypass this, common practice is to designate one ProA column to a specific product, however this then results in column use for only a fraction of demonstrated lifetimes.1,4,5 As affinity resins on average cost 50% more than other chromatographic media, the economics of affinity column reuse and the importance of carryover minimization become apparent.4
Column-related carryover can be reduced through method optimization and the implementation of a rigorous clean-in-place (CIP) protocol, but fundamental interactions with the affinity column will pervade. To this end, selecting a ProA Affinity Column with the lowest demonstrable carryover will result in more efficient downstream analysis, extended column longevity, and greater confidence in titer measurement.
Column carryover was assessed using blank elution cycles following a 1 µg NISTmAb injection. The instrument method was constructed to perform three load and elute gradient cycles within one injection, resulting in 2 appended blank elutions with the benefit of bypassing the sample manager and avoiding injection-related artifacts. This ensured that any carryover observed originated only from the ProA column. Samples were loaded in Dulbecco’s PBS and eluted with 100 mM potassium phosphate and 100 mM potassium chloride at pH 3 using a method consisting of a 1 minute load/wash, a 0.1 minute shift to elution, a 1 minute elution hold, a 0.1 minute return to loading buffer, and a 1 minute re‑equilibration before repeating the elution cycle. The 2.1 x 20 mm BioResolve Protein A Affinity Columns and the commercially available benchmark columns were tested at a flow rate of 1 mL/min and the 3.9 x 5 mm BioResolve configuration columns were tested at 2 mL/min. The absolute carryover values reported here are method specific.
BioResolve Protein A Affinity Columns of both configurations were tested for carryover using varied resin batches and multiple lots of High Performance Surface (HPS) Column hardware (Figure 1). Twelve batches of ProA resin were tested in the 2.1 x 20 mm format, as well as 7 columns containing one batch of resin within varying lots of HPS hardware. Eight 3.9 x 5 mm columns were tested as well, 7 containing one resin batch packed in various lots of hardware, and one with a second batch of ProA resin. For comparison, 2 batches of a comparable on-market ProA column were also assessed.
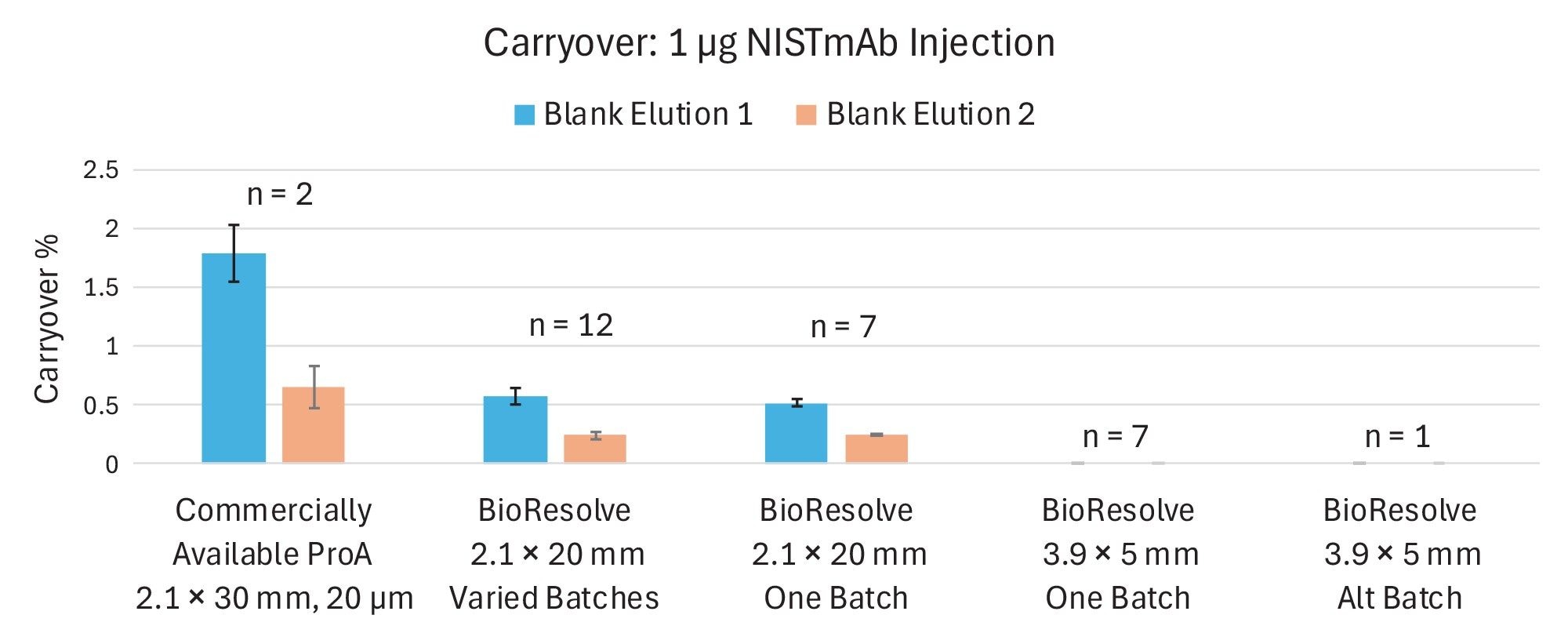
Across the 12 batches of BioResolve ProA resin tested in 2.1 x 20 mm hardware, the average carryover following a 1 µg NISTmAb injection was 0.58% on the first blank elution and 0.25% on the second blank elution. This represents approximately a 65% reduction in observed carryover from the commercially available competitive benchmark in this study, which demonstrated an average carryover of 1.79% and 0.65% during the first and second blank elutions.
Across 7 2.1 x 20 mm columns containing a single batch of BioResolve ProA resin, average carryover was observed to be 0.52% and 0.25% during the first and second blank elutions, consistent with the average carryover reported across the 12 resin batches. When this test sequence was performed on 7 3.9 x 5 mm BioResolve columns also packed with a single batch of ProA resin, no carryover was detected from any column analyzed. To verify this result, another 3.9 x 5 mm column was packed using a secondary batch that had exhibited greater carryover in the 2.1 x 20 mm format, yet this column also yielded no visible carryover peaks. Interestingly, despite the lower linear velocity of the 3.9 × 5 mm column operated at 2 mL/min compared to the 2.1 × 20 mm column at 1 mL/min, carryover was undetectable in this format. While longer residence time might typically be expected to elevate carryover, this was not observed. The geometry of the 3.9 × 5 mm column appears to be inherently well-suited for minimizing carryover.
Carryover is a persistent challenge in Protein A mAb titer analyses, leading to data variability at best and sample contamination at worst. Waters BioResolve Protein A Affinity Columns demonstrate significantly improved performance in minimizing carryover compared to a leading commercial benchmark evaluated in this study. Notably, the 3.9 × 5 mm column format, intended for high-throughput method analyses, further enhances this advantage and exhibits no detectable carryover following a 1 µg NISTmAb injection. This reduction in carryover enhances data integrity, reduces the risk of cross-contamination, and prolongs column lifespan by mitigating sample-induced fouling.
720009259, February 2026