This application note reports on efforts to define ‘universal’ parameters for the BioAccord System applicable to peptide analysis. Glycopeptides are used as a test case representing some of the most labile peptides encountered during typical peptide map analysis.
Optimized MS settings for peptide mapping and monitoring analyses, minimizing in-source fragmentation, and maximizing quantification capacity for labile peptides.
The Waters BioAccord System is a small footprint, SmartMS-enabled LC-MS system designed to assist modern-day biotherapeutic analysis. It leverages technological advancements in liquid chromatography-mass spectrometry instrumentation and informatics to provide workflow-based solutions for routine protein, released glycan, peptide, and oligonucleotide analyses. To enable simplified deployment and operation in labs not traditionally using high-performance LC-MS technology, core functionality includes an automated system startup and calibration, active instrument health checks, and automated workflows for data acquisition, processing, and reporting. This new paradigm for delivering high-performance LC-MS solutions to organizations formerly reliant on MS experts is achieved by significantly minimizing user interventions, resulting in faster turnaround time to high-quality data.
In source fragmentation is a common occurrence with electrospray ionization mass spectrometry instrumentation that generates fragment ions by collisional activation of ionized molecules in the higher-pressure source and post-source regions of the MS inlet. For more labile (susceptible) peptides, this could result in cleavage within peptide backbone amide linkages or side chain modifiers. Modifications such as a glycosylation are particularly susceptible to in-source cleavage of the glycosidic bonds between sugar residues that results in loss of respective peptide signal for the intact glycopeptide. In peptide attribute monitoring studies, the relative abundance of each glycopeptide is assessed based on respective MS signal relative to the total signal for the glycopeptide variants. In-source fragment ions would alter these ratios, reducing accuracy of relative modification levels. The BioAccord System provides users access to a few parameters vital to MS source tuning needed for optimizing performance for various conditions for sample introduction to the MS detector. Optimization of these criteria provides near ideal conditions for peptide analysis, including considerations for minimizing in-source fragmentation. This application note reports on efforts to define ‘universal’ parameters for the BioAccord System applicable to peptide analysis. Glycopeptides are used as a test case representing some of the most labile peptides encountered during typical peptide map analysis.
The mAb digestion standard (p/n: 186009126) was dissolved in 200 μL of LC-MS-grade water containing 0.1% formic acid, to a final concentration of 0.2 μg/μL for MS source optimization experiments. A 5.0-μL injection was used for each LC-MS run, loading 1.0 μg digest on-column. This is recommended loading for optimum sensitivity and chromatographic peak shape.
Trastuzumab (Genentech, CA, USA) and infliximab (Janssen Biotech, Inc, PA, USA) tryptic digestions were prepared by dilution to 1.0 mg/mL in a digestion buffer (0.25 M Tris buffer pH 7.5 and 5.2 M GndHCl). Each sample was reduced and alkylated prior to digestion with trypsin (20/1, w/w) for four hours. The sample was acidified and diluted to a final concentration of 0.2 μg/μL in 0.1% formic acid for LC-MS analysis (1 μg on-column loading).
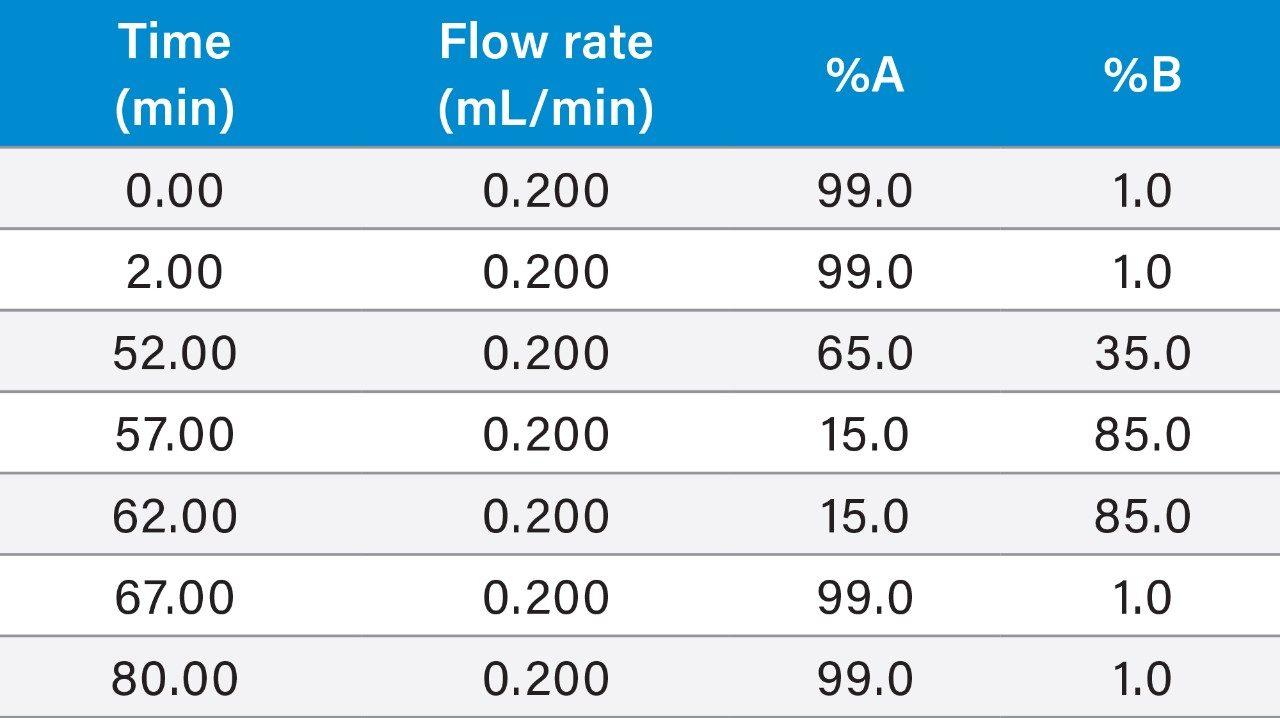
mAb digestion standard: The separation of peptides was performed on an ACQUITY UPLC Peptide CSH C18 Column, 130 Å, 1.7 μm, 2.1 mm, 100 mm (p/n: 186006937) using a 50-min linear gradient (see Table 1), using mobile phase A (0.1% formic acid in water) and B (0.1% formic acid in Acetonitrile).
Trastuzumab and infliximab samples: The separation of peptides was performed on an ACQUITY UPLC Peptide BEH C18 Column, 130 Å, 1.7 μm, 2.1 mm × 100 mm (p/n: 186003555) using the same gradient as above.
LC system: ACQUITY UPLC I-Class PLUS
|
MS system: |
ACQUITY RDa Mass Detector |
|
Ionization mode: |
ESI+ |
|
Acquisition mode: |
Full scan MS with fragmentation (Simultaneous dual-function acquisition at low (fixed) and high (ramped) cone voltage |
|
Acquisition range: |
m/z 50–2000 |
|
Capillary voltage: |
1.2 kV and 1.5 kV |
|
Cone voltage (fixed): |
Test values set at 20 V, 30 V, 40 V, and 50 V |
|
Fragmentation cone voltage: |
60–120 V |
|
Desolvation temp.: |
200–500 °C (test values at 50 °C increment per injection) |
|
Intelligent data capture (IDC): |
on1 |
|
Capillary voltage: |
1.2 kV |
|
Cone voltage: |
20 V |
|
Desolvation temp.: |
350 °C |
UNIFI Scientific Information System under waters_connect with Peptide Map (Exact Mass MS) workflow processing method was used for assignments of peptide identity and a development version of a targeted peptide monitoring software was used for peptide relative quantification.
In-source fragmentation is a known occurrence during electrospray ionization-based mass analysis, resulting in source induced dissociation of thermally labile peptides.2 This phenomenon is occasionally exploited to generate peptide backbone fragment ions for peptide sequence or modification characterization, but they are otherwise viewed as a hinderance as they can negatively affect relative quantitation of labile peptide modifications due to increased complexity of MS spectra and loss of intact peptide variant MS signals.
The ACQUITY RDa Mass Detector source and transfer region are designed to minimize in-source fragmentation of biomolecules. However, quantification of some labile side chain modifications, such as glycosylation, require “softer” MS source conditions to preserve the structure of the glycopeptide moiety.
Optimizing an MS source for low in-source fragmentation has traditionally been challenging for many analytical scientists who are not trained MS experts, largely due to the large number of parameters that could be adjusted. The ACQUITY RDa Mass Detector has been specifically designed such that all parameters have been fixed at nominally optimum values other than the three key source parameters related to sample introduction conditions: Capillary voltage, cone voltage, and desolvation temperature (which are method/sample type specific). These parameters can be accessed through analysis method, as shown in Figure 1.
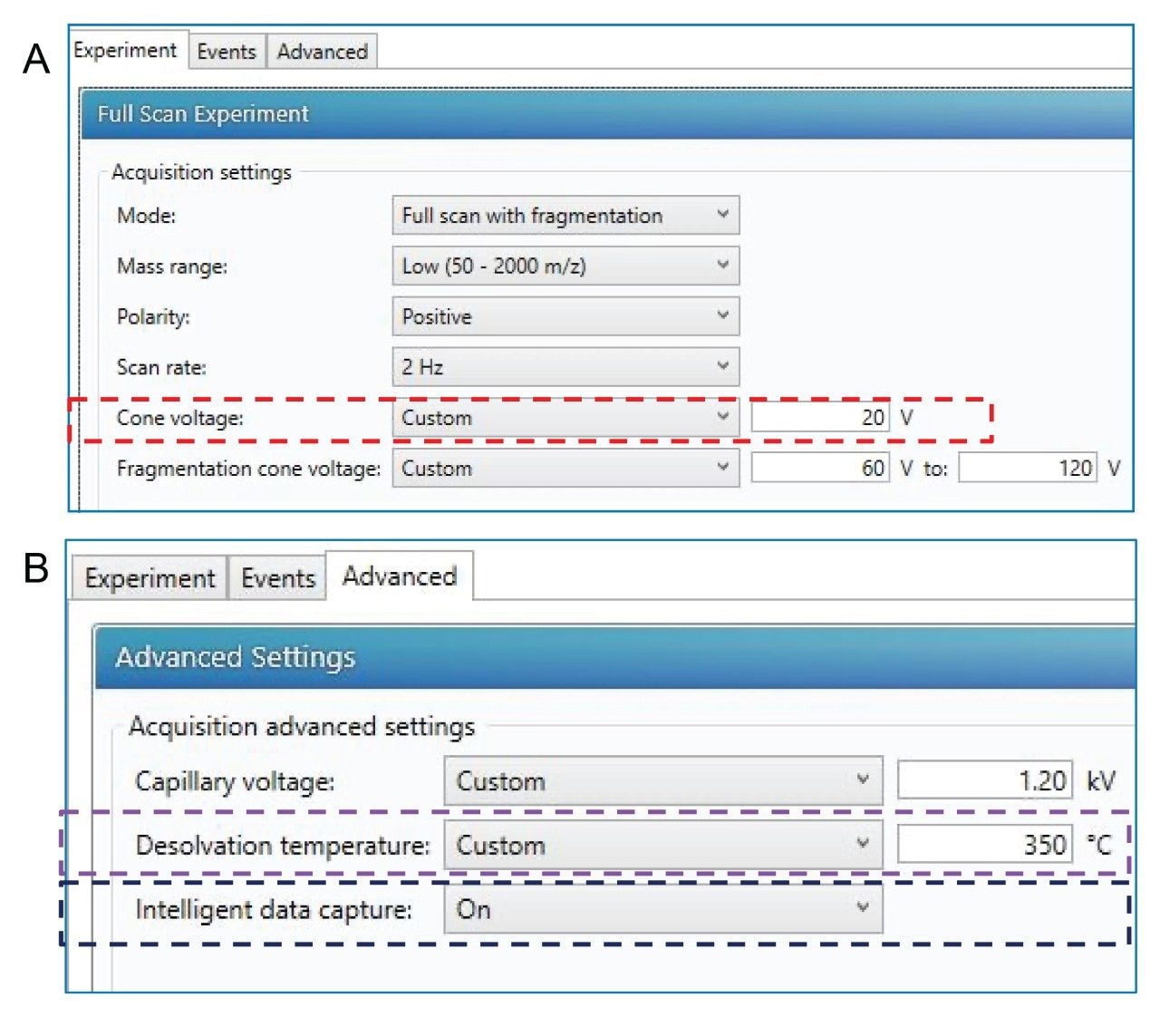
In this study, capillary voltage showed minimal effect on in-source fragmentation levels of glycopeptides (data not shown). The MS response increases with increasing capillary voltage up to 1.2 kV after which little increase is observed up to the maximum of 1.5 kV. A fixed capillary voltage of 1.2 kV was therefore used to evaluate the effects of changing desolvation temperature and cone voltage.
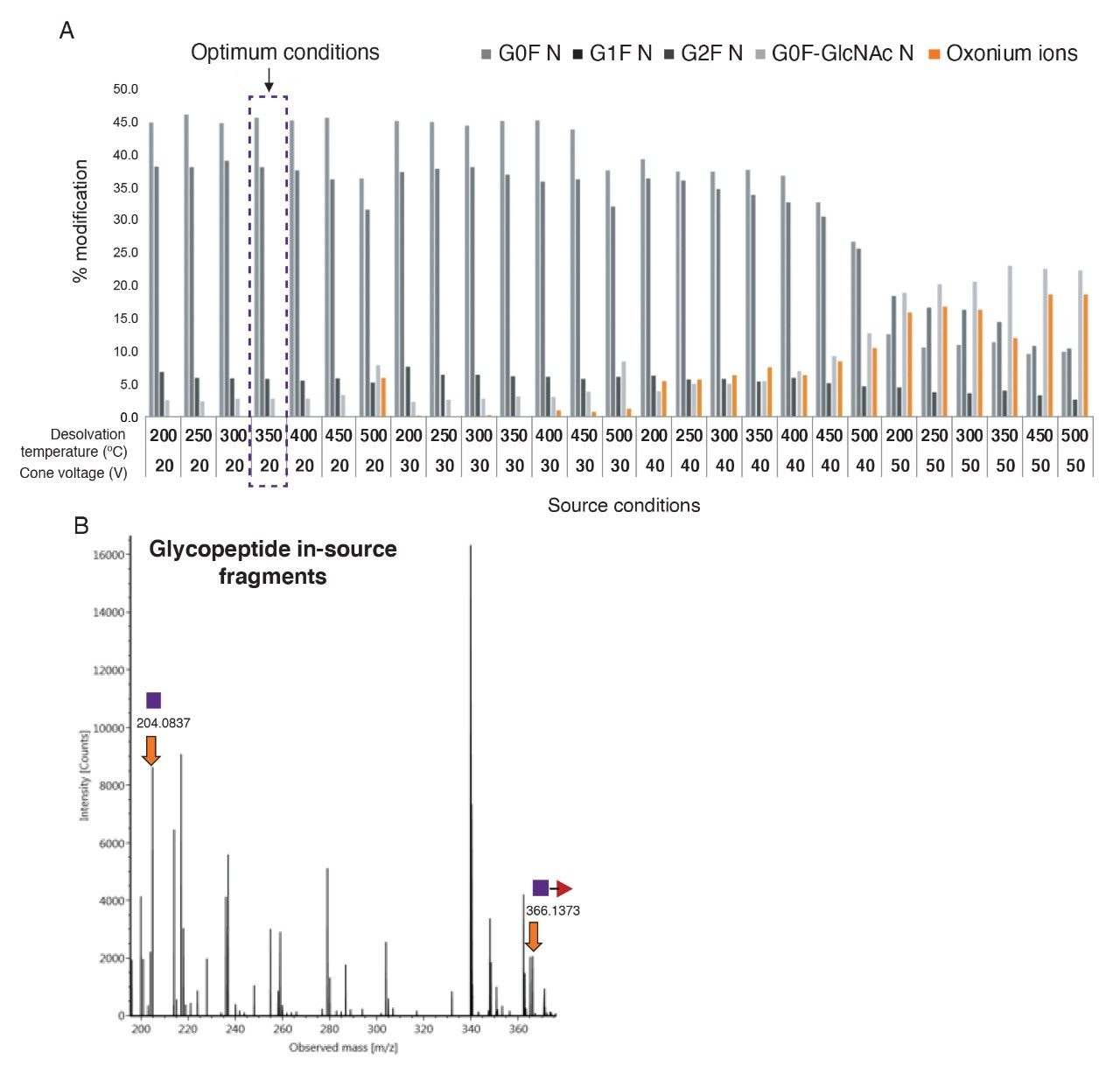
Desolvation temperature and cone voltage were observed to have a greater effect on in-source fragmentation2 and were systematically evaluated to determine the optimum settings for minimization of fragmentation (Figure 2A) of glycopeptides while maintaining absolute MS signal intensity.3
Here, NISTmAb tryptic digest peptides were used as the test sample. To monitor the ion counts, two glycan oxonium ions at m/z 366.1395 Da and 204.0866 Da were selected as markers of in-source fragmentation (Figure 2B). The oxonium ion intensities were monitored at cone voltages of 20, 30, 40, and 50 V over a series of desolvation temperatures from 200–500 °C adjusted in 50 °C increments. The optimum combination of these two parameters was observed at a cone voltage of 20 V and desolvation temperature of 350 °C.
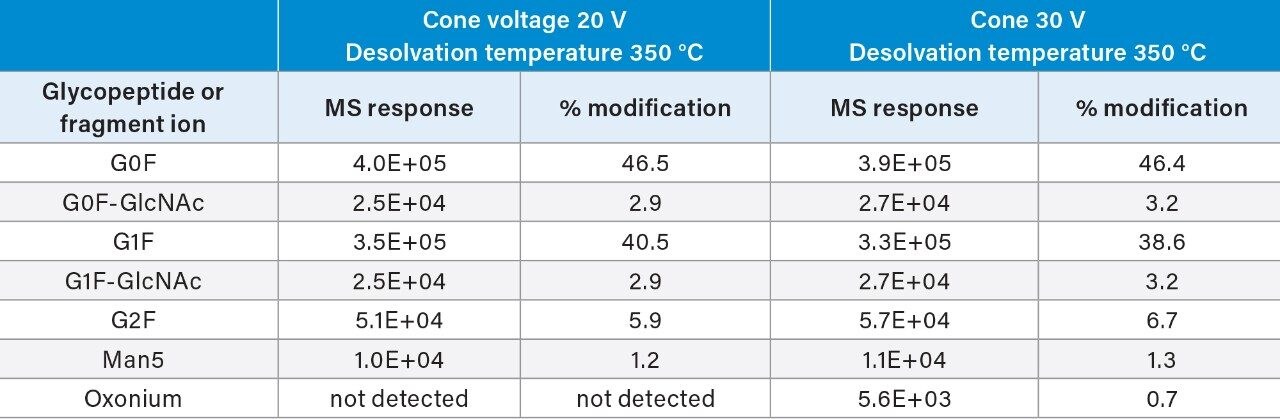
Compared to previously published source parameters for qualitative peptide mapping analysis focused on generating peptide assignments (Waters Application Note, 720006466EN)3 on the BioAccord System (cone voltage at 30 V and desolvation temperature at 350 °C), the newly selected parameters resulted in 0.7% less in-source fragmentation levels for selected glycopeptides. Further, optimized conditions for multiple NISTmAb glycopeptides maintained comparable absolute MS intensity levels and %modification levels for glycopeptides compared to previously reported data (Table 2). These optimized conditions were further verified on two additional BioAccord systems (data not shown).
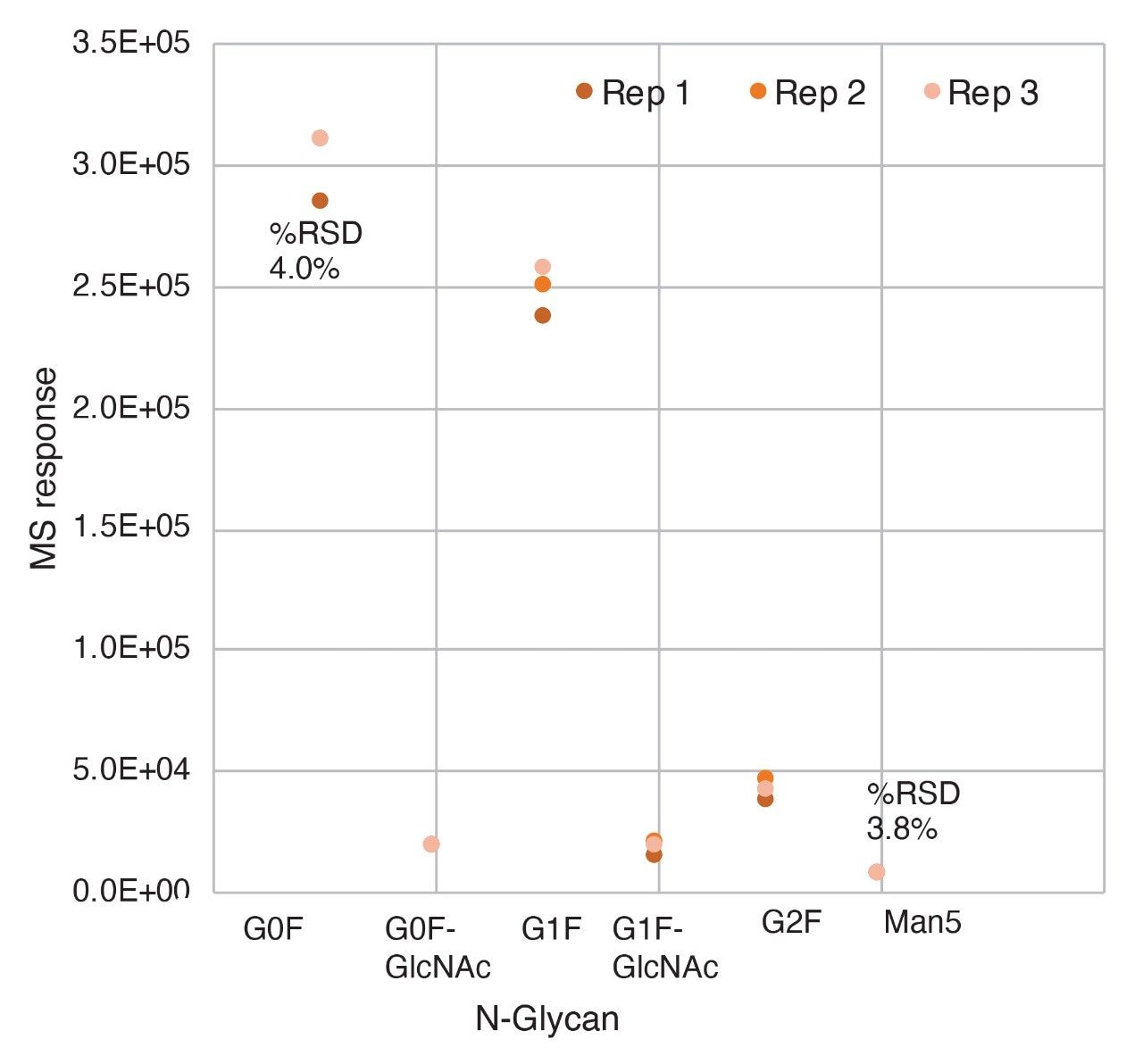
These optimum settings were subsequently used to monitor multiple glycopeptides across three consecutive LC-MS injections. Man5 is a lower abundance glycopeptide with 0.02% abundance relative to base peak peptide signal (VVSAVLTVLHQDWLNGK) in NISTmAb. The replicate injections show high MS sensitivity and repeatability with 3.8% RSD (Figure 3) for the optimized source settings.
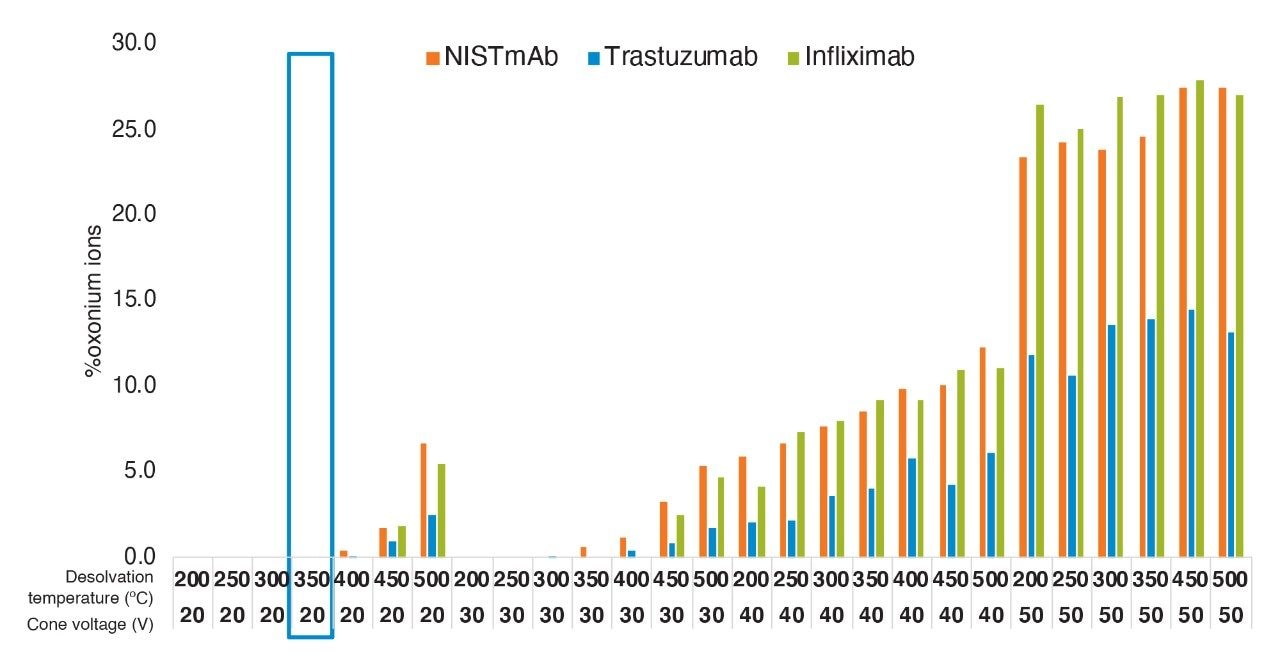
The initial MS source settings optimized using NIST mAb peptides were then confirmed using two additional mAb tryptic digests: trastuzumab and infliximab. In each analysis, the total oxonium ion levels were determined for the same two in-source fragmentation marker ions (m/z 366.1395 Da and 204.0866 Da)
The data showed similar in-source fragmentation levels for all mAb digests (Figure 4). This further confirmed that selected cone voltage and desolvation temperature for mAb digest standard can be applied to other monoclonal antibody molecules when monitoring thermally labile peptides such as glycopeptides.
The integrated analytical workflows of the BioAccord System facilitate automated peptide mapping and product variant attribute monitoring for biotherapeutics. Here, we demonstrated that generic optimized MS source parameters can be used for monitoring labile glycopeptides on the BioAccord System. A capillary voltage of 1.2 kV, cone voltage of 20 V, and desolvation temperature of 350 °C are ideal for minimizing in source fragmentation levels, while maintaining MS sensitivity for low abundance, poorly ionizing glycopeptides such as Man5. Based on relative abundance measurements, the selected conditions enable relative quantification of low abundance labile peptides with highly repeatable %modification levels for glycopeptides across multiple injections. We recommend these source conditions for both qualitative peptide mapping and quantitative peptide attribute monitoring on the BioAccord System to maximize data quality for biotherapeutic analysis.
720006855, April 2020