The goal of the work described herein, was to develop a sensitive and robust method for the LC-MS/MS quantification of NMDA containing the drug product, ranitidine. This application highlights accurate NDMA quantification, achieving LOQs of 0.75 ppb (11 picograms on-column), using the Waters ACQUITY UPLC H-Class PLUS System and XSelect HSS T3 Column for chromatographic separation, and the Xevo TQ-S cronos tandem MS configured with APCI probe for highly sensitive MS detection.
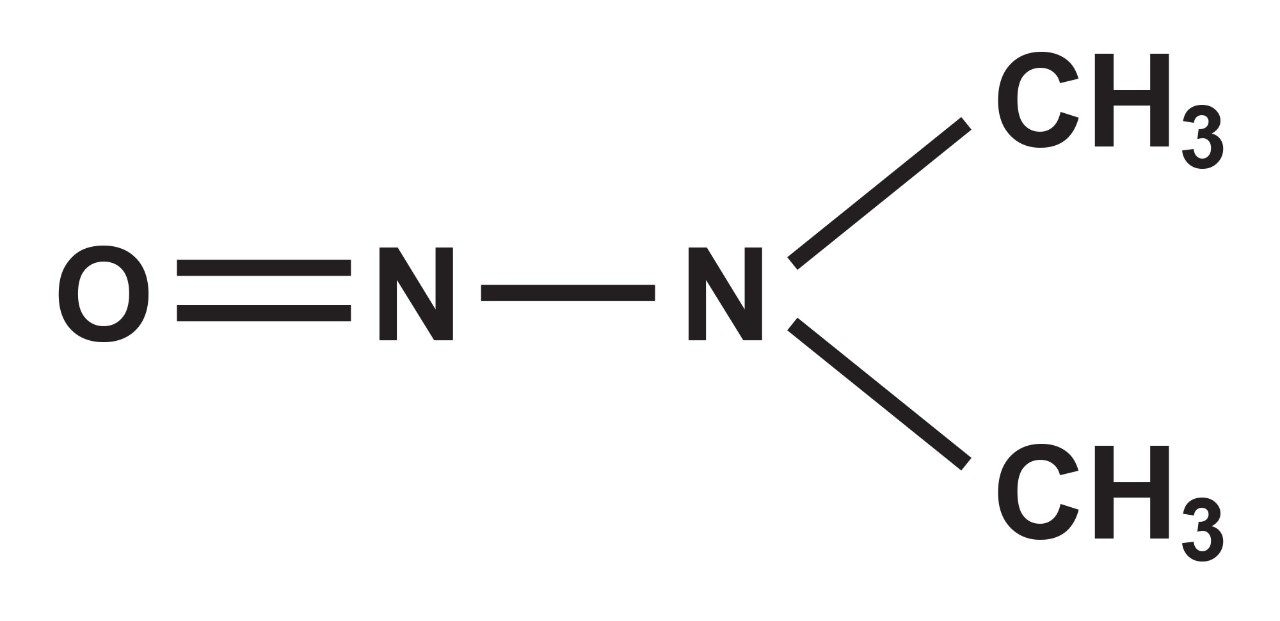
Genotoxic impurities (GTIs), arising from the synthesis/manufacturing from many drug products, specifically suspected carcinogenic N-nitroso compounds, have been found in several medicines. This has resulted in many drug recalls.
Through the testing thus far, FDA have found levels of NDMA in ranitidine that are similar exposure levels from eating common foods like grilled or smoked meats.1
Based on methods described in the 2018 ICH Guidance M7(R1) Assessment and Control of DNA Reactive (Mutagenic) Impurities in Pharmaceuticals to Limit Potential Carcinogenic Risk,1 the FDA has set the acceptable daily intake limit for NDMA at 96 ng/day (0.32 ppm) for ranitidine.
The goal of the work described herein, was to develop a sensitive and robust method for the LC-MS/MS quantification of NMDA containing the drug product, ranitidine.
Active pharmaceutical ingredient (API)
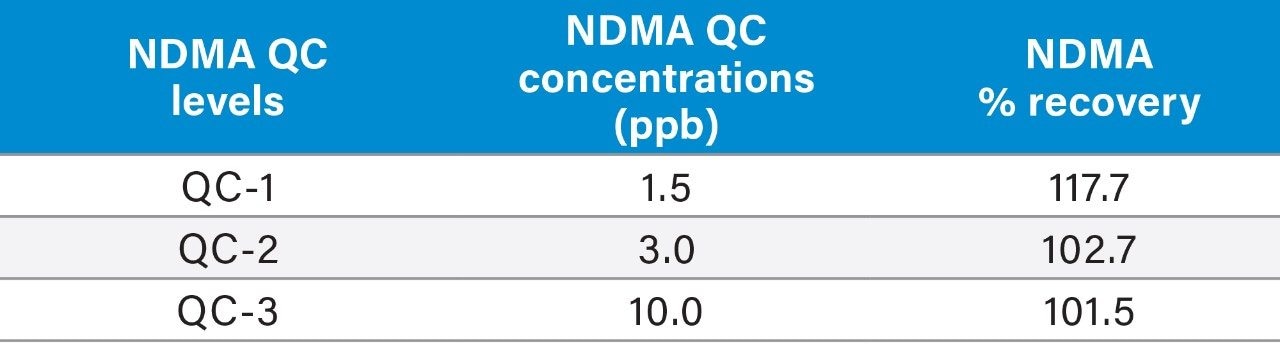
Three QC levels of NDMA (1.5, 3, and 10 ng/mL) containing 30 mg/mL ranitidine API was prepared.
|
LC system: |
ACQUITY UPLC H-Class PLUS |
|
Vials: |
Waters Total Recovery Vials [p/n 186007197C] |
|
Column: |
XSelect HSS T3 5 μm, 4.6 × 150 mm (p/n 186004791) |
|
Column temp.: |
40 °C |
|
Sample temp.: |
10 °C |
|
Injection volume: |
15 μL |
|
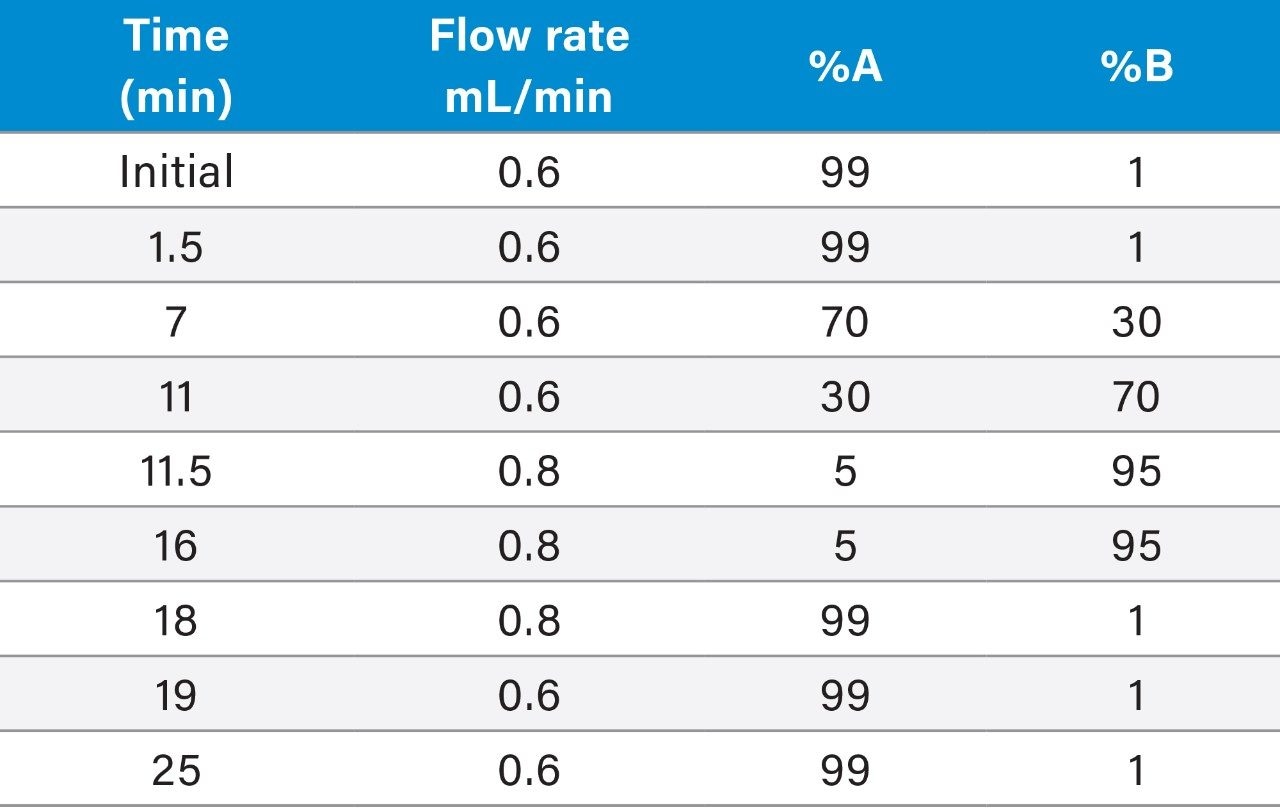
Flow rate: |
0.6 mL/min |
|
Mobile phase A: |
0.1% Formic acid in water |
|
Mobile phase B: |
0.1% Formic acid in methanol |
|
Diluent: |
50% methanol in water |
|
Needle wash: |
50% methanol in water |
|
MS system: |
Xevo TQ-S cronos |
|
Ionization mode: |
APCI positive ion mode |
|
Corona voltage: |
4.7 kV |
|
Desolvation gas: |
1000 L/Hr |
|
Cone gas: |
20 L/Hr |
|
Desolvation temp.: |
600 °C |
|
MS software: |
MassLynx v4.2, TargetLynx XS |

Using the Waters Xevo TQ-S cronos Tandem Quadrupole MS coupled to the ACQUITY UPLC H-Class PLUS System (Figure 2) and chromatographic separation using the Waters XSelect HSS T3 5 m, 4.6 × 150 mm Column, and MRM transitions listed in Table 2, a lower Limit of Quantification (LOQ) of NDMA 0.75 ppb (S/N 30) was observed (Figure 3). Linear dynamic range (R2 0.9998) of this assay was 0.75–1200 ppb (Figure 3), with no carry over observed in the blank sample proceeding the highest calibration standard. Representative QC performance is highlighted in Table 3. Use of the Waters XSelect HSS T3 Column provided necessary retention of NDMA, while providing adequate resolution fromdetecon.

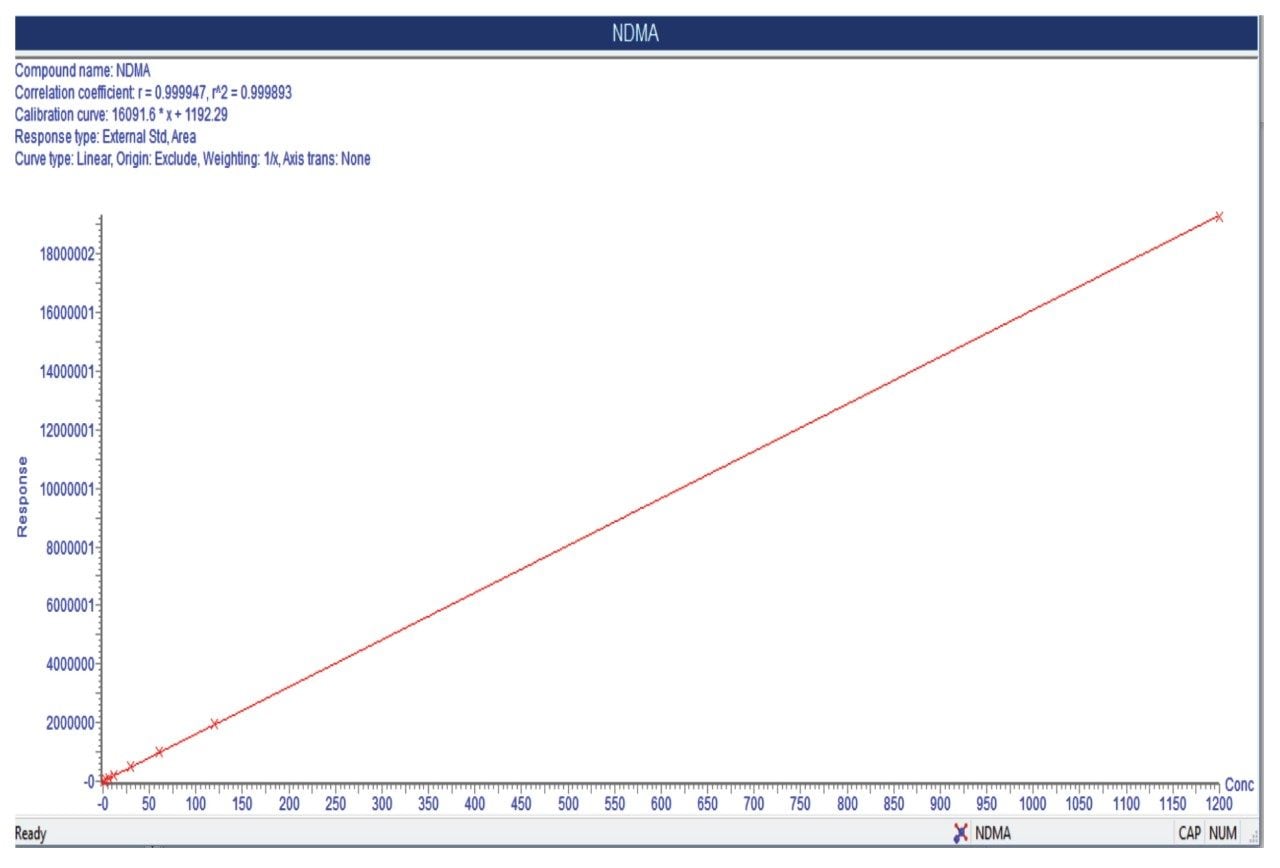
This application highlights accurate NDMA quantification, achieving LOQs of 0.75 ppb (11 picograms on-column), using the Waters ACQUITY UPLC H-Class PLUS System and XSelect HSS T3 Column for chromatographic separation, and the Xevo TQ-S cronos tandem MS configured with APCI probe for highly sensitive MS detection.
720006790, March 2020