In this Application note, Waters reversed phase columns were screened to separate a series of commercially available and relevant synthetic peptide sequences under different gradient-based separation conditions. Based on the screening results, a general guidance for column selection is provided.
Peptides are gaining more and more attention as potential biotherapeutics. Currently, more than 100 peptides are marketed worldwide. Therapeutic peptides have been derived from three sources: natural sources, recombinant technology, and chemical synthesis. The synthetic peptide approach has the advantage over the other two approaches in that the synthetic peptide can be generated in a quick and well-controlled way using solid phase peptide synthesis (SPPS). In addition, chemical modifications that can extend the chemical and structural diversity and stability of the peptides can be introduced fairly easily.1,2
Impurities in the final desired synthetic peptide product can be produced both during manufacturing and upon storage. Since these impurities can potentially affect the safety and efficacy of the therapeutic peptide, they need to be monitored and characterized.
Selecting optimal columns and separation methods for synthetic peptide impurity analysis is often not straightforward. In this study, Waters reversed phase columns were screened to separate a series of commercially available and relevant synthetic peptide sequences under different gradient-based separation conditions. Based on the screening results, a general guidance for column selection is provided.
All synthetic peptide samples were purchased from Bachem. Lyophilized materials were reconstituted in water to a concentration of 2 mg/mL based on the mass of the lyophilized peptide. The samples were further diluted to lower concentrations in 0.1% formic acid. Table 1 lists the peptides and their properties.
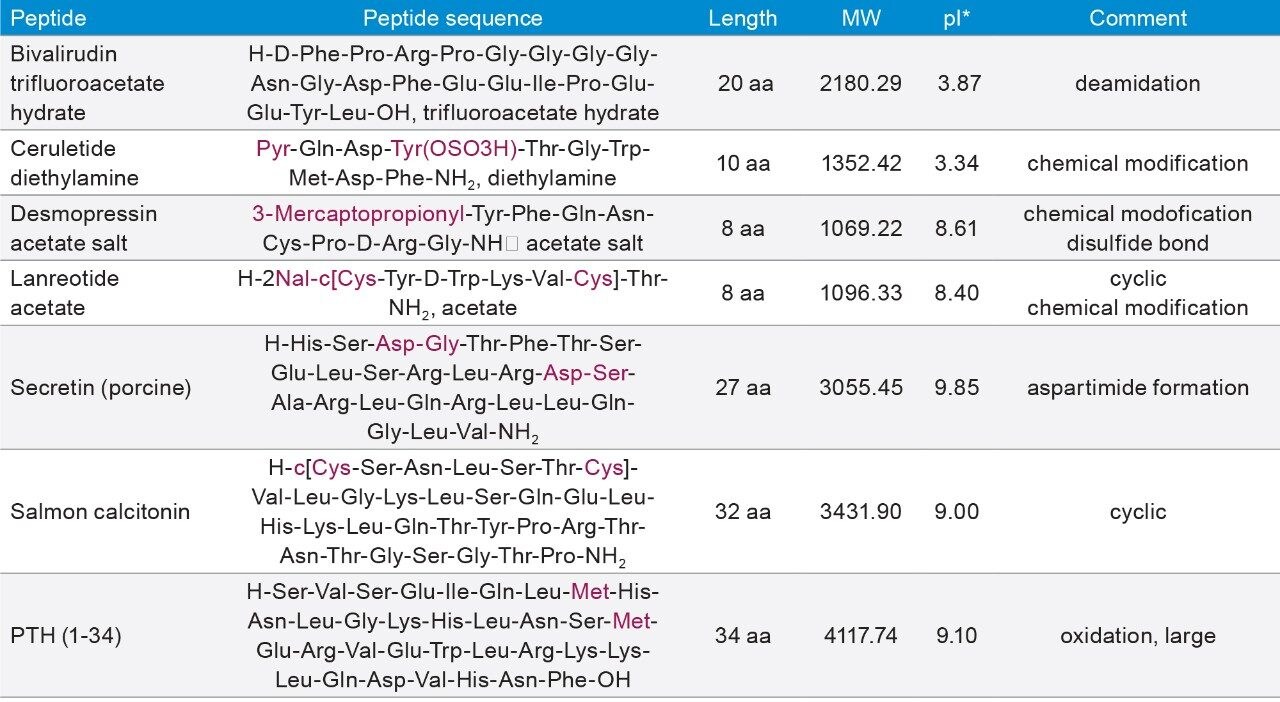
Table 1. List of synthetic peptides and their properties.
*: pIs are obtained from BioLynx without chemical modifications.
|
System: |
ACQUITY UPLC H-Class Bio |
|
|
Sample temp.: |
4 °C |
|
|
Analytical column temp.: |
60 °C |
|
|
Flow rate: |
0.3 mL/min (general gradient) |
|
|
Injection volume: |
2.5–10 μL |
|
|
Columns: |
ACQUITY UPLC Peptide BEH C18, 300A, 1.7 μm, 2.1 x 150 mm (p/n: 186003687) ACQUITY UPLC Peptide BEH C18, 130A, 1.7 μm, 2.1 x 150 mm (p/n: 186003556) ACQUITY UPLC Peptide CSH C18, 130A, 1.7 μm, 2.1 x 150 mm (p/n: 186006938) ACQUITY UPLC Peptide HSS T3, 100A, 1.8 μm, 2.1 x 150 mm (p/n: 186008756) ACQUITY UPLC CSH Phenyl-Hexyl, 130A, 1.7 μm, 2.1 x 150 mm (p/n: 186005408) ACQUITY UPLC CSH Fluoro-Phenyl, 130A, 1.7 μm, 2.1 x 150 mm (p/n: 186005353) ACQUITY UPLC BEH C8, 130A, 1.7 μm, 2.1 x 150 mm (p/n: 186003377) |
|
|
Detection: |
ACQUITY UPLC TUV Detector with 5 mm titanium flow cell, 214 nm |
|
|
Sample collection/vials: |
LCGC Certified Clear Glass 12 x 32 mm Screw Neck Total Recovery Vial, with cap and Preslit PTFE/Silicone Septa 1 mL volume (p/n: 186000385C) |
|
|
Mobile phase A: |
0.1 % (v/v) trifluoroacetic acid (TFA) or 0.1% (v/v) formic acid (FA) or 20 mM ammonium formate, pH 10 in water |
|
|
Mobile phase B: |
0.1 % (v/v) trifluoroacetic acid (TFA) or 0.085% (v/v) formic acid (FA) or 20 mM ammonium formate, pH 10 in acetonitrile |
|
|
Data management: |
MassLynx v4.1 |
|
Time |
Flow Rate(mL/min) |
%A |
%B |
Curve |
|---|---|---|---|---|
|
0.00 |
0.3 |
74 |
26 |
- |
|
2.00 |
0.3 |
74* |
26* |
11 |
|
22.00 |
0.3 |
54 |
46 |
6 |
|
24.00 |
0.3 |
5 |
95 |
6 |
|
24.01 |
0.3 |
74 |
26 |
11 |
|
34.00 |
0.0 |
74 |
26 |
11 |
*The %A and %B composition changes with different mobile phases and different peptides. However, the gradient slope was kept constant as Δ20%B per 20 mins.
Solid phase peptide synthesis (SPPS) is a common technique used for chemical synthesis, and offers a number of advantages over solution phase synthesis. The general principle of SPPS is one of repeated cycles of deprotection-wash-coupling-wash. This technique uses a solid support and adds amino acids in a sequential fashion. All soluble reagents and by-products are washed away. After the desired peptide has been synthesized, it is cleaved from the support with all amino acid blocking groups removed.
To start the SPPS, C-terminus of the first amino acid is coupled to an activated solid support, commonly a chemically unreactive polystyrol. The first amino acid, C-terminal, is coupled to a resin (there is a linker between the amino acid and the support). The resin acts as the C-terminal protecting group, the immobilized peptide can be retained during a filtration process while liquid-phase reagents and by-products of synthesis are flushed away.3
After completion of the synthesis, the desired peptide is cleaved from the resin. During each step of the synthesis, impurities will be introduced, which include amino acid deletion, amino acid insertion, truncated peptides, racemerization, by-products formed during synthesis, etc. Some impurities can still be present even after initial single step purification and isolation. In addition, peptide product degradation could occur during storage.4,5
Chromatographic methods are often employed to assess the levels of these impurities in synthetic peptide preparations, and reversed-phase chromatography is the most commonly used chromatographic technique.
In 2017, an application note was published by Waters, in which the performance of 10 reversed-phase columns were compared side-by-side for a peptide mapping application.6 While all of the columns that were evaluated were effective in this application, four columns were the most recommended for peptide mapping for the purpose of protein characterization and quality testing. These four columns are: Peptide BEH C18, 130Å; Peptide BEH C18, 300Å; Peptide CSH C18, 130Å; and Peptide HSS T3, 100Å.
In this study, a synthetic peptide impurity analysis was carried out by screening the four above mentioned columns, as well as three other columns based on the differences in selectivity and retentivity. Figures 1–7 show chromatograms of seven synthetic peptides separated on Waters’ reversed-phase columns under different mobile phase conditions. The results for the individual peptides are summarized below.
Bivalirudin (Brand name: Angiomax, Angiox) is a specific and reversible direct thrombin inhibitor.7 It is used with aspirin to decrease the clotting ability of the blood and to help prevent harmful clots from forming in blood vessels. Specifically, it is used in patients who are having certain heart and blood vessel procedures, such as coronary angioplasty.8
Bivalirudin is a 20 amino acid peptide, with a monoisotopic mass of 2178.986 Da. It has an asparagine (Asn) residue, which could undergo deamidation. In addition, the poly-glycine motif in the sequence is of great interest.
Figures 1a, 1b, and 1c show separations of bivalirudin and its impurities using formic acid, TFA, and ammonium formate (pH 10) mobile phases, respectively. A few observations were made upon close examination of the chromatograms:
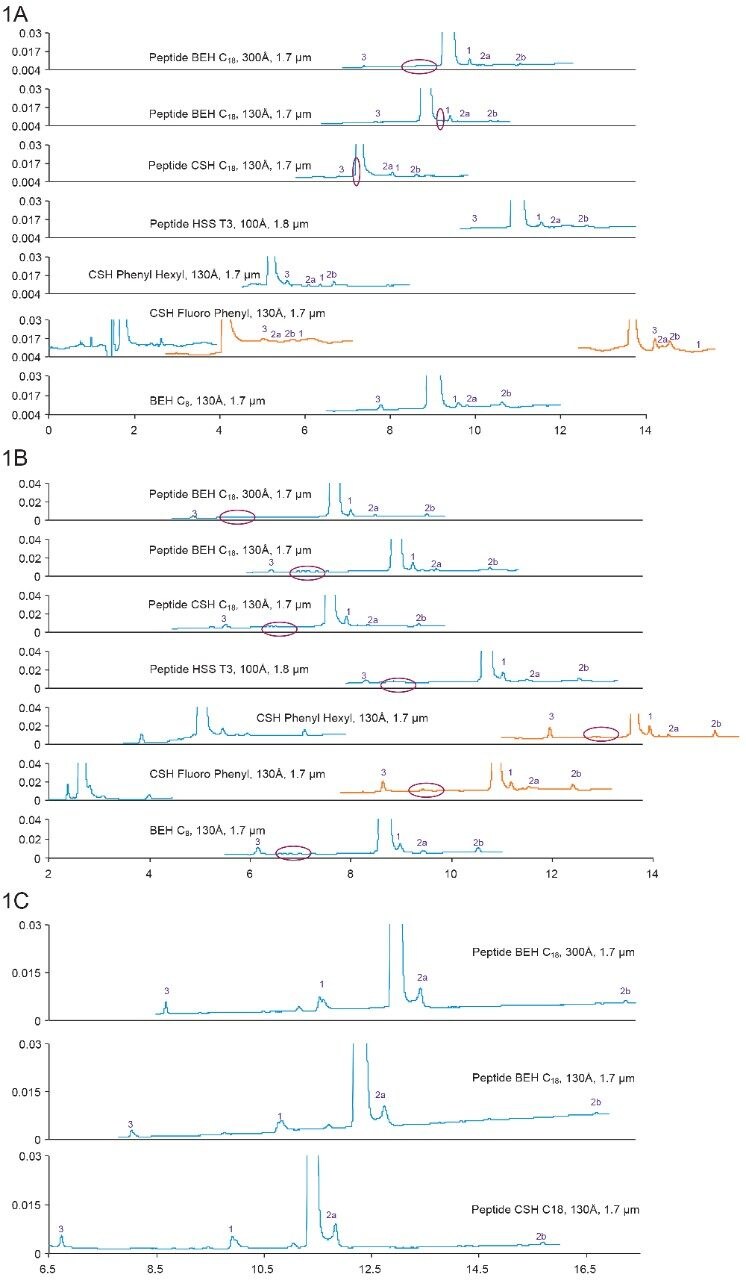
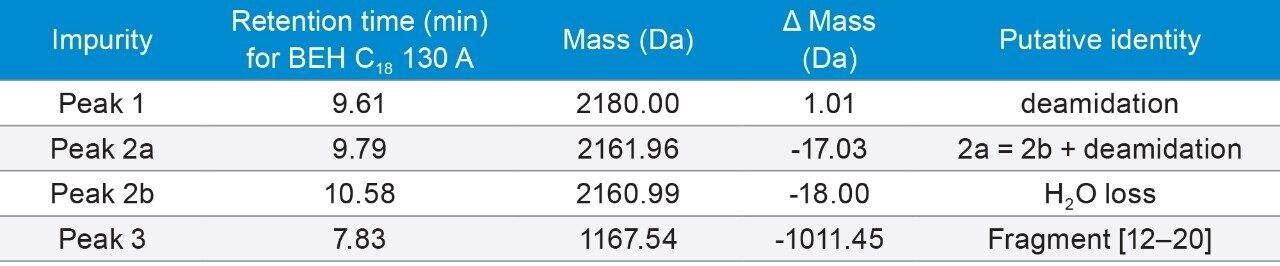
1. Selectivity
In order to show selectivity of the columns, major peaks were identified using mass spectrometry. The putative identities are listed in Table 2. As predicted, deamidation was observed as one of the major impurities. Selectivity changes not only among columns, but also under different mobile phase conditions, were observed as shown in Figures 1a–1c. The change in selectivity among columns can be explained by subtle differences in column chemistry such as base particle, surface charge, ligand type, and ligand density; while the drastic changes in selectivity under different mobile phases can be explained by the interaction (both hydrophobic and ionic) between the peptide and its impurities, and the stationary phase under different pH and ion pairing conditions.
2. Retentivity
Under the same gradient conditions (18–38% B in 20 min), the tested columns exhibit different retentivity. CSH Columns have the least retentivity, while the HSS T3 Column has the most retentivity. This has been observed on many other synthetic peptides, and is in agreement with peptide mapping results.6 Among the CSH family, CSH Fluoro Phenyl Column has the least retentivity, followed by CSH Phenyl Hexyl and Peptide CSH C18 columns. Since the main peak eluted near the void on the CSH Fluoro Phenyl Column, lower starting organic percentage was used in this case (orange traces).
3. Loading capacity/peak capacity
High loading capacity is important when one wants to see very low abundance impurities. High loading capacities are also very beneficial for purification work.
As can be seen, the peak width of the main peak is generally narrower on CSH Columns than on other columns, especially under FA mobile phases. This has been seen with many other synthetic peptides, and is also in agreement with peptide mapping results that showed CSH Columns as generally having highest peak capacity.6 As a result, more sample can be loaded onto CSH Columns without sacrificing resolution.
Unique impurity peaks were found on some columns and were marked with orange ovals (Figure 1A).
Under FA-mobile phase conditions, a group of impurities that appeared only on the Peptide BEH C18, 300Å Column were identified as fragments [6–20], [7–20], [8–20], [9–20], [10–20], with shorter fragments eluting out later (Figure 1A). Interestingly, all of these fragment peaks were observed under TFA-mobile phase conditions on all of the columns (Figure 1B).
The unique peak observed on the Peptide BEH C18, 130Å Column has a mass that is 129 Da lower than the main peak, while the unique peak was observed on the Peptide CSH C18, 130Å Column is likely to be a glycine insertion (Figure 1A).
4. Additional peaks were also found under different mobile-phase conditions. For example, fragments [3–20] and [11–20] were identified under TFA conditions, and Fragment [1–11] was identified under ammonium formate (pH 10) conditions. Under ammonium formate (pH 10) conditions, Peak 1 split into two peaks with the same mass (Figure 1C). This behavior suggests that these two peaks are diasteromers.
Ceruletide (Brand name: Takus, Tymtran) stimulates gastric, biliary, and pancreatic secretion and certain smooth muscle. It is used as a diagnostic aid in pancreatic malfunction.9 Ceruletide is also known as cerulein or caerulein.
Ceruletide is a 10 amino acid peptide, with a monoisotopic mass of 1351.449 Da. It has a chemical modification – sulfonic acid – which is predicted to make the molecule extremely acidic. In addition, it has a pyroglutamic acid residue at the N-terminus, a methionine (Met) residue that could be prone to oxidation, and an amidated C-terminus.
Figures 2a, 2b, and 2c showed chromatographic separation of ceruletide and its impurities on the columns tested under different mobile phases. Table 3 lists a few major impurities.
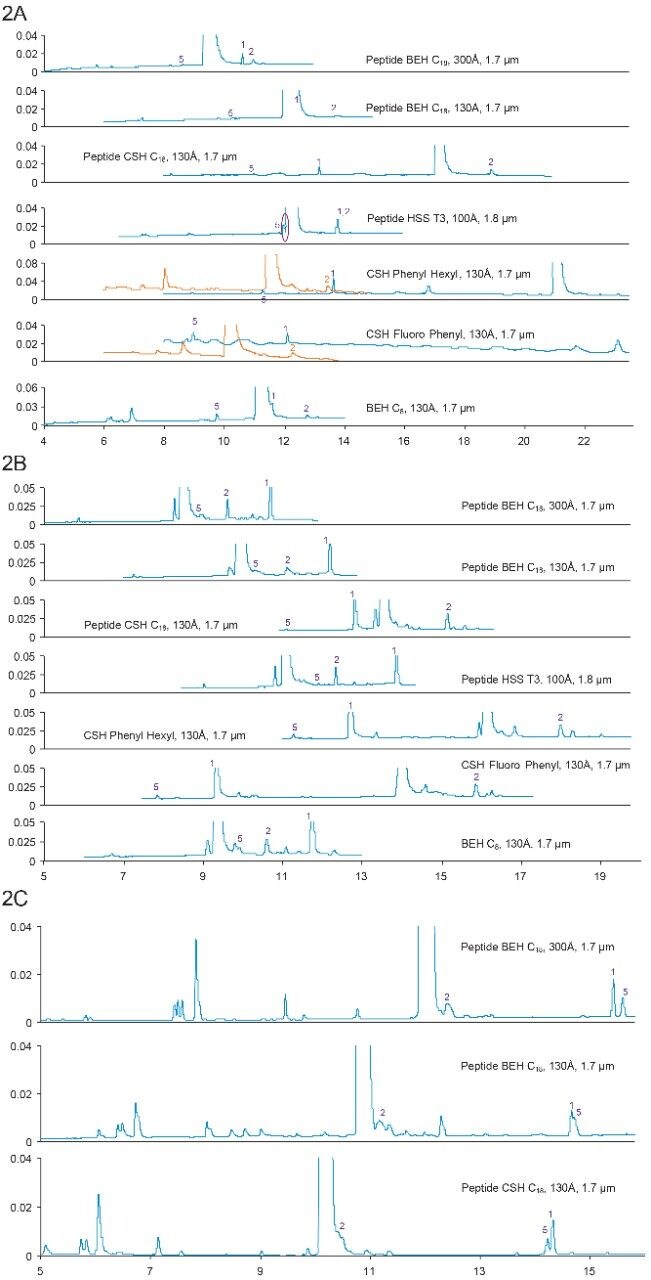
Figure 2A. Separation of ceruletide with formic acid mobile phases. Gradient: 18–38% B in 20 min. Orange traces: 28–48% B in 20 min for CSH Phenyl Hexyl;35–55% B in 20 min for CSH Fluoro Phenyl.
2B. Separation of ceruletide with TFA mobile phases. Gradient: 18–38% B in 20 min.
2C. Separation of ceruletide with ammonium formate (pH 10) mobile phases. Gradient: 10–30% B in 20 min.
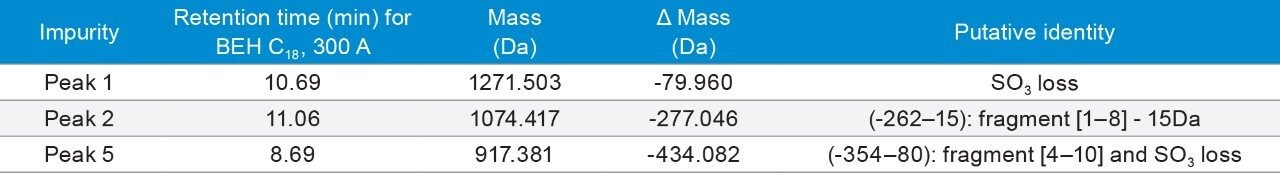
The most interesting observation is the retentivity of ceruletide on various columns. Typically, under the same gradient conditions, peptides have the lowest retentivity on CSH Columns and the highest retentivity on a Peptide HSS T3 Column. However, it is not the case for ceruletide. Instead, ceruletide has the highest retentivity on CSH Columns, and have similar retentivity on a Peptide HSS T3 column as the Peptide BEH C18, 130Å Column. The main peak didn’t elute on the CSH Fluoro Phenyl Column, while for the previous peptide, bivalirudin, the main peak came out at the void on the CSH Fluoro Phenyl Column. Indeed, the retentivity of Ceruletide on CSH Columns and a Peptide HSS T3 Column is reversed.
Our hypothesis is that since ceruletide is a very acidic peptide, it is still negatively charged even under the acidic conditions (FA and TFA) due to the sulfonic acid moiety. Since CSH particles have a controlled low level of positive charge, the negatively charged peptide will interact with positively charged CSH particles. As a result, the peptide eluted later on the CSH Columns. On the other hand, Peptide HSS T3 particles are silica-based. The negatively charged silanol group would repulse the negatively charged ceruletide. As a result, the retentivity is greatly decreased on the Peptide HSS T3 Column. It’s worth noting that the retention time difference under FA conditions is much larger than that under TFA conditions.
Interestingly, the retentivity of ceruletide became “normal” under ammonium formate (pH 10) mobile phase conditions (Figure 2C) – it was the least retained on the Peptide CSH C18 Column. Consistent with our hypothesis, under pH 10, the positive charges on the CSH particles are neutralized. Therefore, the ionic interaction between ceruletide and the CSH particles is dramatically reduced. As a result, ceruletide had the least retentivity on the Peptide CSH C18 Column, which is typically observed for other peptides. Tests were not run on HSS T3 Columns because the particles are silica-based, which is not stable at pH 10.
2. Selectivity
Similarly to what has been seen on bivalirudin, column chemistry and mobile-phase conditions play critical roles in changing the selectivity of the separation. For example, under FA and TFA conditions, Peak 1 eluted before the main peak for all CSH Columns, while for other columns, it eluted after the main peak. Under pH 10 mobile phases, Peaks 1 and 5 eluted very closely, while under acidic conditions (FA and TFA), the two peaks were much more separated.
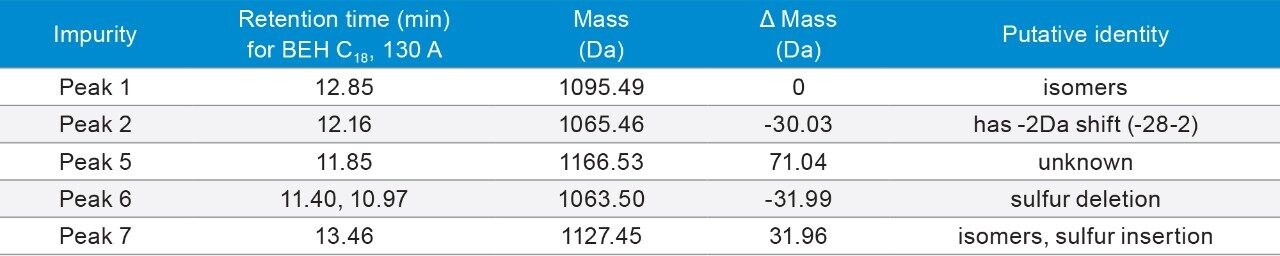
3. Under FA conditions, a unique peak was identified on the Peptide HSS T3, 100Å Column, which is likely to be pyroglutamic acid deletion and sulfur trioxide (SO3) deletion. Under TFA condition, a new peak was observed right before the main peak on most of the columns, and it is likely an isomer of the main peak. Other impurities include amino acid deletion, dimer, +1 Da (presumably deamidation), -64 Da, +16 Da (presumably oxidation), with combination of SO3 loss and fragments.
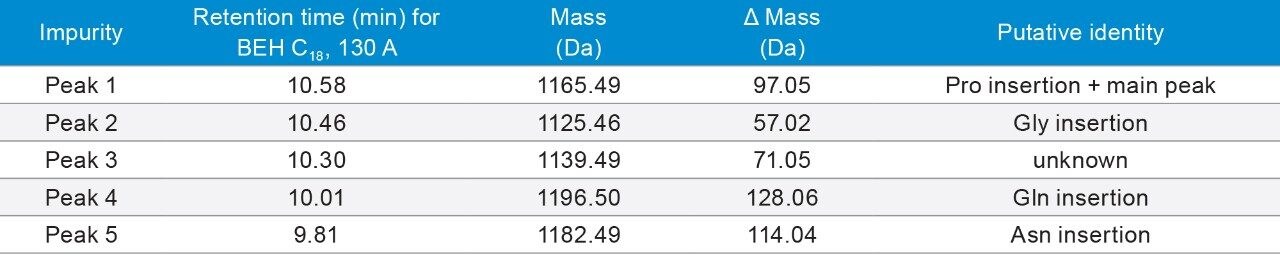
Desmopressin (Brand name: DDAVP, Minirin, Stimate) is a synthetic form of vasopressin. It is used to treat diabetes insipidus, bedwetting, hemophilia A, von Willebrand disease, and high blood urea levels.10 Desmopressin has eight amino acids, with a monoisotopic mass of 1068.427 Da. It has a 3-mercaptopropionyl modification at N-terminus, and an amidation at C-terminus. Desmopressin is a cyclic peptide, with a disulfide bond formed between the modified moiety and cysteine (Cys) residue. It also has an asparagine (Asn) residue that could undergo deamidation.
Peaks indicated by the asterisk (*) are +1 Da peaks of the main peak, which could be deamidation of Asn. It was not clear at first whether these impurities occurred during chromatographic separation or during storage. When a new sample was diluted from the stock solution and injected onto one of the columns, the peak area reduced significantly (data not shown). Therefore, it is likely that these impurities are degradants that occurred during storage.
As shown in Table 4, multiple occasions of amino acid insertion were found as putative impurities.
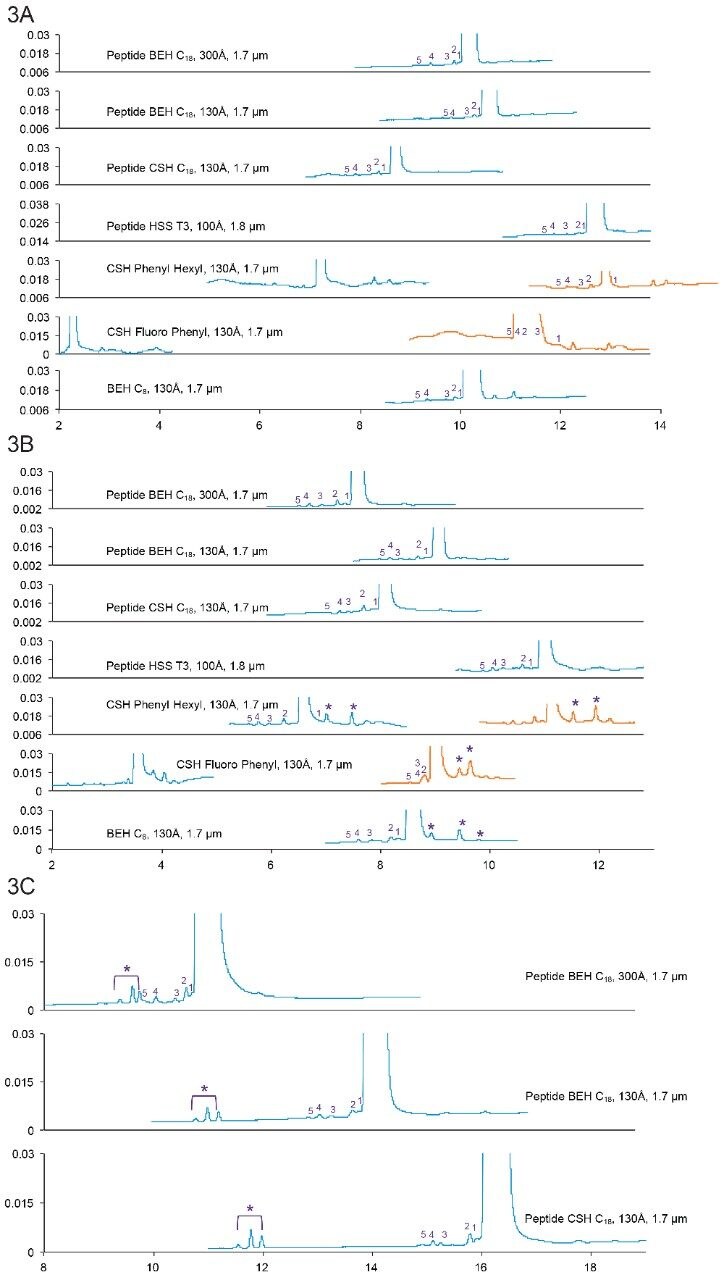
Figure 3A. Separation of Desmopressin with formic acid mobile phases. Gradient: 10–30% B in 20 min. Orange trace: 5–25% B in 20 min.
3B. Separation of Desmopressin with TFA mobile phases Gradient: 14–34% B in 20 min. Orange trace: 8–28% B in 20 min. 3C. Separation of Desmopressin with ammonium formate (pH 10) mobile phases. Gradient: 12–32% B in 20 min.
Lanreotide (Brand name: Somatuline Autogel, Somatuline Depot) is a synthetic analogue of somatostatin, a naturally occurring inhibitory hormone which blocks the release of several other hormones, including growth hormone, thyroid-stimulating hormone (TSH), insulin, and glucagon.11 Lanreotide has eight amino acids, with a monoisotopic mass of 1095.476 Da. It has a chemical modification (2-naphthyl) that increases the hydrophobicity of the molecule, a disulfide bond, and an amidated C terminal.
Under FA-mobile phases, Peak 1 (an isomer) was well-separated from the main peak. However, under TFA-mobile phases, only a shoulder peak appeared next to the main peak (data not shown). This again, indicates that selectivity is different using different mobile phases.
Two isomers of Peak 6 were found separated on most of the columns, as the main component under both peaks has the same mass.
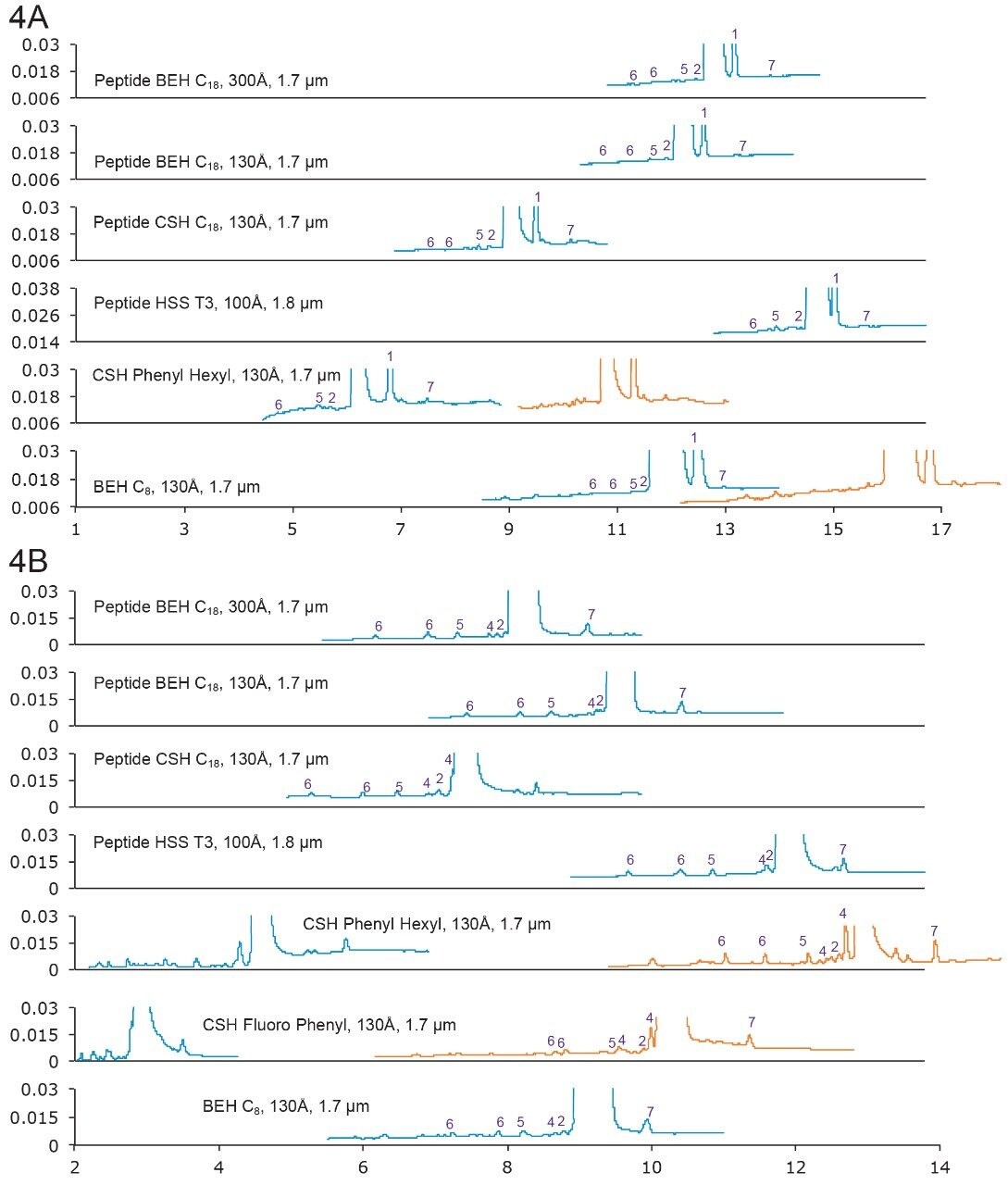
Figure 4A. Separation of lanreotide with formic acid mobile phases. Gradient: 12–32% B in 20 min. Orange trace: 8–28% B in 20 min.
4B. Separation of lanreotide with TFA mobile phases. Gradient: 20–40% B in 20 min. Orange trace: 12–32% B in 20 min.
This peptide (Brand name: SecreFlo) is the synthetic form of natural secretin. Its main effect is to regulate the pH of the small intestine’s contents through the control of gastric acid secretion and buffering with bicarbonate. Natural secretin was the first hormone to be discovered.12
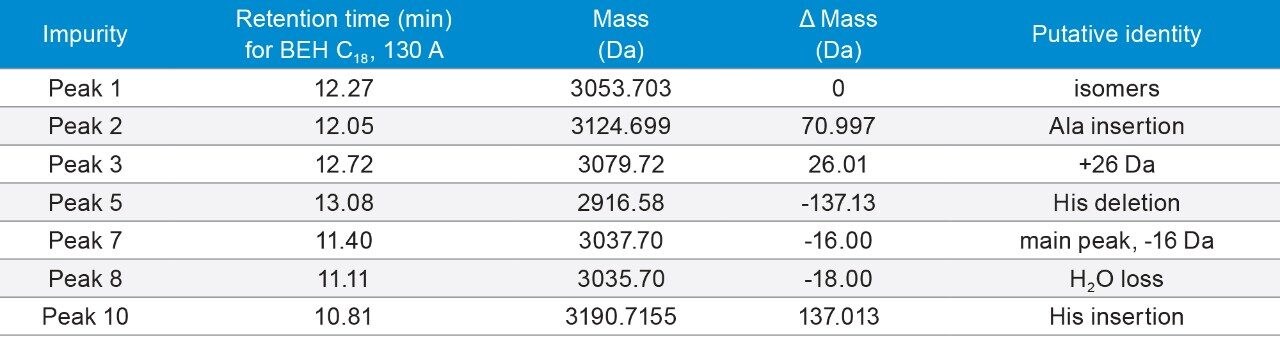
Secretin has 27 amino acids, with a monoisotopic mass of 3054.632 Da. Its C-terminus is amidated. It contains Asp-Gly and Asp-Ser motifs that can undergo aspartimide formation, which generates isomers.13 Indeed, three and five isomers were observed under FA and TFA conditions, respectively.
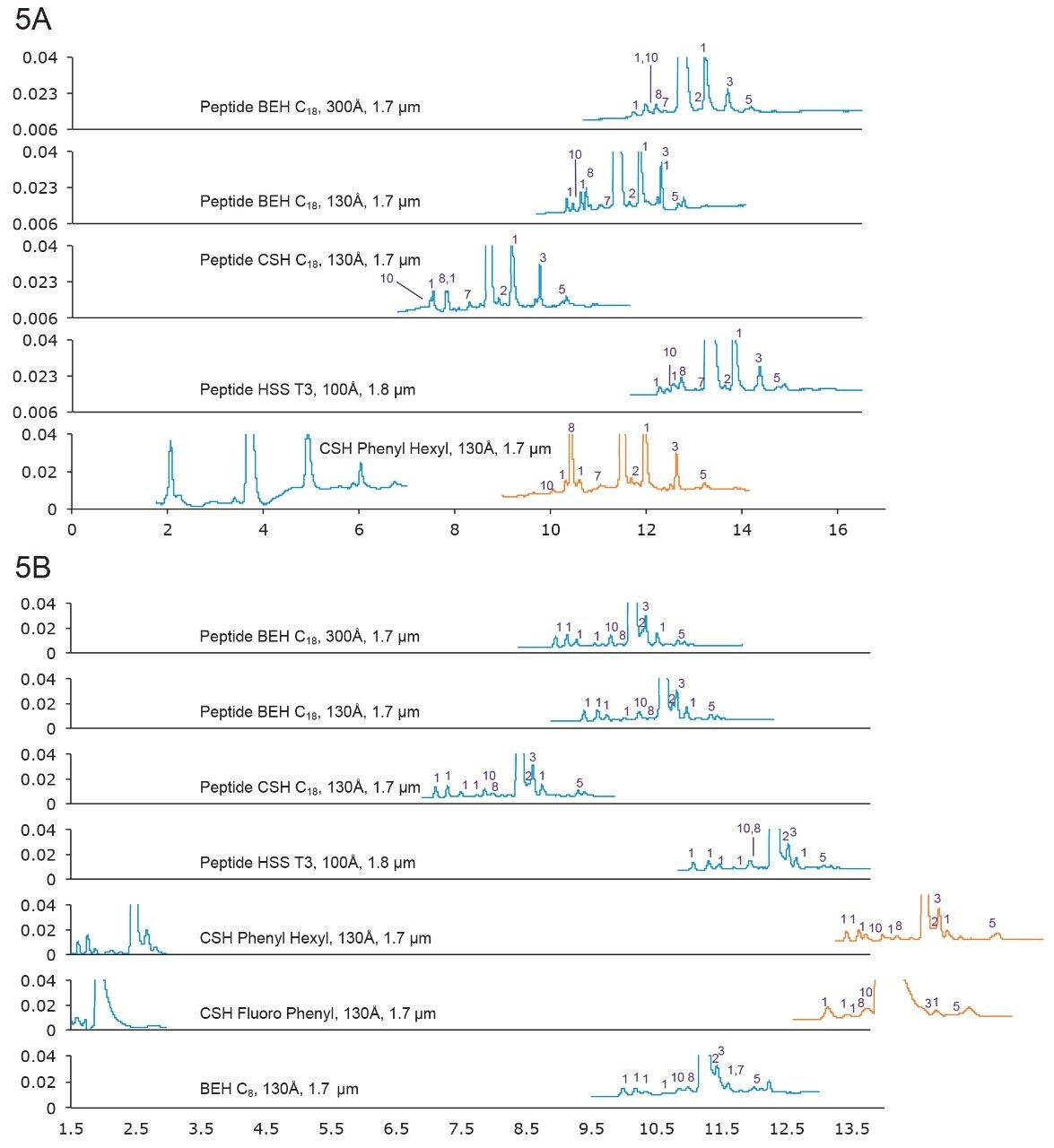
Figure 5A. Separation of Secretin with formic acid mobile phase. Gradient: 18–38% B in 20 min. Orange trace: 12–32% B in 20 min.
5B. Separation of Secretin with TFA mobile phase. Gradient: 26–46% B in 20 min. Orange trace: 16–36% B in 20 min.
Calcitonin-salmon (Brand name: Miacalcin) is a synthetic form of a hormone that occurs naturally in the thyroid gland used to treat Paget’s disease of bone, postmenopausal osteoporosis, or high levels of calcium in the blood.14
Calcitonin is a 32 amino acid peptide, with a monoisotopic mass of 3429.713 Da. It’s a cyclic peptide, with the C-terminus amidated.
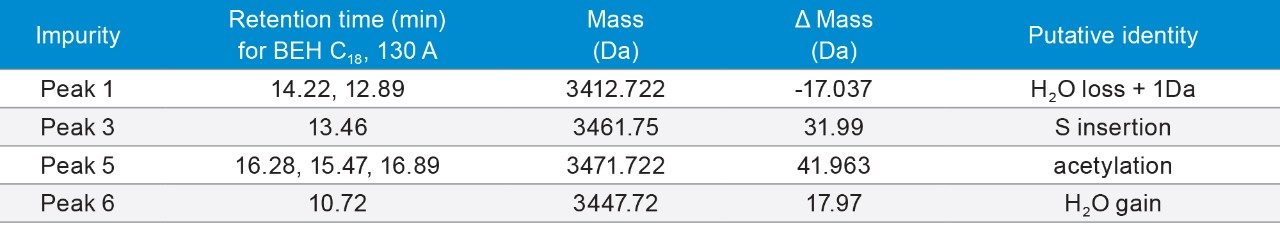
Some of the major impurities include sulfur insertion and acetylation, as shown in Figures 6a and 6b, and listed in Table 7.
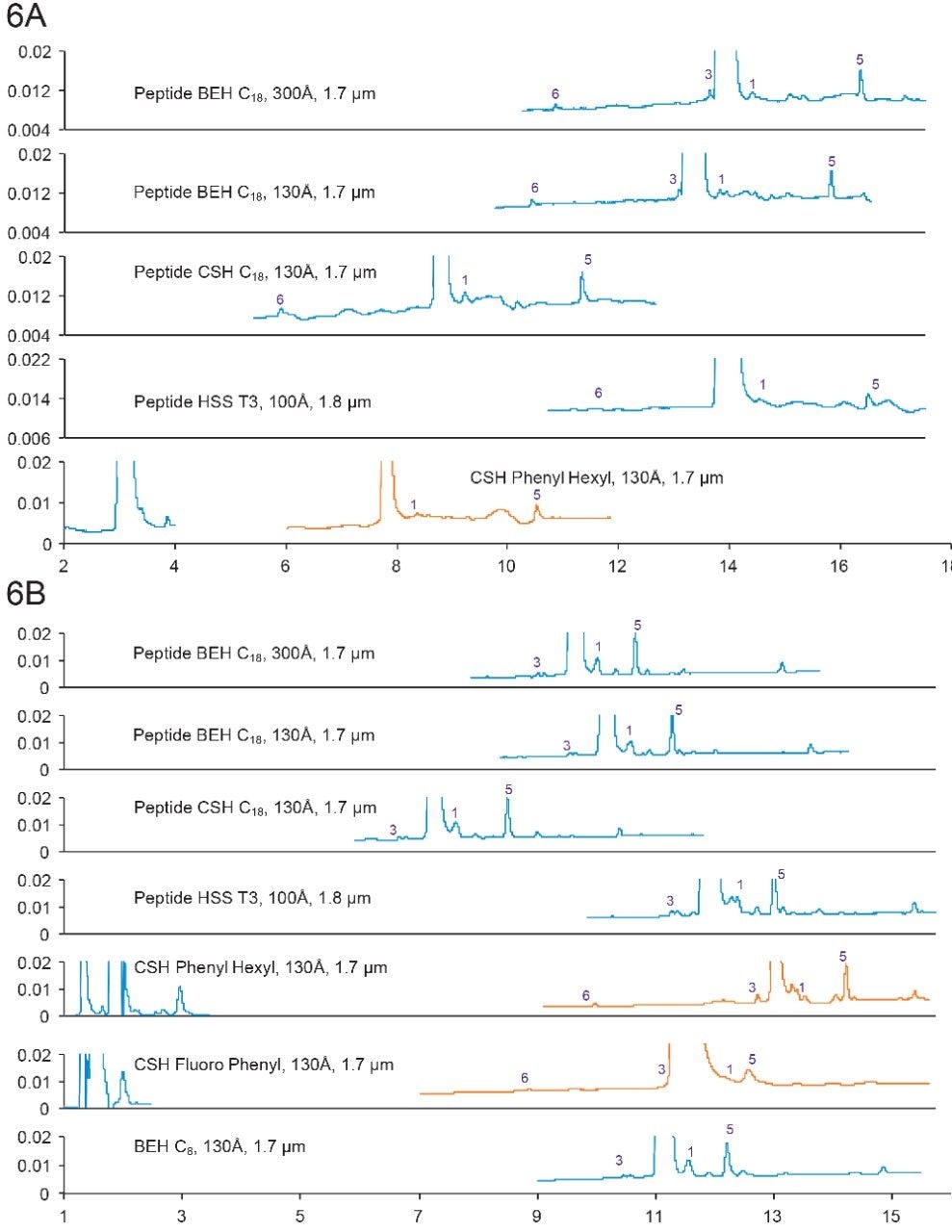
Figure 6A. Separation of Calcitonin with formic acid. Gradient: 18–38% B in 20 min. Orange trace: 15–35% B in 20 min.
6B. Separation of Calcitonin with TFA. Gradient: 28–48% B in 20 min. Orange trace: 18–38% B in 20 min.
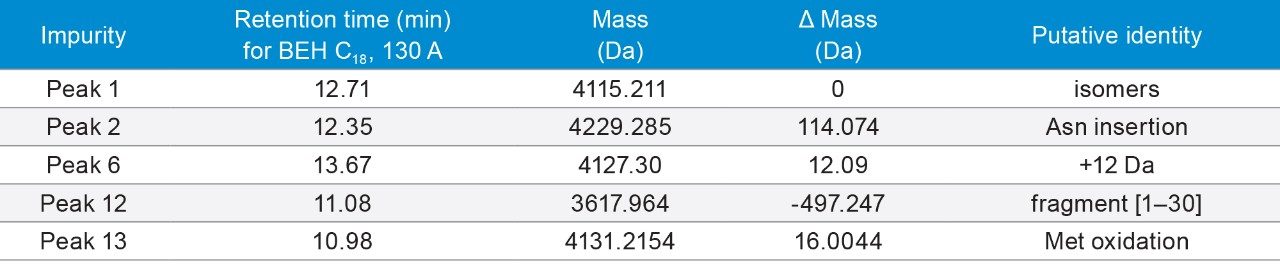
p-TH (1–34) (Brand name: Forteo) is a peptide fragment (34 amino acids) of the naturally occurring human parathyroid hormone (PTH) that is an important regulator of calcium and phosphorus metabolism.15 It is used to treat osteoporosis.
p-TH (1–34) has a monoisotopic mass of 4115.131 Da. It contains two methionine (Met) residues, which are prone to oxidation. Indeed, an oxidation peak (Peak 13) was found as one of the major impurities.
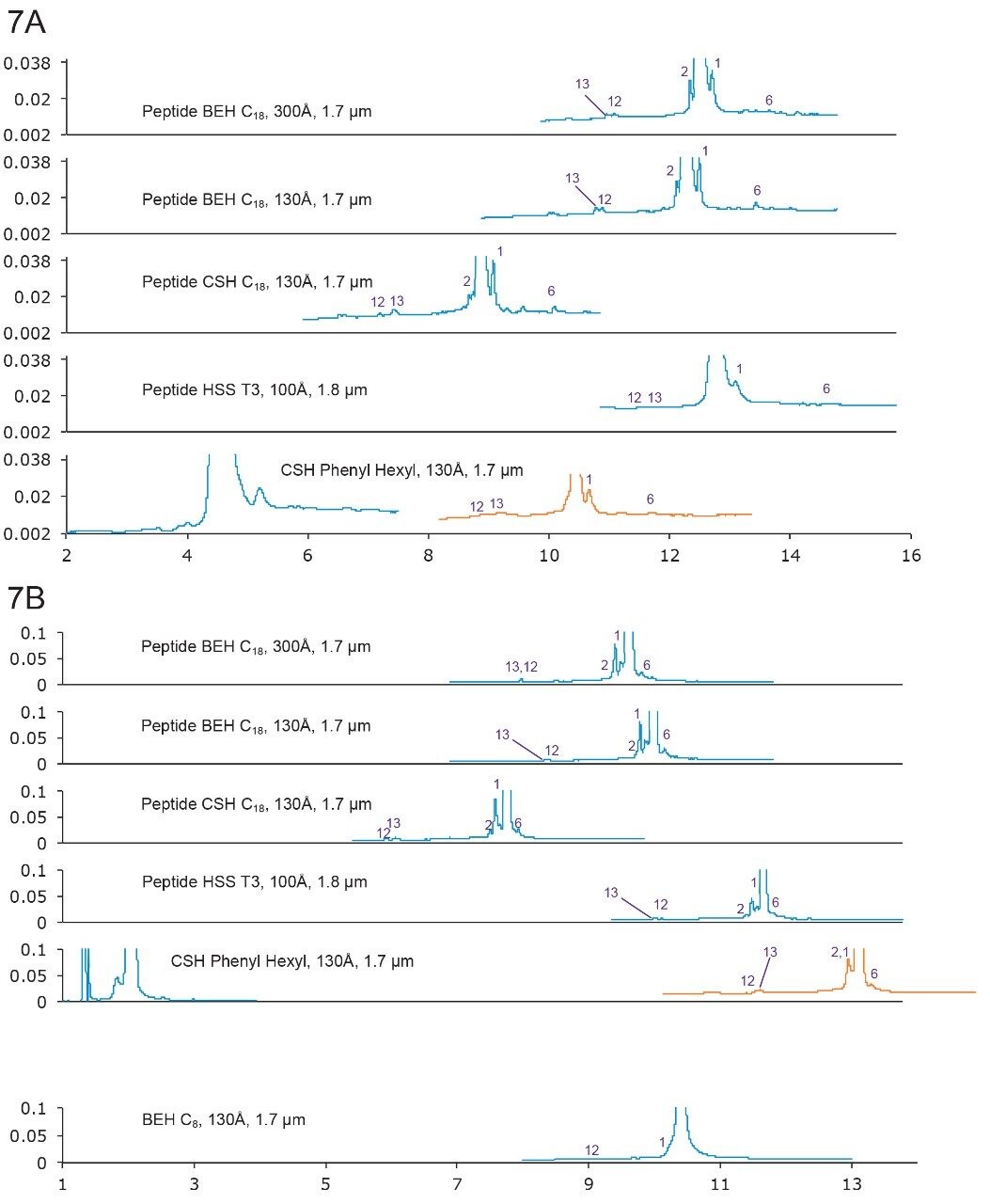
Figure 7A. Separation of p-TH with formic acid. Gradient: 15–35% B in 20 min. Orange trace: 10–30% B in 20 min.
7B. Separation of p-TH with TFA. Gradient: 24–44% B in 20 min. Orange trace: 14–34% B in 20 min.
In this study, a variety of synthetic peptides were separated on several reversed-phase columns under different mobile-phase conditions. Based on the chromatographic results, it is clear that there is no single column that is the most effective for all of the synthetic peptide impurity analyses attempted. As a result, screening columns for this application can be beneficial. Additionally, screening multiple columns with different selectivities for the analysis will increase the confidence that the final developed method is effectively monitoring the impurity profile in its entirety. In some cases, it is conceivable that complete coverage of the impurities may even require the use of separate analyses with two different columns.
However, screening all of the reversed-phase columns that may be effective for the analysis of a peptide may not be feasible or reasonable. As a starting point, the analyst may find value in comparing the characteristics of the peptide that they are analyzing to those tested in this study to potentially narrow down the column options in order to find the column chemistry with the best selectivity characteristics for their method. While all of the columns evaluated, among numerous other reversed-phase columns manufactured by Waters, are reproducibly manufactured and are well-suited to provide consistent separations, only four of these chemistries are currently branded as “peptide” columns. These “peptide” branded columns have been shown to perform consistently over several years of manufacturing for peptide separations and are routinely batch tested with a peptide mixture to provide additional assurance of reproducibility in peptide separations. These columns and their significant characteristics are:
For general use
For more retention in FA-mobile phases, use 300Å column for higher molecular weight peptides (>2100 Da) or bulky peptides (e.g., PEGylated peptide); use 130Å column for lower molecular weight peptides (<1400 Da)
Significant selectivity differences compared to BEH C18 phases
Narrowest peak width in both FA and TFA (highest loadability)
Lower retention than HSS T3 and BEH C18 phases (with exceptions, see ceruletide)
Highest peptide retention (with exceptions, see ceruletide)
Great retention for early eluting (short, hydrophilic) peptides
The other three columns tested in this study also have their own characteristics. Both the CSH Phenyl Hexyl and CSH Fluoro Phenyl columns have significant selectivity differences compared to BEH C18 phases, and they provide lower retention than CSH C18, HSS T3, and BEH C18 phases (with exceptions, see ceruletide). Although BEH C8, 130Å Column performed similarly to BEH C18, 130Å Column for the peptides tested in this study, it may provide better separation for large, hydrophobic peptides. For the columns that were not included in this study, many have been successfully used in peptide-related separations. For example, CORTECS C18, CORTECS UPLC C18+, and CORTECS UPLC T3 columns have been shown to separate synthetic peptides and tryptic peptides of monoclonal antibodies very well.6 They can be excellent choices for synthetic peptide impurity analysis.
720006244, April 2018