This application note presents a simple isocratic separation of 16 cannabinoids that can be used for the analysis of both plant material and concentrates to ensure the quality and safety of cannabis products.
The Alliance HPLC System is a highly reliable and robust instrument that can be used for routine analysis of cannabis and cannabis extracts.
As the legalization of cannabis for both medicinal and recreational use continues to advance, the need for simple and reliable analytical methods for the analysis of these products is desired by many industry stakeholders (producers, regulators, and consumers). The cannabis plant (Cannabis sativa) is a complex natural product that is known to produce at least 100 different cannabinoids.1 Traditionally, laboratories have focused their analysis on five primary compounds: delta-9-tetrahydrocannabinol (d9-THC), delta-9-tetrahydrocannabinolic acid (THC-A), cannabidiolic acid (CBD-A), cannabidiol (CBD), and cannabinol (CBN). Recently, more attention has been paid to some of the other minor cannabinoids, particularly in the medicinal market, which requires methods that are capable of higher resolution separations. Many of the minor cannabinoids have shown some medicinal effects so it is important to have the ability to separate and identify them. It is also desirable to have methods that minimize peak co-elutions of minor and major cannabinoids and provide accurate quantitative results. Several industry standardization bodies are currently developing harmonized methods for cannabinoid profiling to minimize the inherent variability in sampling, extraction, and instrumental analysis with an aim to improve potency result accuracy. High Performance Liquid Chromatography (HPLC) is the preferred methodology for potency determination (Total THC, Total CBD, or THC/CBD ratio) as it can identify and measure the structurally similar cannabinoids and their different forms (e.g. free form and corresponding acid forms of THC and CBD) in a single analysis. This application note presents a simple isocratic separation of 16 cannabinoids that can be used for the analysis of both plant material and concentrates to ensure the quality and safety of cannabis products.
|
LC system: |
Alliance HPLC |
|
Column: |
CORTECS Shield RP18 2.7 μm, 4.6 mm × 150 mm (p/n: 186008685) |
|
Analytical flow rate: |
2.0 mL/min |
|
Mobile phase A: |
Water with 0.1% TFA |
|
Mobile phase B: |
Acetonitrile |
|
Isocratic: |
41:59 mobile phase A/mobile phase B |
|
Oven temp.: |
35 °C |
|
Detector: |
2998 PDA |
|
Detection wavelength: |
228 nm at 4.8 nm resolution |
|
Injection volume: |
5 μL |
|
Software: |
Empower 3 CDS |
US DEA exempt reference standard solutions were obtained from Cerilliant Corporation, Round Rock, TX. These pre-dissolved solutions have been previously shown to be suitable for the generation of calibration curves2 when handled in an appropriate manner.3 Table 1 lists the cannabinoid standards used in this application note.
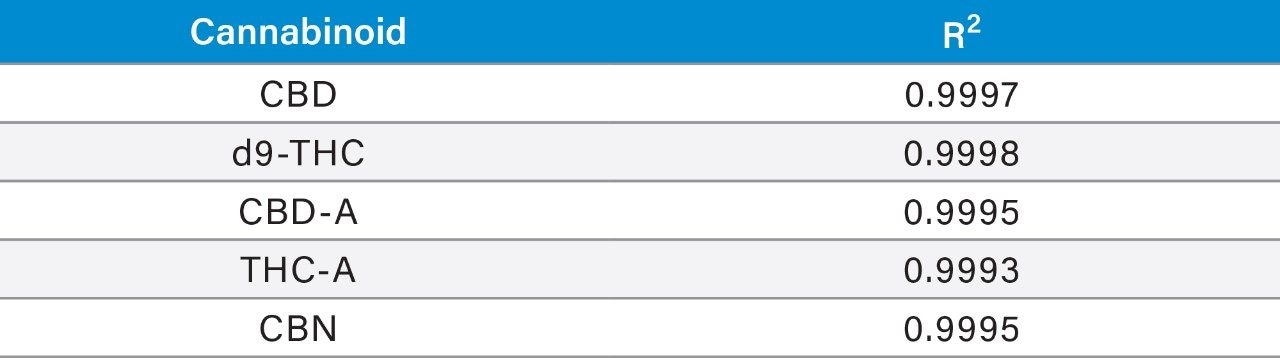
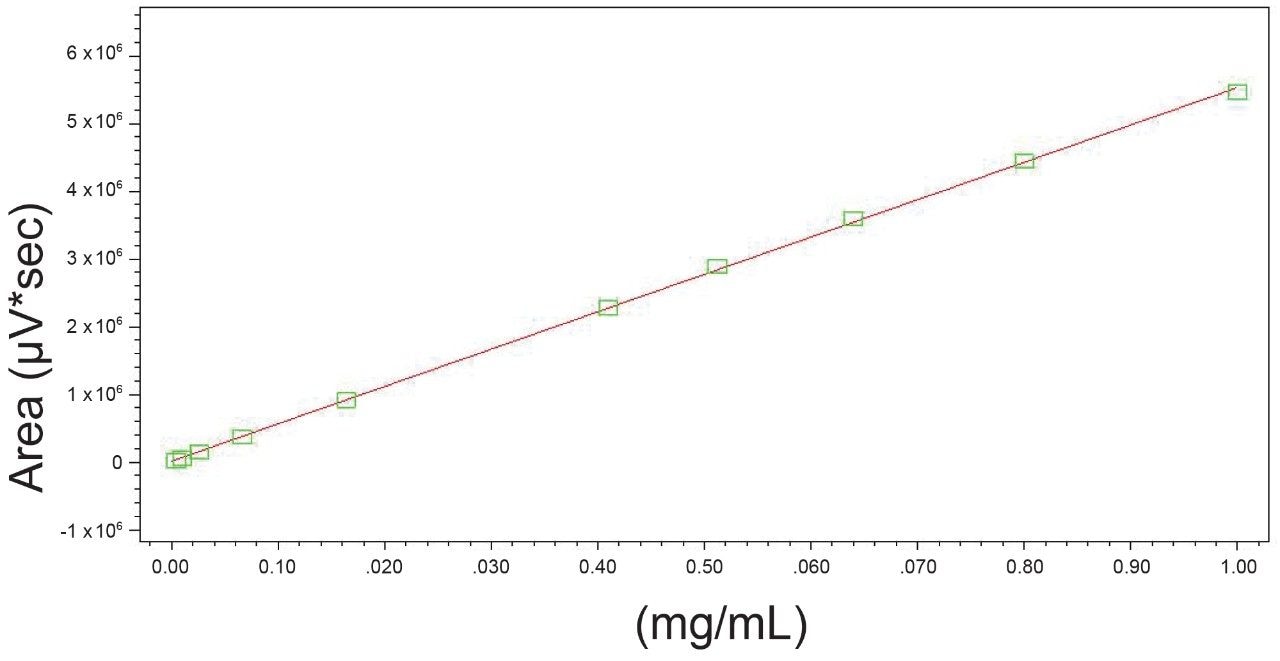
Linearity of major cannabinoids d9-THC, THC-A, CBD, CBD-A, and CBN was determined for 10 concentrations between 0.004 mg/mL and 1.000 mg/mL, prepared via serial dilution in methanol using DEA exempt standards.
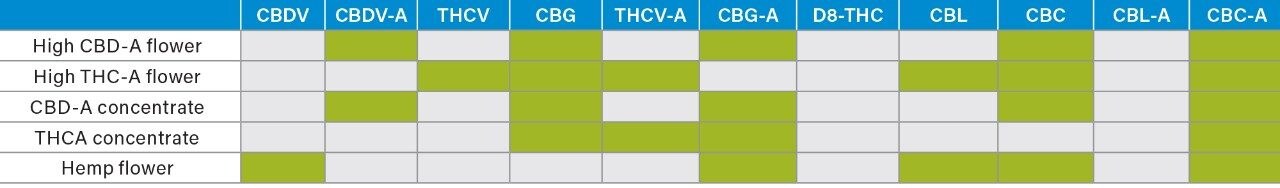
Four representative, pre-prepared samples were obtained from a local cannabis testing laboratory and one from a hemp processing laboratory. Samples were prepared as follows: For flower, a portion of homogenized plant material (Table 2) was added to acetonitrile or ethanol and sonicated for 20 minutes. The subsequent extract was filtered through a 0.22 µm syringe tip filter, diluted with methanol, and placed into a 2 mL sample vial ready for analysis. Concentrates were prepared similarly with isopropanol being used as the extraction solvent.

16 cannabinoids were fully separated in 26 minutes by HPLC using a reversed-phase isocratic method as shown in Figure 1. Resolution of all 16 compounds was calculated 2.5 or greater (Table 3).
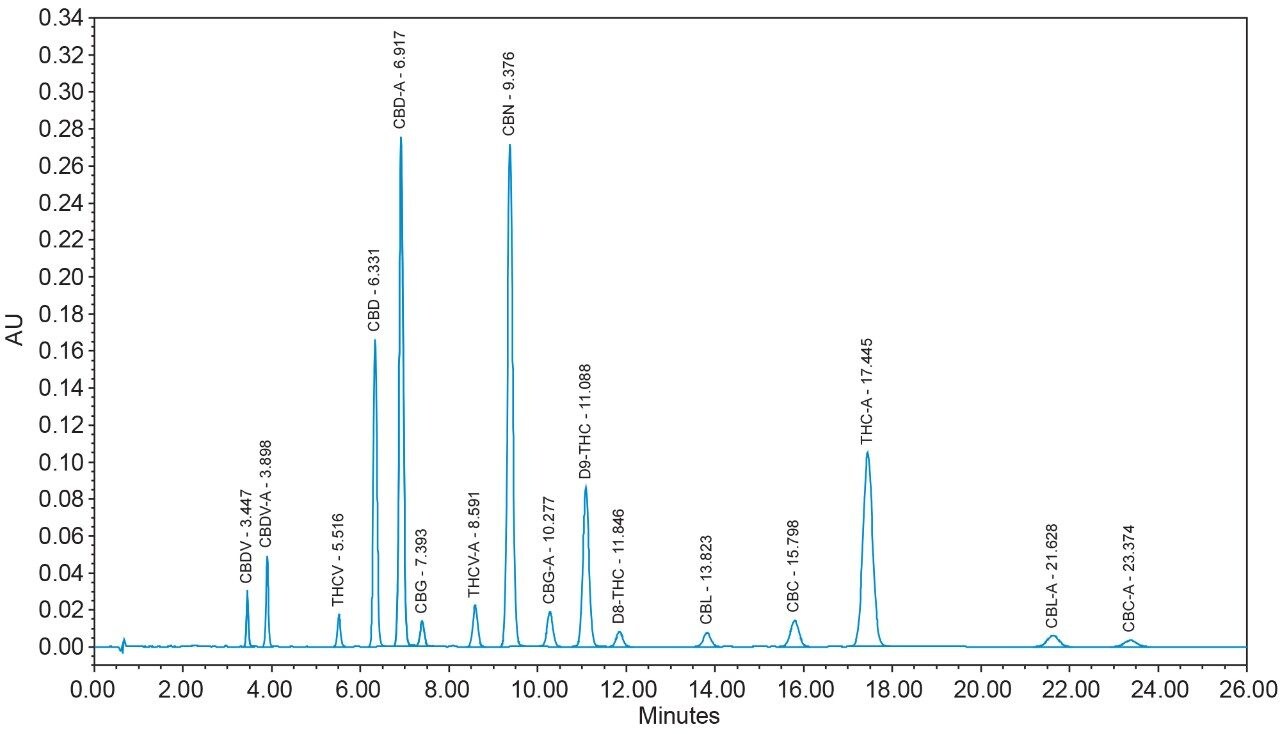
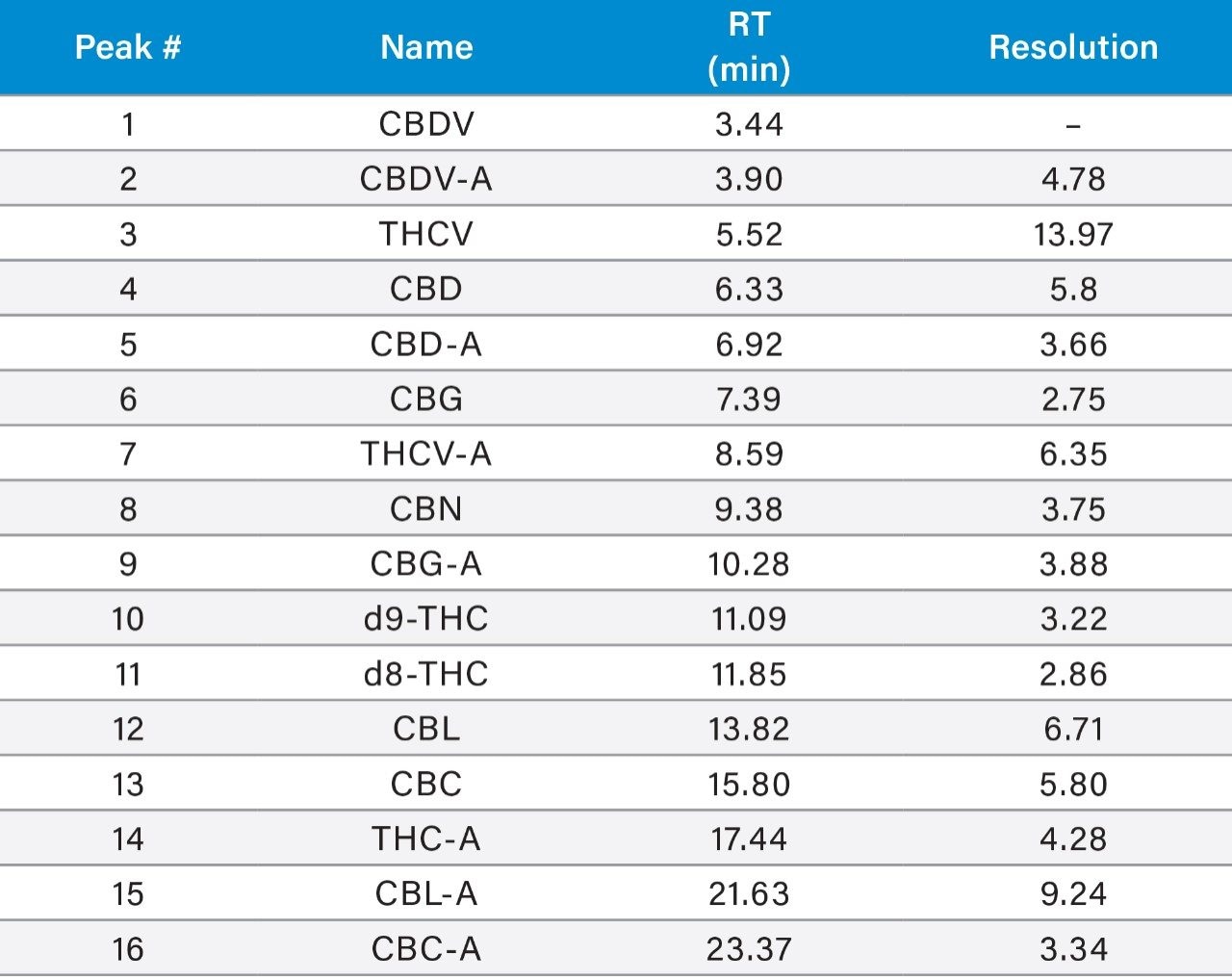
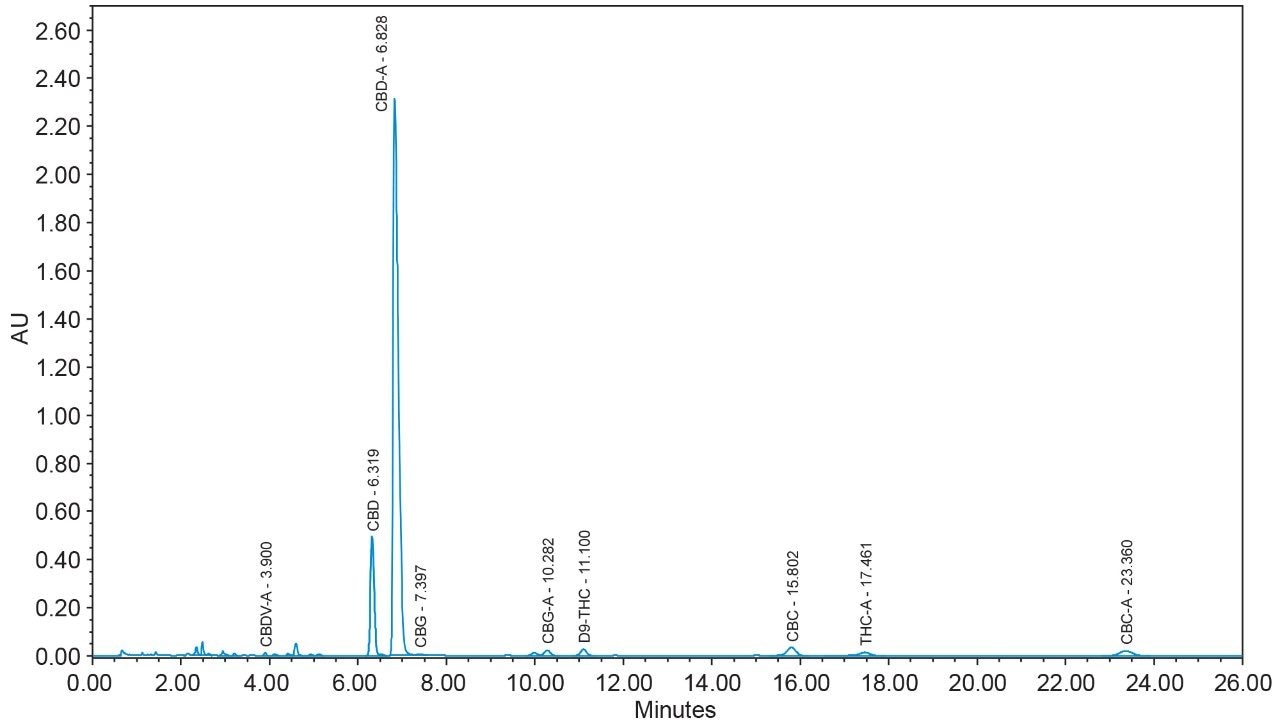
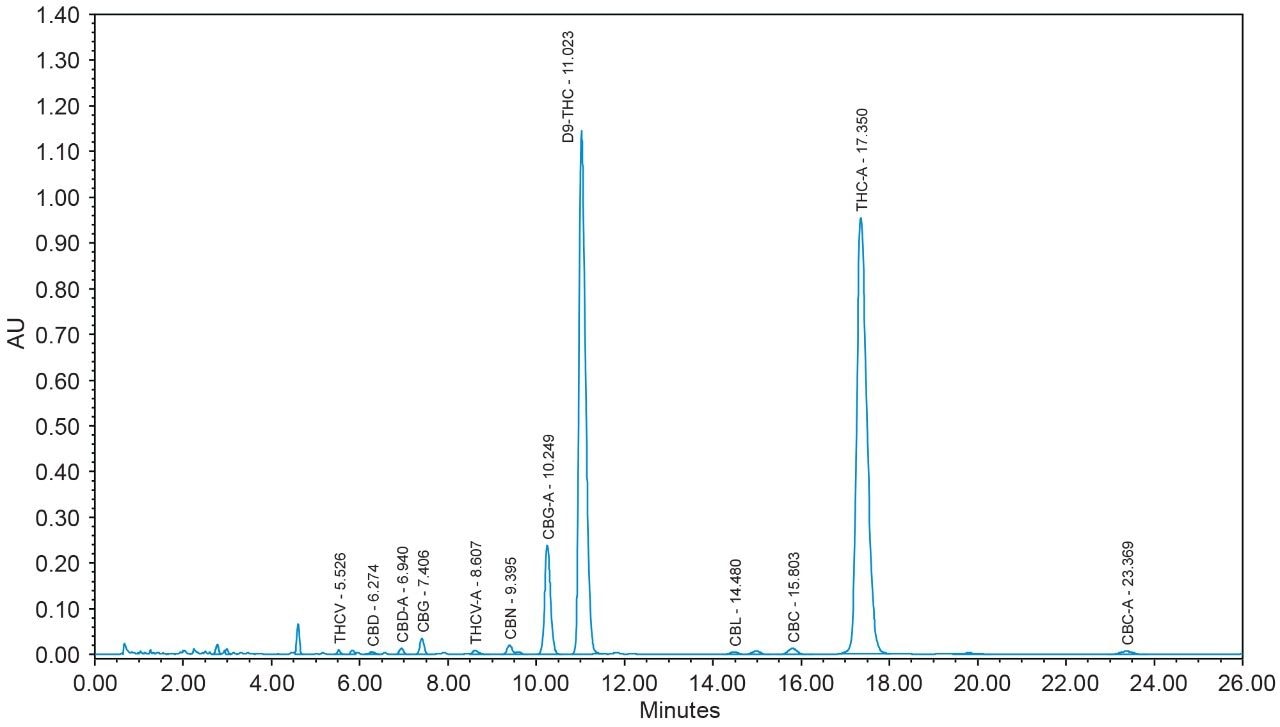
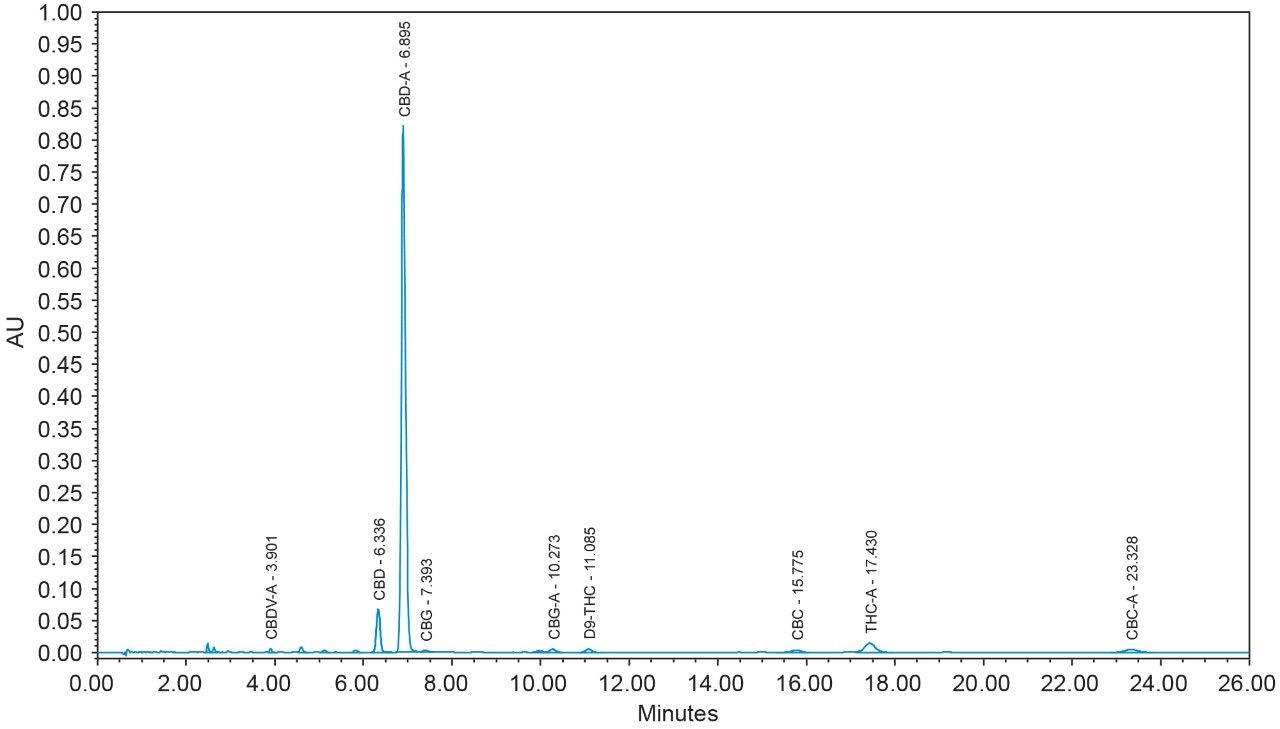
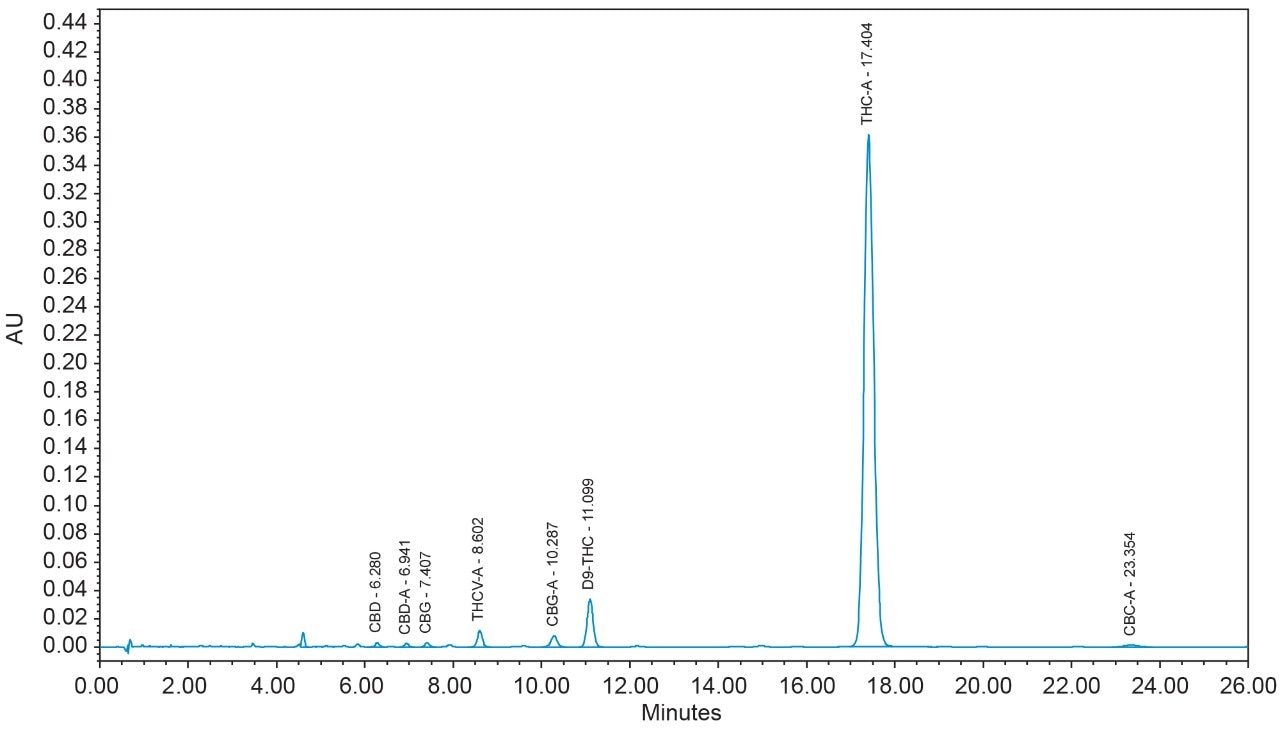
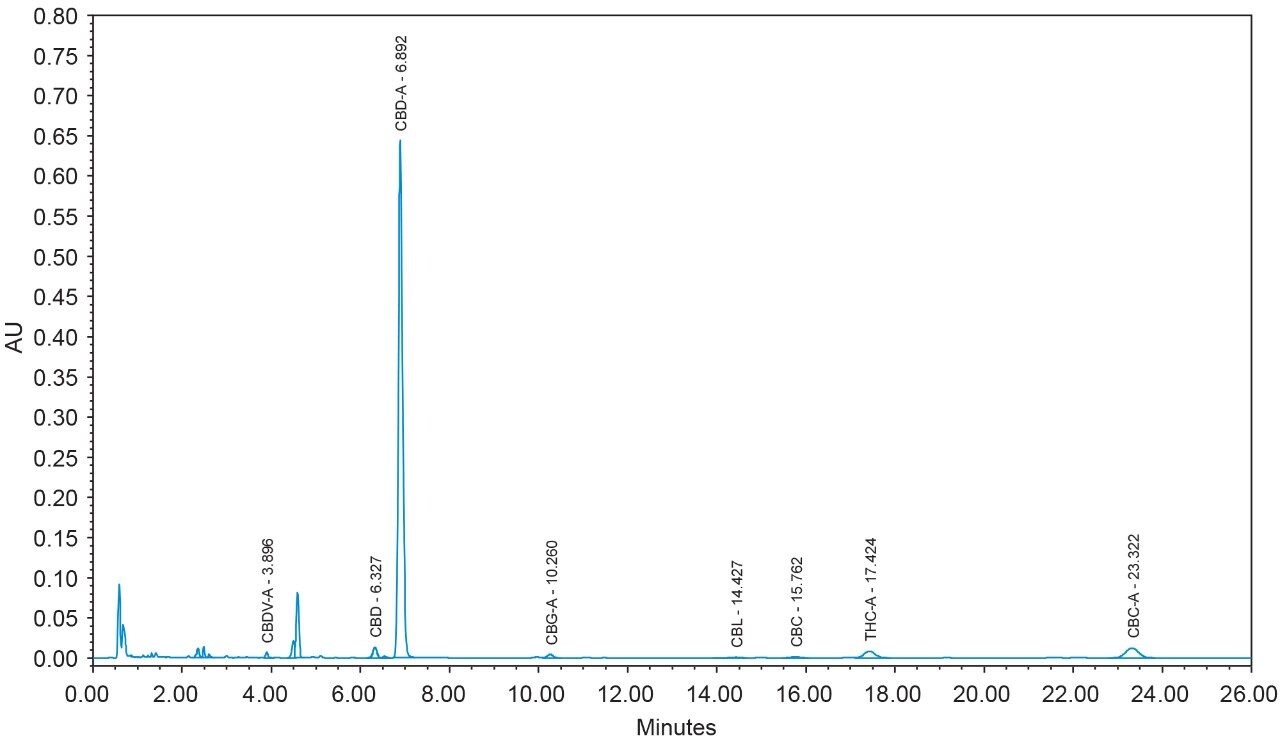
Many HPLC methods have been published as the cannabis industry has continued to expand.4,5,6 These methods are presented using a variety of columns, mobile phase combinations, pH adjustments, and gradient profiles, but more importantly, with only a subset of target cannabinoids. It is possible for any cannabinoid to be present in any cannabis sample. In addition, any cannabis sample can have any cannabinoid as a major or minor constituent; therefore good chromatographic resolution of all cannabinoids of interest is essential. For HPLC methods it is recommended that a resolution of >2 between the peak of interest and the closest potential interfering peak is achieved.7 To ensure the most accurate and reliable results, every effort should be made to separate as many cannabinoids as possible in a given analysis. Further, methods that are simple, with easy to prepare reagents, reduce operator errors which in turn generates higher quality results. In the method presented here, the mobile phase consists of 0.1% trifluoroacetic acid (TFA) in water and acetonitrile which are mixed using the automated solvent blending capability of the Waters Alliance System.8 This significantly reduces variability that can happen when manually preparing mixed mobile phases. Detection is accomplished using a Waters 2998 Photodiode Array (PDA) Detector operated in single wavelength mode (228 nm) for simplicity. Even when operated in this mode, full scan PDA data is collected and is available for library matching or peak purity assessment when desired.9 Separations were achieved using a CORTECS Shield RP18 Column (p/n: 186008685). This solid-core column provides alternative selectivity when compared to typical C18 bonded chemistries, especially for phenolic compounds. The instrument was fully controlled and all data were collected and processed using Empower 3 Chromatography Data Software (CDS).
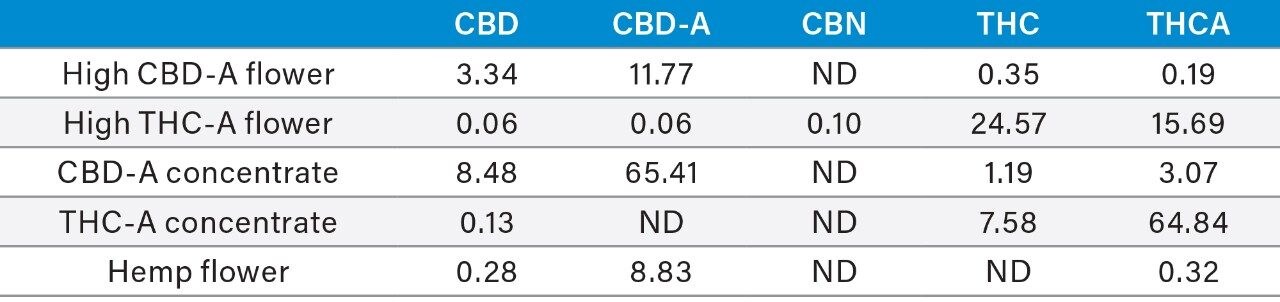
Table 5. Quantitative analysis results of representative samples presented as %dry weight.
ND=Not Detected.
The authors thank ProVerde Labs and Cattis LLC for the donation of the prepared samples.
Waters does not support, encourage or promote the use of its products or services in connection with an illegal use, cultivation, or trade of cannabis or cannabis products. Waters products are intended to be used for cannabis related purposes only in compliance with all applicable laws in a manner that promotes public safety and/or in connection with federally approved research, or state approved medical research.
720006426, November 2018