
This is an Application Brief and does not contain a detailed Experimental section.
This application brief describes a method that employs QuEChERS extraction, combined with Atmospheric Pressure GC (APGC) and Xevo G2-XS QTof high resolution mass spectrometry (HRMS).
QuEChERS extraction, combined with APGC and QTof allows dioxins analysis to be performed without the need for an expert operator, quicker and cheaper than traditional dioxin sample preparation and analysis.
The objective of this work was to develop a dioxin method that was faster and more cost effective than the traditional magnetic sector technique using APGC high resolution mass spectrometry (HRMS) analysis on a Waters Xevo G2-XS QTof while exceeding the minimum performance limits required for EPA method 1613.
Dioxins and dioxin-like compounds are ubiquitous persistent organic pollutants (POPs) linked to various diseases including cancer.1 They are restricted under the Stockholm Convention2 and are monitored for their occurrence and toxicity by regulatory agencies worldwide.
The classical analytical method for testing dioxins in sediment using magnetic sector instruments is considered the “gold” reference standard. However it requires an expert operator and specialized instrumentation.3 Traditional sample preparation times can exceed several days and use a large amount of costly and hazardous solvents.
Since sediment chemistry can vary spatially and temporally, it is necessary to analyze a large number of samples to properly characterize any site being evaluated for dioxin contamination.4 This translates to an extreme expenditure of time for sample prep and massive solvent usage. Within the last decade, a single phase acetonitrile extraction known as QuEChERS (Quick, Easy, Cheap, Effective, Rugged, and Safe) has been employed to prepare food samples for pesticide analysis in as little as 30 minutes.5 This technique was modified and adapted as a rapid extraction and cleanup for the analysis of dioxins and furans in sediment samples and was used in the preparation of samples in this study. In this new approach, samples were investigated using the Xevo G2-XS QTof equipped with Atmospheric Pressure GC (APGC).
A modified QuEChERS sample preparation method for the screening of dioxins and furans in sediment was developed which reduced sample preparation time from 10 samples in four to five days, to as many as 30 samples in one day.6 This study also exploits the use of an APGC source (Figure 1a) coupled to the Xevo G2-XS QTof (Figure 1b) as an alternative to a traditional magnetic sector instrument.

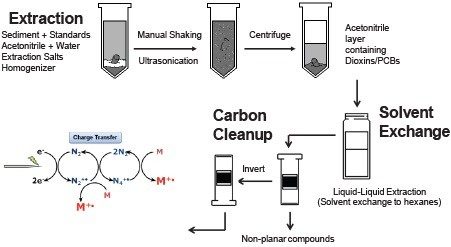
Wet sediment samples were fortified with 13C-labeled standards and extracted using a modified QuEChERS method. The separated organic layer was solvent exchanged to hexane by liquid-liquid extraction. The extract was cleaned by a carbon column and then concentrated for instrumental analysis using a magnetic sector GC HRMS system and a Xevo G2-XS QTof equipped with an APGC source. The column used in this analysis was a Restek Rtx-Dioxin2 at 20 m, 30 m, and 40 m lengths.
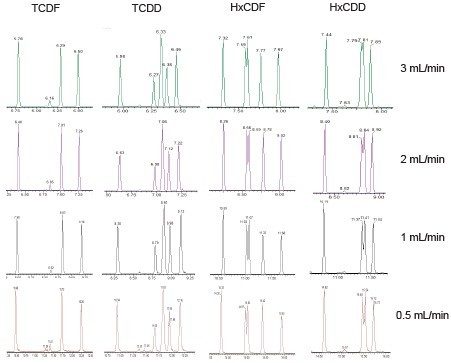
The capabilities and performance of the APGC-Xevo G2-XS QTof proved to be similar or better than the magnetic sector MS for the analysis of dioxins. Unlike conventional EI (electron ionization) systems, the APGC source allows for higher flow rates to improve analysis times. The effect of increased flow rate on the chromatographic resolution for four different congener classes are shown in Figure 3. Although chromatographic resolution decreases with increased column flow rates, adequate separation is maintained for quantitative analysis, in large part due to the selectivity of the stationary phase (Rtx-Dioxin2). Only the pair of HxCDD congeners appear to co-elute, but given their identical TEFs (toxic equivalency factors), the impact of the reduced chromatographic resolution on TEQ (toxic equivalency quantity) is expected to be negligible.
Column length was also evaluated and the results are summarized in Figure 4. Shorter columns reduced backpressure, resulting in higher flow rates, and further reduced runtimes (<15 min/sample) with minimal loss in separation. APGC is sufficiently versatile to provide ultimate chromatographic performance (using a 40 m Rtx-Dioxin2 column at optimum flows) that satisfies the regulation, despite the method requirement for EI ionization and magnetic sector MS. When needed, high throughput and increased capacity is possible (using a 20 m Rtx-Dioxin2 column at flows >3 mL/min) while preserving separation of critical isomers.
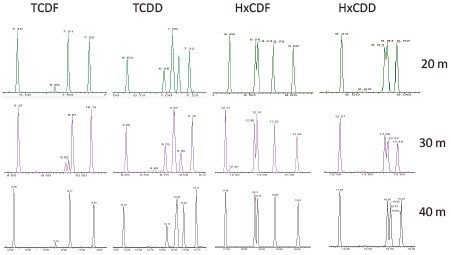
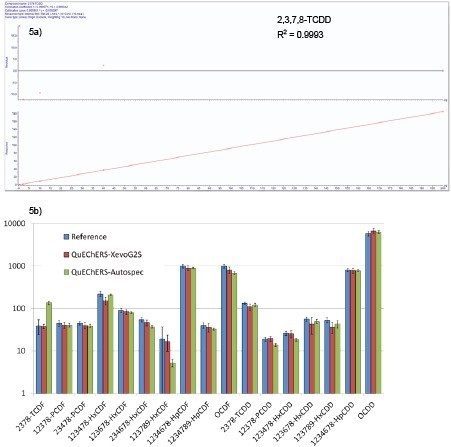
Figure 5a shows results from a calibration curve of 2,3,7,8-TCDD from 0.5 to 200 pg with good linearity and R2 of 0.9993 using APGC-Xevo G2-XS QTof.
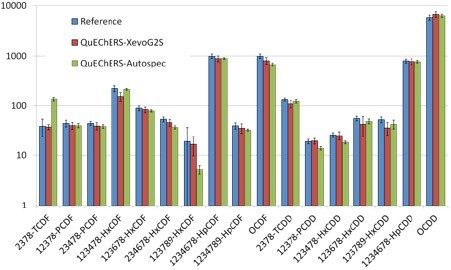
Shown in Figure 5b are the results of the certified reference material compared to the APGC-Xevo G2-XS and magnetic sector. Results from the APGC-Xevo G2-XS compare favorably to the reference and the magnetic sector. It is worth noting, however, that the magnetic sector results for 2,3,7,8-TCDD and 1,2,3,7,8,9-HxCDF differ from the reference while the APGC results for those same congeners compare more favorably to the reference (Figure 5b).
QuEChERS has been proven to be an effective sample extraction/clean-up method for the analysis of a large number of sediment samples from site remediation activities,7 hence reducing the time and solvent as compared to the classic preparation. APGC along with the Xevo G2-XS QTof decreased instrumental run time due to its ability to handle higher flow rates than the GC-HRMS system. The combined method of QuEChERS extraction with APGC-QTof analysis provided a sample throughput increase of 15x over traditional techniques. The Xevo G2-XS QToF offers a flexible platform with inlet options including APGC, ESI, APCI, and UniSpray to name a few, thus permitting the instrument to perform other analysis when needed. It can operate in a non-targeted acquisition mode that can meet the limits of detection of dioxin regulatory method EPA1613, and can provide additional analytical information such as elemental composition on non-target analytes that can be encountered with both classical and generic sample preparation approaches.
720006099, October 2017