
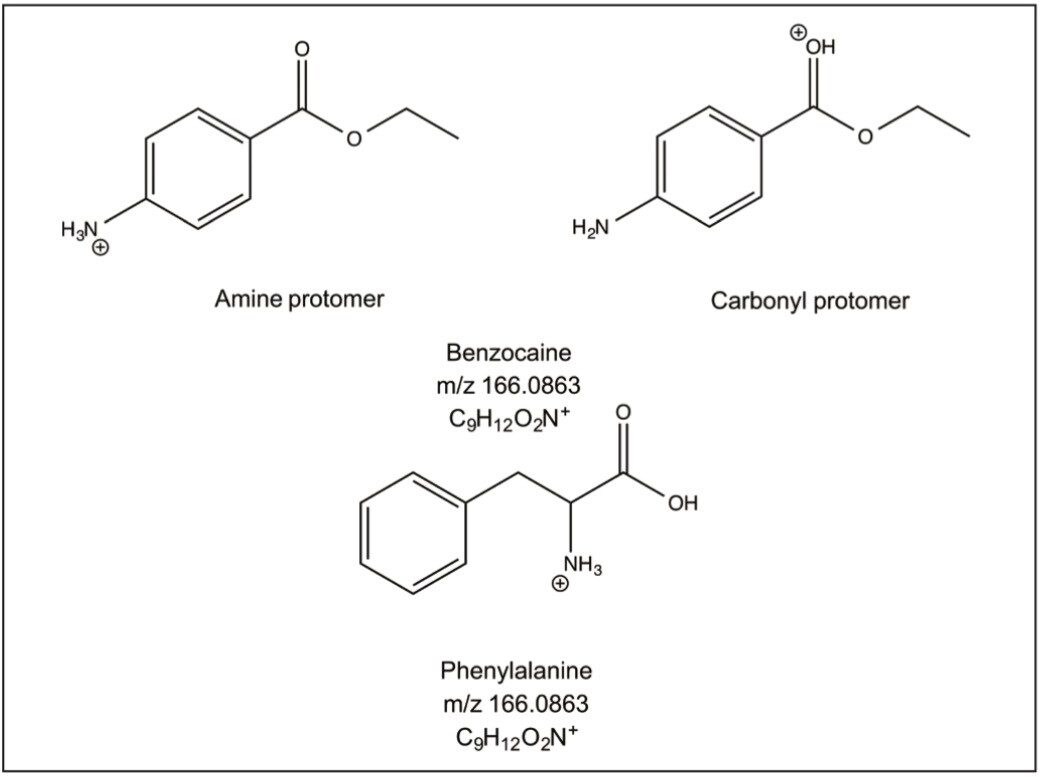
In this application note, the SYNAPT G2-Si HDMS Travelling-Wave (T-Wave) Ion Mobility Mass Spectrometer (IMMS) has been used to separate the structural isomers benzocaine and phenylalanine.
As part of the newborn metabolic screening protocol, the early identification of babies at risk of a metabolic disorder allows for the correct medical treatment and management of selected inherited disorders. A particularly effective method for the analysis of certain metabolites from dried blood spots, which may indicate a metabolic disorder, is electrospray ionisation combined with tandem mass spectrometry.1 A number of inherited diseases result in the disruption of the metabolism and transformation of amino acids.
For example, approximately 1 in 12,000 children in the UK is born with an inherited metabolic disorder called phenylketonuria (PKU). PKU, a deficiency in phenylalanine hydroxylase inhibits the conversion of phenylalanine to tyrosine, with an accumulation of phenylalanine and a decrease in tyrosine. Thus, the phenylalanine to tyrosine ratio is useful in diagnosing PKU. If phenylketonuria is not detected and treated, the levels of phenylalanine in the child’s blood will increase resulting in brain damage and mental retardation.
Any change in the way blood samples are collected prior to submission for MS analysis can adversely affect the measured concentrations of the targeted metabolites being measured. This was recently highlighted in the screening protocol for PKU whereby the hospital reported an increase in PKU abnormal.1 The abnormally high levels were due to a contamination issue that was identified as an interference with benzocaine. The midwives used alcohol swabs which contained benzocaine to clean the skin prior to taking a blood spot via a heel prick of the newborns. Benzocaine is a structural isomer of phenylalanine and confirmed as the source of the PKU false positives.
The SYNAPT G2-Si HDMS Travelling-Wave (T-Wave) Ion Mobility Mass Spectrometer (IMMS) has been used to separate the structural isomers benzocaine and phenylalanine. Benzocaine has two protonation site isomers (protomers) that are detected at different mobility drift times to one another.2,3 The individual drift times of these protomers are importantly different to the ion mobility of phenylalanine.
Benzocaine (Sigma-Aldrich, St Louis, MO) and Phenylalanine (Sigma-Aldrich) stock solutions were prepared at 1 mg/mL and 0.4 mg/mL (MeOH) respectively. The samples were further diluted to 0.5 ng/μL (MeOH) and mixed prior to analysis by direct infusion MS.
|
MS system: |
SYNAPT G2-Si |
|
Ionization mode: |
ESI positive |
|
Capillary voltage: |
2.5 kV |
|
Cone voltage: |
20 V |
|
Source temp.: |
110 °C |
|
Desolvation temp.: |
250 °C |
The ion signal was attenuated such that only ~5% of the ions were transmitted to the ion mobility cell.
|
N2 parameters |
|
|---|---|
|
T-Wave ion mobility cell: |
N2 pressurized at a flow rate of 90 mL/min |
|
T-Wave velocity: |
750 m/s |
|
T-Wave amplitude: |
27 V |
|
CO2 parameters |
|
|
T-Wave ion mobility cell: |
CO2 pressurized at a flow rate of 65 mL/min |
|
T-Wave velocity: |
1000 m/s |
|
T-Wave amplitude: |
40 V |
Benzocaine has two protomers that are detected at different mobility drift times, see Figure 1. The individual drift times of these protomers are importantly different to the ion mobility of phenylalanine. Protonation on either the amine nitrogen or the carbonyl oxygen generates protonation site isomers that interact differently with the mobility drift gas.
As a consequence of the different sites of protonation, infusion of benzocaine into the T-Wave ion mobility section of the instrument results in two resolved ion mobility peaks detected at different drift times (data not shown). Addition of the structural isomer phenylalanine to a solution of benzocaine results in three components being separated and detected using ion mobility as shown in Figure 2. The drift times of the mobility separated m/z 166 ions are shown using N2 mobility drift gas (Figure 2A) and CO2 mobility drift gas (Figure 2B).
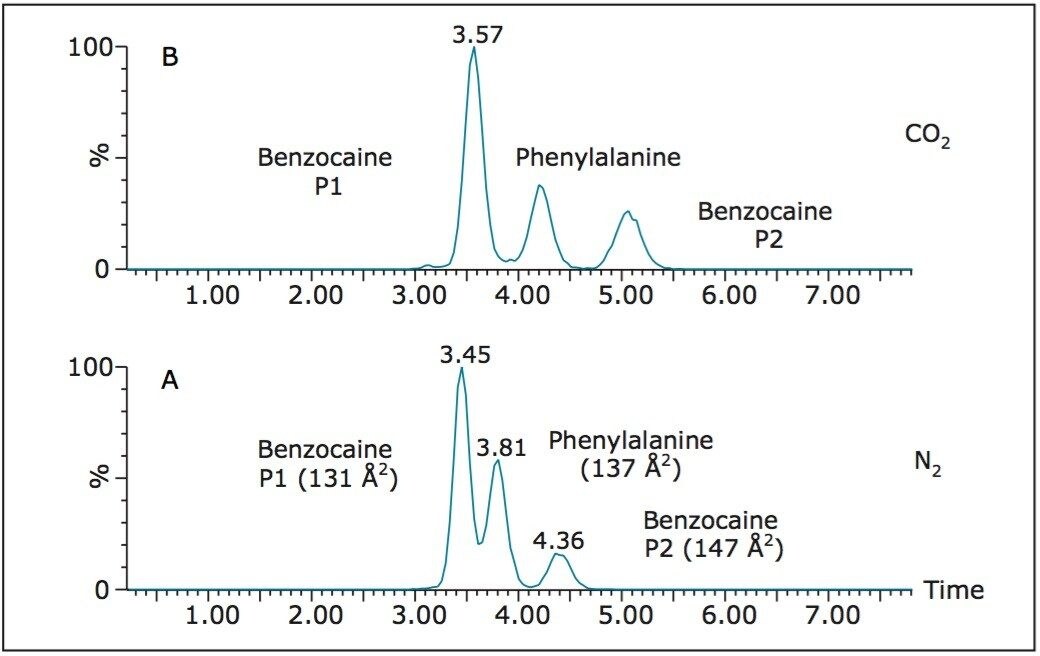
The T-Wave ion mobility rotationally-averaged collision cross-sections measured in N2 for phenylalanine, and the protomers of benzocaine were 137 Å2, 131 Å2, and 147 Å2, respectively. The results show that one of the protomers of benzocaine has a much greater interaction with the mobility gases of N2 and CO2 than the other protomer and phenylalanine. Following mobility separation, the ion species were fragmented by applying a potential difference of 15 V between the mobility and transfer T-Wave section of the instrument. By combining the mass spectra over the individual peaks in the drift times, tandem mass spectra were independently obtained. Figure 3A shows the MS/MS spectrum of the peak centred around 3.45 ms for one of the protomers of benzocaine. Figure 3B shows the MS/MS spectrum of the second peak centred around 4.36 minutes for the other protomer of benzocaine. Figure 3C shows the MS/MS spectrum of the peak centred around 3.81 minutes and is consistent with fragmentation of phenylalanine. The resulting product ions obtained, and abundances for fragmentation of benzocaine, are due to different fragmentation pathways as a direct result of the different positions of protonation, as shown in Figure 3.
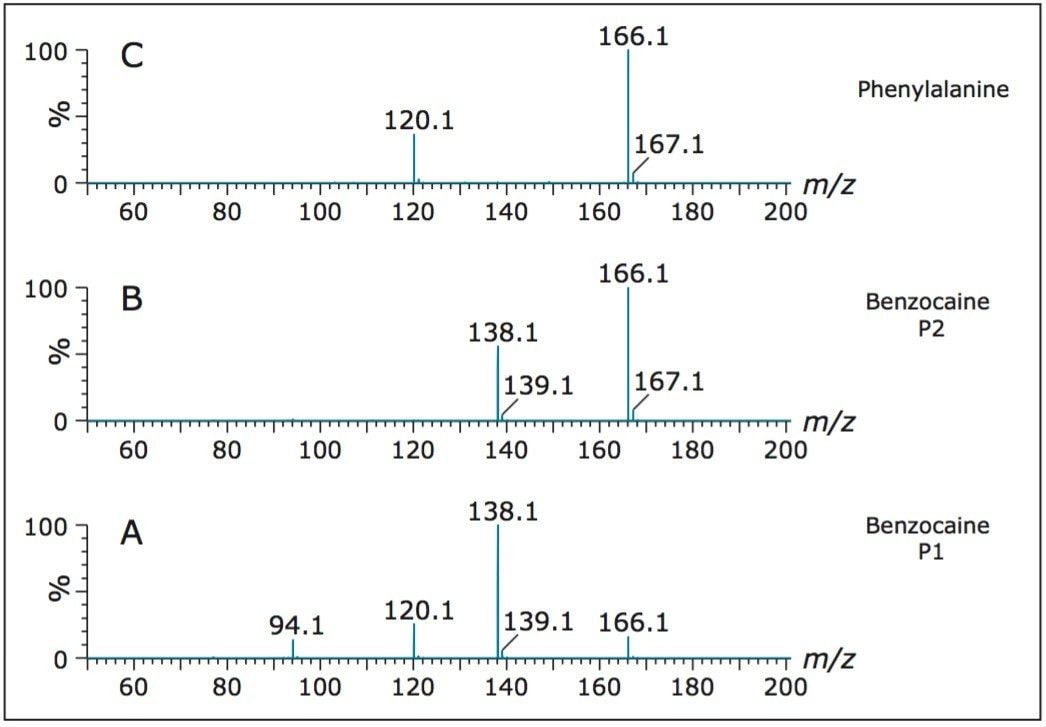
The resulting product ions obtained, and abundances for fragmentation of benzocaine, are due to different fragmentation pathways as a direct result of the different positions of protonation. Both protomers produce a fragment at m/z 138. Both protomers primarily fragment by the neutral loss of C2H4 (ethene). The protomer with the fastest drift time shows two other abundant product ions at m/z 120 and m/z 94. Accurate mass measurement confirms these ions to be loss of water from m/z 138 and formation of the the anilinium cation. These product ions suggest protonation on the carbonyl oxygen and not the amide nitrogen
It can be clearly seen that the separation for this simple mixture, although adequate in N2, is superior in CO2. This application demonstrates the utility of the ion mobility-MS/MS assay, which is particularly relevant for further multiple reaction monitoring bio-analytical method development in the absence or presence of benzocaine. This will not only have important consequences for the quantitative Phe/Tyr ratio analysis, but the combination of ion mobility to screening protocols will augment other metabolic studies and applications whereby interferences may be present.
720005367, April 2015