
The purpose of this application note is to demonstrate the CORTECS UPLC HILIC Column for determination of highly polar basic compounds. In this application, a number of these compounds are determined in a spiked river water sample. Sample preparation was performed using the EPA 1694 method modified for use with a smaller sample volume (100 mL rather than 1000 mL) and a smaller Oasis HLB Cartridge (150 mg compared with 1000 mg). The same enrichment factor was maintained and similar performance was observed compared with the protocol given in method 1694.1 Sample preparation time was reduced from about 4 hours to about 1 hour.
Highly polar basic compounds, such as albuterol, can be determined in environmental water samples by ion-pairing reversed-phase liquid chromatography or by HILIC (Hydrophilic Interaction LIquid Chromatography). Although volatile ion-pairing reagents are available for use with LC-MS, significant reduction in MS response is usually observed compared with response with no ion-pairing reagent. Therefore the HILIC approach with no ion-pairing reagents is preferred for trace-level LC-MS analysis.
The purpose of this application note is to demonstrate the CORTECS UPLC HILIC Column for determination of highly polar basic compounds. In this application, a number of these compounds are determined in a spiked river water sample. Sample preparation was performed using the EPA 1694 method modified for use with a smaller sample volume (100 mL rather than 1000 mL) and a smaller Oasis HLB Cartridge (150 mg compared with 1000 mg). The same enrichment factor was maintained and similar performance was observed compared with the protocol given in method 1694.1 Sample preparation time was reduced from about 4 hours to about 1 hour.
|
System: |
ACQUITY UPLC |
|
Column: |
CORTECS UPLC HILIC, 2.1 x 100 mm, 1.6 µm |
|
Injection volume: |
30 µL |
|
Temperature: |
30 °C |
|
Mobile phase A: |
Ammonium Acetate Buffer (18 mM acetic acid/13 mM ammonium acetate) |
|
Mobile phase B: |
Acetonitrile |
|
Flow rate: |
0.45 mL/min |
|
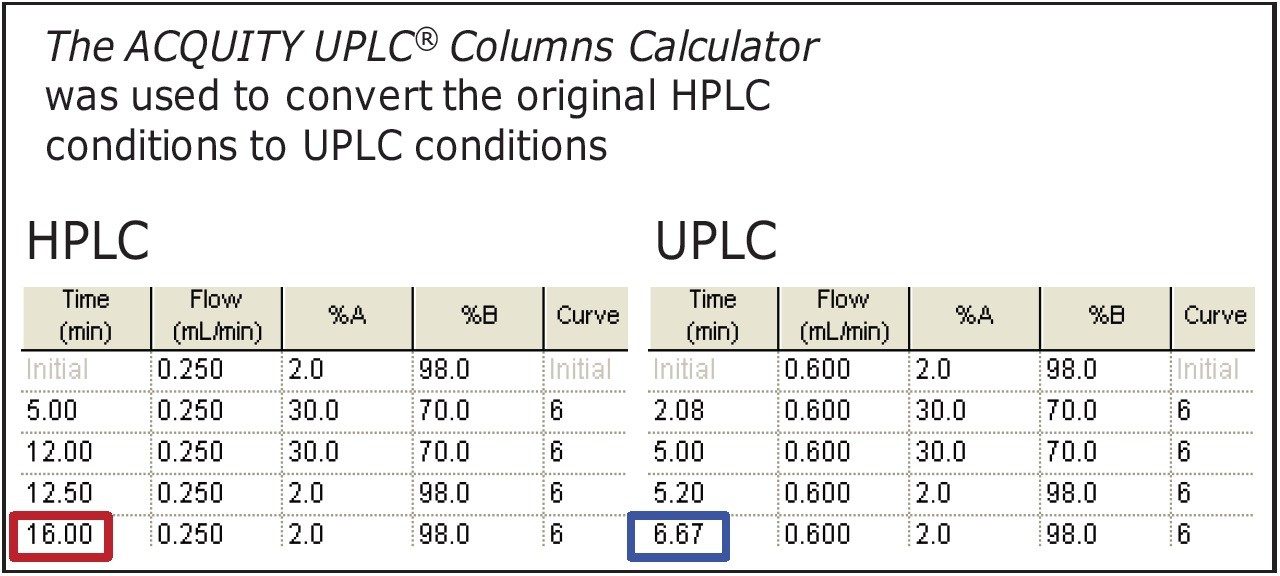
Gradient: |
See Table 1 |
|
Instrument: |
Xevo TQ MS |
|
Mode: |
Electrospray positive (ES+) |
|
Source temp.: |
150 °C |
|
Desolvation temp.: |
500 °C |
|
Desolvation gas: |
1200 L/hr |
|
Collison gas (Argon): |
0.18 mL/min |
|
Cone gas: |
30L/hr |
|
Compound |
MRM(m/z) |
Cone(V) |
Collision(eV) |
|---|---|---|---|
|
Cimitidine |
253.0>116.9 |
30 |
20 |
|
253.0>158.9 |
30 |
15 |
|
|
Clenbuterol |
277.0>167.9 |
24 |
26 |
|
277.0>202.9 |
24 |
18 |
|
|
Albuterol |
240.2>148.0 |
28 |
15 |
|
240.2>222.0 |
28 |
10 |
|
|
Metformin |
130.9>59.9 |
25 |
15 |
|
130.9>71.9 |
25 |
20 |
|
|
Ranitidine |
315.0>129.9 |
25 |
25 |
|
315.0>175.9 |
25 |
20 |
The surface water sample (100 mL taken from a local river) was adjusted to pH 11 with concentrated ammonium hydroxide. An Oasis HLB Cartridge (6 cc, 150 mg) was conditioned with 2 mL methanol and equilibrated with 2 mL pH 11 water. The sample was loaded at approximately 5 mL/min using a vacuum manifold. After loading, the cartridge was washed with 2 mL of pH 11 water and was eluted with 1 mL methanol followed by 2 mL of 2% formic acid in methanol. The eluent was evaporated and reconstituted in 0.4 mL 20:80 methanol/acetonitrile.
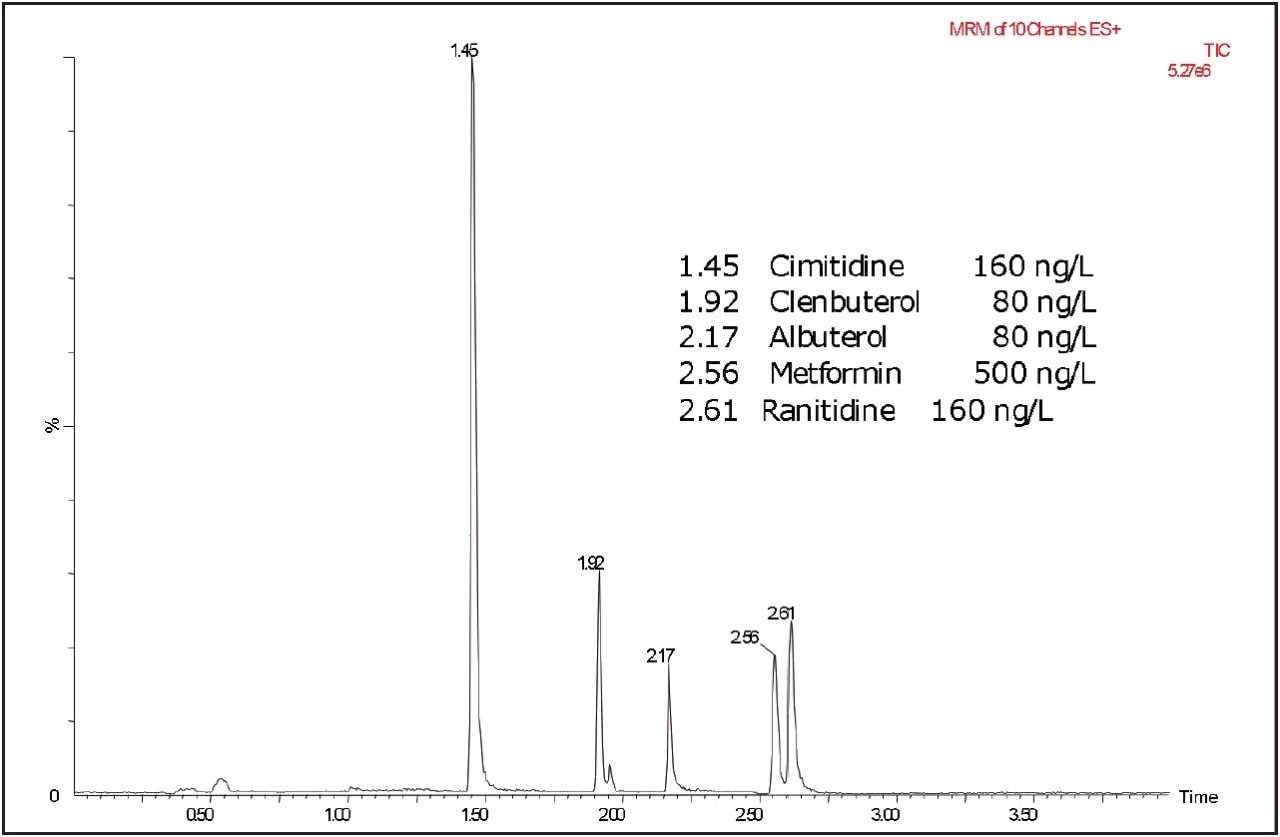
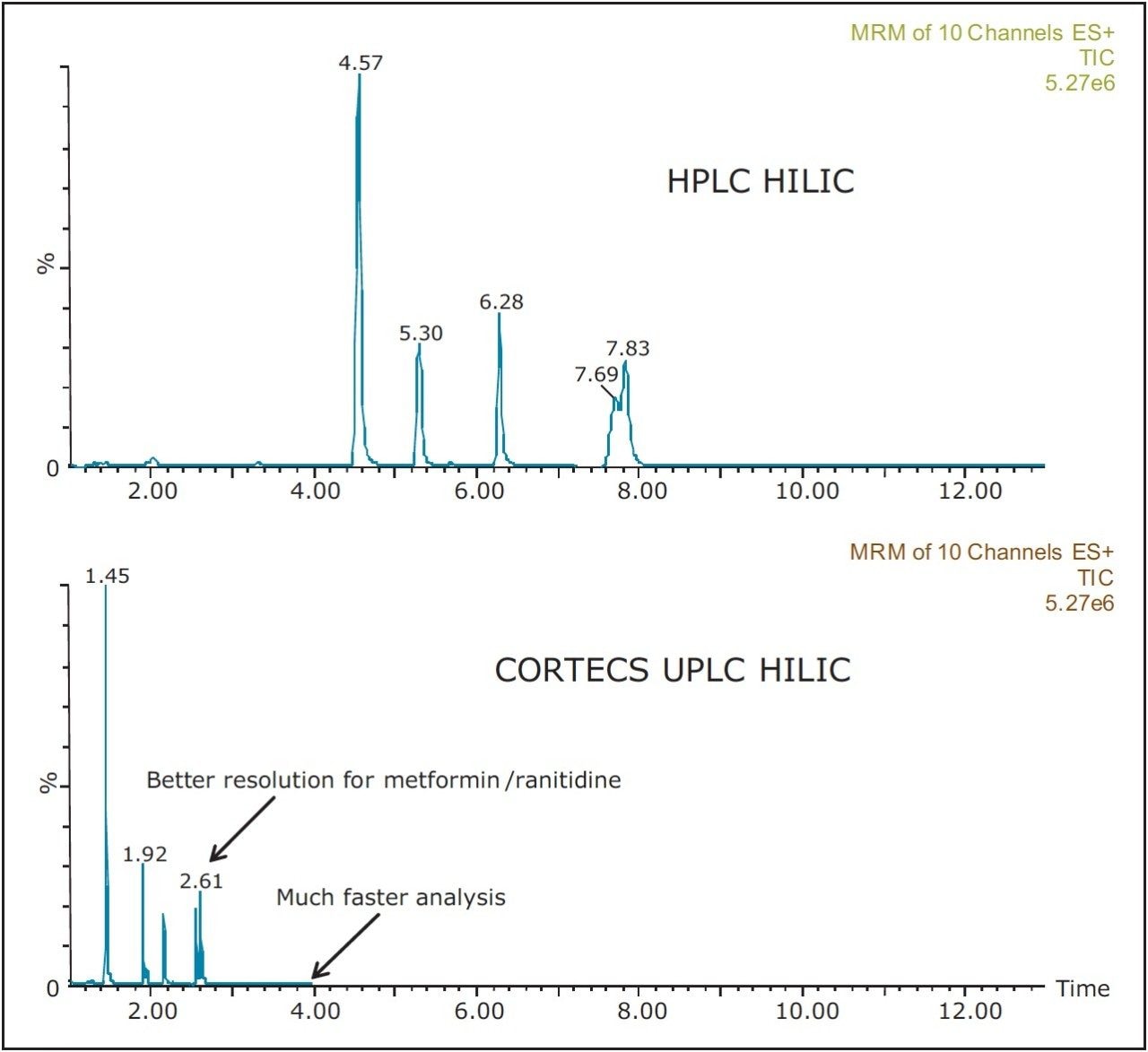
Figure 1 shows an LC-MS/MS chromatogram obtained from a spiked sample of river water. Figure 2 shows a comparison of the same sample analyzed by UPLC and by HPLC. The CORTECS UPLC HILIC Column gave much improved resolution for metformin and ranitidine in less than half the time.
720005141, August 2014